What is Life? Project PART 6: The molecules of life
... Proteins are an important molecular component of all living things. Your body contains tens of thousands of different proteins. Each protein has a unique three-dimensional shape that corresponds to a specific function. Proteins make up many of the structures in your body; every time you look at your ...
... Proteins are an important molecular component of all living things. Your body contains tens of thousands of different proteins. Each protein has a unique three-dimensional shape that corresponds to a specific function. Proteins make up many of the structures in your body; every time you look at your ...
NUTRIENT Handout
... All of the nutrients fit into one of these classes. Sometimes the things we ANALYZE, however, are not so clear cut. For example, we don't analyze just for "carbohydrates" because some of the carbohydrates are very digestible and some are very indigestible. For purposes of ANALYSIS, we often use a ve ...
... All of the nutrients fit into one of these classes. Sometimes the things we ANALYZE, however, are not so clear cut. For example, we don't analyze just for "carbohydrates" because some of the carbohydrates are very digestible and some are very indigestible. For purposes of ANALYSIS, we often use a ve ...
Unit 2 PPT - Faculty Sites
... chain owing to bend-producing amino acids and interactions among R groups far apart on the chain. 4. Quaternary structure is the linkage of two or more polypeptide chains by hydrogen bonds and other interactions. ...
... chain owing to bend-producing amino acids and interactions among R groups far apart on the chain. 4. Quaternary structure is the linkage of two or more polypeptide chains by hydrogen bonds and other interactions. ...
2.3: Carbon-Based Molecules
... Structure of Lipids • Fatty acids bonded to glycerol. – Glycerol-3 hydroxyl groups – Fatty acids-long carbon chains Two different types of fatty acids found in fats and oils: oSaturated fatty acids oUnsaturated fatty acids ...
... Structure of Lipids • Fatty acids bonded to glycerol. – Glycerol-3 hydroxyl groups – Fatty acids-long carbon chains Two different types of fatty acids found in fats and oils: oSaturated fatty acids oUnsaturated fatty acids ...
Biological Molecules
... properties d. Hydrogen bonding between cellulose molecules forms bundles called microfibrils that reinforce the cell wall e. Can not be digested by animals; no enzyme to digest the 1-4 linkage ...
... properties d. Hydrogen bonding between cellulose molecules forms bundles called microfibrils that reinforce the cell wall e. Can not be digested by animals; no enzyme to digest the 1-4 linkage ...
Ch 3 organic molecules
... most carnivores have not what do they eat? How is this related to corn fuels?? ...
... most carnivores have not what do they eat? How is this related to corn fuels?? ...
Chemistry of Proteins Model Making
... Proteins are the main structural and growth components of cells in tissues such as skin, hair, muscle and blood. Other proteins serve in regulatory capacity as enzymes and hormones. Proteins always contain nitrogen in addition to carbon, hydrogen and oxygen. Phosphorus and sulfur are also found in m ...
... Proteins are the main structural and growth components of cells in tissues such as skin, hair, muscle and blood. Other proteins serve in regulatory capacity as enzymes and hormones. Proteins always contain nitrogen in addition to carbon, hydrogen and oxygen. Phosphorus and sulfur are also found in m ...
Answer
... 47. Are lipids polar or nonpolar? What happens to lipids when they are placed in water? Non polar, they separate from water 48. Compared to carbohydrates, what is true about the ratio of carbon & hydrogen atoms to oxygen atoms? If a compound has more bonds, what can it store more of in those bonds? ...
... 47. Are lipids polar or nonpolar? What happens to lipids when they are placed in water? Non polar, they separate from water 48. Compared to carbohydrates, what is true about the ratio of carbon & hydrogen atoms to oxygen atoms? If a compound has more bonds, what can it store more of in those bonds? ...
Nutrients - HRSBSTAFF Home Page
... that help to maintain homeostasis are referred to as homeostatic mechanisms. ...
... that help to maintain homeostasis are referred to as homeostatic mechanisms. ...
GLYCOLYSIS and respiration review worksheet
... 3. What molecule actually enters the Krebs cycle, serving as a common link for the breakdown of not only sugars but also fats and amino acids? (HINT: Next step after pyruvate...) ...
... 3. What molecule actually enters the Krebs cycle, serving as a common link for the breakdown of not only sugars but also fats and amino acids? (HINT: Next step after pyruvate...) ...
CS4030: Tutorial 1- Biological Issues (from Bioinformatics ch 1)
... 2. Diagram the ”Central Dogma” of molecular biology complete with labels that indicate the portions that correspond to transcription and translation and indicate what enzymes are responsible for those important steps. 3. Examine the chemical structures of the amino acid R groups shown in figure 1.5b ...
... 2. Diagram the ”Central Dogma” of molecular biology complete with labels that indicate the portions that correspond to transcription and translation and indicate what enzymes are responsible for those important steps. 3. Examine the chemical structures of the amino acid R groups shown in figure 1.5b ...
Food - cbbiology
... Phospholipid: a lipid where one of the fatty acids have been replaced with a phosphate group or has a phosphate group added to it Sources of lipids: butter, oils, margarine, cream ...
... Phospholipid: a lipid where one of the fatty acids have been replaced with a phosphate group or has a phosphate group added to it Sources of lipids: butter, oils, margarine, cream ...
Biology Organic Molecules Notes
... SPI3210.1.3 – I can distinguish among proteins, carbohydrates, lipids, and nucleic acids. SPI3210.1.4 – I can identify positive tests for carbohydrates, lipids, and proteins. ...
... SPI3210.1.3 – I can distinguish among proteins, carbohydrates, lipids, and nucleic acids. SPI3210.1.4 – I can identify positive tests for carbohydrates, lipids, and proteins. ...
Macromolecule Basics
... The Basic Molecules • Carbohydrates • This is the most common organic molecule • It makes up most plant matter • They are made from carbon, hydrogen and oxygen • Their building block is a single sugar called a monosaccharide (mono = single) • When 2 sugars combine it is called a disaccharide (di = ...
... The Basic Molecules • Carbohydrates • This is the most common organic molecule • It makes up most plant matter • They are made from carbon, hydrogen and oxygen • Their building block is a single sugar called a monosaccharide (mono = single) • When 2 sugars combine it is called a disaccharide (di = ...
Chapter 2
... • Molecules that consist of multiple monomers • Carbohydrates, lipids, proteins, nucleic acids ...
... • Molecules that consist of multiple monomers • Carbohydrates, lipids, proteins, nucleic acids ...
Respiratory Substrates
... carried to Krebs cycle • Some can enter Krebs directly • Number of hydrogen atoms per mole accepted by NAD then used in electron transport chain is slightly more than the number of hydrogen atoms per mole of glucose, so proteins release slightly more energy than equivalent masses of glucose ...
... carried to Krebs cycle • Some can enter Krebs directly • Number of hydrogen atoms per mole accepted by NAD then used in electron transport chain is slightly more than the number of hydrogen atoms per mole of glucose, so proteins release slightly more energy than equivalent masses of glucose ...
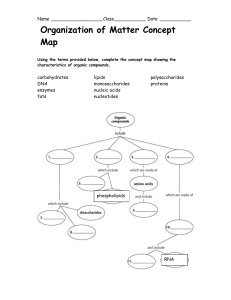
Polymers vs. monomers wkst. and concept map
... Name __________________ Class ___________ Date ___________ 7. Match the POLYmer on the left to the macromolecules on the right. ...
... Name __________________ Class ___________ Date ___________ 7. Match the POLYmer on the left to the macromolecules on the right. ...
Cellular, Element, and Molecular Building Blocks of Living Systems
... • Molecular Bonding: occur between molecules and weaker than covalent bonding. • Hydrogen Bonding: Partially positive hydrogen atom in a polar molecule is attracted to a slightly negative atom (O, N, or F) • van der Waals interactions: Temporary and unequal electron distributions around atoms. ...
... • Molecular Bonding: occur between molecules and weaker than covalent bonding. • Hydrogen Bonding: Partially positive hydrogen atom in a polar molecule is attracted to a slightly negative atom (O, N, or F) • van der Waals interactions: Temporary and unequal electron distributions around atoms. ...
Bell Work 3-19-12 - Science is a Blast
... • It allows reactions to happen in environments that would not normally result in a timely ...
... • It allows reactions to happen in environments that would not normally result in a timely ...
ANPS 019 Beneyto-Santonja 08-29
... Water molecules and solutions The polar water molecule is attracted to any solute with charged atoms: ions, proteins, carbohydrates, etc. Substances that interact readily with water are hydrophilic most substances Substances that do not interact with water hydrophobic lipid molecules Organ ...
... Water molecules and solutions The polar water molecule is attracted to any solute with charged atoms: ions, proteins, carbohydrates, etc. Substances that interact readily with water are hydrophilic most substances Substances that do not interact with water hydrophobic lipid molecules Organ ...
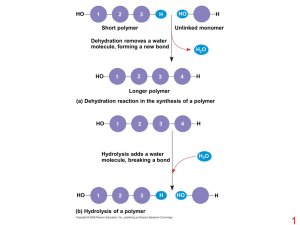
Organic Compounds
... carbohydrates (sugars and starches), lipids (fats), proteins, and nucleic acids (DNA and RNA). • These molecules are usually in the form of polymers, long chains of similar subunits. Because they are large, these molecules are called macromolecules. The subunits are called monomers. • The cell also ...
... carbohydrates (sugars and starches), lipids (fats), proteins, and nucleic acids (DNA and RNA). • These molecules are usually in the form of polymers, long chains of similar subunits. Because they are large, these molecules are called macromolecules. The subunits are called monomers. • The cell also ...
Topic 3.2: Carbohydrates, Lipids, and Proteins
... in animals, and of fructose, sucrose, and cellulose in plants • 3.2.5: Outline the role of condensation and hydrolysis in the relationships between monosaccharides, disaccharides and polysaccharides; between fatty acids, glycerol and triglycerides; and between amino acids and polypeptides • 3.2.6: S ...
... in animals, and of fructose, sucrose, and cellulose in plants • 3.2.5: Outline the role of condensation and hydrolysis in the relationships between monosaccharides, disaccharides and polysaccharides; between fatty acids, glycerol and triglycerides; and between amino acids and polypeptides • 3.2.6: S ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.