Quale Vita? - uniroma1.it
... constant, in some cases they do not make H bonds with solutes, in other cases they attack the organic material (e.g. ammonia) ...
... constant, in some cases they do not make H bonds with solutes, in other cases they attack the organic material (e.g. ammonia) ...
Slide 1
... 1. An atom is the smallest basic unit of matter 2. An element is one type of atom 3. An atom has a nucleus and electrons ...
... 1. An atom is the smallest basic unit of matter 2. An element is one type of atom 3. An atom has a nucleus and electrons ...
Slide ()
... cytosine; G = guanine; T = thymine; U = uridine (replaces thymine in RNA). In RNA, adenine is complementary to thymine of DNA; uridine is complementary to adenine of DNA; cytosine is complementary to guanine and vice versa. “Stop” = peptide chain termination. The three-letter and singleletter abbrev ...
... cytosine; G = guanine; T = thymine; U = uridine (replaces thymine in RNA). In RNA, adenine is complementary to thymine of DNA; uridine is complementary to adenine of DNA; cytosine is complementary to guanine and vice versa. “Stop” = peptide chain termination. The three-letter and singleletter abbrev ...
Biology 1 Unit 2 2. Chemistry: Atoms, Compounds, Water, pH
... Valence electrons – electrons that can be gained, lost, or shared in a chemical reaction Macromolecules (or macronutrients) – the large molecules that make up living organisms, such as proteins, carbohydrates, lipids, and nucleic acids Monomer – a small molecule that may be chemically bonded to othe ...
... Valence electrons – electrons that can be gained, lost, or shared in a chemical reaction Macromolecules (or macronutrients) – the large molecules that make up living organisms, such as proteins, carbohydrates, lipids, and nucleic acids Monomer – a small molecule that may be chemically bonded to othe ...
study guide - Dorman High School
... a. carbohydrate (mono-, di-, and polysaccharide) b. protein (amino acid, polypeptide, dipeptide) c. lipid (glycerol, fatty acid, triglyceride, phospholipid, steroid) d. nucleic acid (DNA, RNA, purines, pyrimidines, ribose, deoxyribose) ...
... a. carbohydrate (mono-, di-, and polysaccharide) b. protein (amino acid, polypeptide, dipeptide) c. lipid (glycerol, fatty acid, triglyceride, phospholipid, steroid) d. nucleic acid (DNA, RNA, purines, pyrimidines, ribose, deoxyribose) ...
7th-cells-and-heredity-unit-1-lesson-2-chemistry-of-life
... • Used to build and repair body structures • Enzymes (a type of protein) help chemical processes happen in cells. • Found in meat, beans, nuts ...
... • Used to build and repair body structures • Enzymes (a type of protein) help chemical processes happen in cells. • Found in meat, beans, nuts ...
Anatomy and Physiology Chapter #2
... 2.2 Structure of Matter Matter is anything that has mass (weight) and takes up space. Matter is found in various forms, gases, liquids, and solids Elements make up all matter. Elements are composed of tiny particles called atoms. The smallest complete units of elements are atoms. ...
... 2.2 Structure of Matter Matter is anything that has mass (weight) and takes up space. Matter is found in various forms, gases, liquids, and solids Elements make up all matter. Elements are composed of tiny particles called atoms. The smallest complete units of elements are atoms. ...
Anatomy and Physiology Chapter #2
... 2.2 Structure of Matter Matter is anything that has mass (weight) and takes up space. Matter is found in various forms, gases, liquids, and solids Elements make up all matter. Elements are composed of tiny particles called atoms. The smallest complete units of elements are atoms. ...
... 2.2 Structure of Matter Matter is anything that has mass (weight) and takes up space. Matter is found in various forms, gases, liquids, and solids Elements make up all matter. Elements are composed of tiny particles called atoms. The smallest complete units of elements are atoms. ...
Biomolecules You Are What You Eat Handout
... How do plants store glucose? What are some of the forms plants use to store starch? How do humans store carbohydrate energy? How do humans store energy over long periods of time? What are lipids? What keeps lipids from dissolving in water? What are the two ingredients of fats? What is glycerol? ...
... How do plants store glucose? What are some of the forms plants use to store starch? How do humans store carbohydrate energy? How do humans store energy over long periods of time? What are lipids? What keeps lipids from dissolving in water? What are the two ingredients of fats? What is glycerol? ...
Carbon-Based Molecules
... as they are in starches (like potatoes), or they may be different, as they are in proteins. ...
... as they are in starches (like potatoes), or they may be different, as they are in proteins. ...
Organic compounds are covalent compounds composed of carbon
... Carbon atoms bond together to form a structure called a backbone. It makes the molecule very strong. There are three types of structures for organic compounds. ...
... Carbon atoms bond together to form a structure called a backbone. It makes the molecule very strong. There are three types of structures for organic compounds. ...
2.4 review
... 9) Be able to describe the difference between the primary, secondary and tertiary & quarternary structure of a protein. 10) When a protein loses its shape and no longer works, it has become__________. 11) Be able to give at least 6 examples of protein functions in our body. 12) What is a proteome? 1 ...
... 9) Be able to describe the difference between the primary, secondary and tertiary & quarternary structure of a protein. 10) When a protein loses its shape and no longer works, it has become__________. 11) Be able to give at least 6 examples of protein functions in our body. 12) What is a proteome? 1 ...
Compounds for Life
... Chemical changes- a new chemical is formed Evidence of chemical changes can include: -bubbles forming when substances are added/mixed, a color change, burning/fire Examples: Rust forming on metal, Silver tarnishing (changing colors), Burning a pile of wood, Cooking bread and seeing it rise ...
... Chemical changes- a new chemical is formed Evidence of chemical changes can include: -bubbles forming when substances are added/mixed, a color change, burning/fire Examples: Rust forming on metal, Silver tarnishing (changing colors), Burning a pile of wood, Cooking bread and seeing it rise ...
Organic Chemistry Notes Powerpoint
... (fats), proteins, and nucleic acids (DNA and RNA). • These molecules are usually in the form of polymers, long chains of similar subunits. Because they are large, these molecules are called macromolecules. The subunits are called monomers. • The cell also contains water, inorganic salts and ions, an ...
... (fats), proteins, and nucleic acids (DNA and RNA). • These molecules are usually in the form of polymers, long chains of similar subunits. Because they are large, these molecules are called macromolecules. The subunits are called monomers. • The cell also contains water, inorganic salts and ions, an ...
Final Review - Chemistry Courses: About: Department of
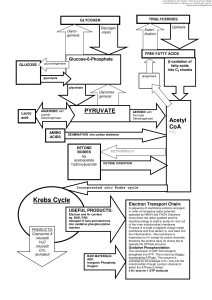
... 14.Electron transport, ATP synthase, P/O ratio 15. purpose of light reactions, dark reactions 16. energetics of FA synthesis and degradation 17. nitrogen processing, catabolism of AA 18. medical applications of nucleotide metabolism 19. nucleic acid structure on atomic level ...
... 14.Electron transport, ATP synthase, P/O ratio 15. purpose of light reactions, dark reactions 16. energetics of FA synthesis and degradation 17. nitrogen processing, catabolism of AA 18. medical applications of nucleotide metabolism 19. nucleic acid structure on atomic level ...
MASTERY 2.01 ______ 2.04 ______ Biology I Name: Unit 2
... explain the relationship between the two organic molecules? A. Both proteins and carbohydrates provide energy. B. Specific proteins called enzymes break down carbohydrates to release quick energy. C. Antibodies are proteins that protect the body from harmful substances, such as carbohydrates. D. Pro ...
... explain the relationship between the two organic molecules? A. Both proteins and carbohydrates provide energy. B. Specific proteins called enzymes break down carbohydrates to release quick energy. C. Antibodies are proteins that protect the body from harmful substances, such as carbohydrates. D. Pro ...
File
... carbon, blue for nitrogen, red for oxygen, and white for hydrogen. Part A: Models of Carbohydrates Most organisms rely on carbohydrates for their main source of energy. Carbohydrates include simple sugars and their polymers. 1. Glucose is a simple sugar, or monosaccharide, which means that it is a s ...
... carbon, blue for nitrogen, red for oxygen, and white for hydrogen. Part A: Models of Carbohydrates Most organisms rely on carbohydrates for their main source of energy. Carbohydrates include simple sugars and their polymers. 1. Glucose is a simple sugar, or monosaccharide, which means that it is a s ...
Dehydration Synthesis
... are so large that they are called *macromolecules which means giant molecules. Examples: Carbohydrates, lipids, proteins and nucleic acids. Most macromolecules are fomed through a process called *polymerization in which large compounds are built by ...
... are so large that they are called *macromolecules which means giant molecules. Examples: Carbohydrates, lipids, proteins and nucleic acids. Most macromolecules are fomed through a process called *polymerization in which large compounds are built by ...
Name
... Due on the day you take Chapter 5 test 1. Describe monomer and polymer and be able to relate it to the macromolecules discussed in this chapter. ...
... Due on the day you take Chapter 5 test 1. Describe monomer and polymer and be able to relate it to the macromolecules discussed in this chapter. ...
3.3 teacher Notes
... Building Blocks of Cells • The parts of a cell are made up of large, complex molecules, often called biomolecules. • Large, complex biomolecules are built from a few smaller, simpler, repeating units arranged in an extremely precise way. • The basic unit of most biomolecules contain atoms of carbon ...
... Building Blocks of Cells • The parts of a cell are made up of large, complex molecules, often called biomolecules. • Large, complex biomolecules are built from a few smaller, simpler, repeating units arranged in an extremely precise way. • The basic unit of most biomolecules contain atoms of carbon ...
Biochemistry (Macromolecules)
... C. The monomer “building blocks” are called Amino Acids (There are 20 different Amino Acids that can be involved in making proteins. Proteins and enzymes usually have hundreds to thousands of Amino acids in their structure.) D. Amino Acids have 4 different parts to them: 1. Carboxyl end (COOH) – Thi ...
... C. The monomer “building blocks” are called Amino Acids (There are 20 different Amino Acids that can be involved in making proteins. Proteins and enzymes usually have hundreds to thousands of Amino acids in their structure.) D. Amino Acids have 4 different parts to them: 1. Carboxyl end (COOH) – Thi ...
Biochemistry
... • Two major factors contribute to uniqueness – Each amino acid has distinct properties • R groups – Sequence of amino acids bound together • Varying combinations lead to distinct proteins • Changes in types or positions of amino acids • Sequence also affects levels of protein structure ...
... • Two major factors contribute to uniqueness – Each amino acid has distinct properties • R groups – Sequence of amino acids bound together • Varying combinations lead to distinct proteins • Changes in types or positions of amino acids • Sequence also affects levels of protein structure ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.