Hemoglobin, or haemoglobin, is an iron
... Hemoglobin (Hb) is an iron-containing protein in the red blood cells of mammals and other vertebrates. Hb is released under certain pathological conditions, such as malarial infection and allergic drug reaction. This free Hb is toxic and causes damage to blood vessels and surrounding tissues. Haptog ...
... Hemoglobin (Hb) is an iron-containing protein in the red blood cells of mammals and other vertebrates. Hb is released under certain pathological conditions, such as malarial infection and allergic drug reaction. This free Hb is toxic and causes damage to blood vessels and surrounding tissues. Haptog ...
Mrs
... You will now join them together by cutting out the necessary –OH and –H. You only cut them out when you join 2 together. Cut on the solid black lines of the–OH and –H. Save the -OH and –H and put them next to each other to make water. How many water molecules do you make from joining these 6 subunit ...
... You will now join them together by cutting out the necessary –OH and –H. You only cut them out when you join 2 together. Cut on the solid black lines of the–OH and –H. Save the -OH and –H and put them next to each other to make water. How many water molecules do you make from joining these 6 subunit ...
Solutions - Seattle Central
... compound. A molecule of a compound is the smallest particle that has the specific properties of the compound CH4 (methane). ...
... compound. A molecule of a compound is the smallest particle that has the specific properties of the compound CH4 (methane). ...
Ch7METABOLISM
... A few hours into the fast, most of the cells are depending on fatty acids, except brain, nerve, and red blood cells. Body proteins must always breakdown to some extent during fasting to provide glucose for these cells. However, the body cannot afford to breakdown its own protein at a high rate. ...
... A few hours into the fast, most of the cells are depending on fatty acids, except brain, nerve, and red blood cells. Body proteins must always breakdown to some extent during fasting to provide glucose for these cells. However, the body cannot afford to breakdown its own protein at a high rate. ...
Final Exam: Multiple Choice Portion Biochem Block Spring 2016
... A) fructose is cleaved into two molecules of guanine, absorbing energy B) glucose is cleaved into two molecules of pyruvate, releasing energy C) glucose is produced from carbon dioxide D) electrons flow from NADH to oxygen, producing ATP 25. Which statement describes best what happens in the citric ...
... A) fructose is cleaved into two molecules of guanine, absorbing energy B) glucose is cleaved into two molecules of pyruvate, releasing energy C) glucose is produced from carbon dioxide D) electrons flow from NADH to oxygen, producing ATP 25. Which statement describes best what happens in the citric ...
chemistry of life
... 2.2 Structure of Matter Matter is anything that has mass (weight) and takes up space. Matter is found in various forms, gases, liquids, and solids Elements make up all matter. Elements are composed of tiny particles called atoms. The smallest complete units of elements are atoms. ...
... 2.2 Structure of Matter Matter is anything that has mass (weight) and takes up space. Matter is found in various forms, gases, liquids, and solids Elements make up all matter. Elements are composed of tiny particles called atoms. The smallest complete units of elements are atoms. ...
Ch8 sec4Life with Carbon
... made of proteins. Hormones, Insulin & hemoglobin is Protein. – Chains of amino Acids Proteins are long chains of amino acids. Different proteins are made up of different sequence of amino acids. – Food Proteins Become your Proteins: Proteins are found in meat, fish, eggs, and milk products. The body ...
... made of proteins. Hormones, Insulin & hemoglobin is Protein. – Chains of amino Acids Proteins are long chains of amino acids. Different proteins are made up of different sequence of amino acids. – Food Proteins Become your Proteins: Proteins are found in meat, fish, eggs, and milk products. The body ...
Biochemistry CDT Practice
... A. Water sticks to things it cannot dissolve. B. Water is able to exist in three states of matter at room temperature. C. Water is able to dissolve a large variety of chemicals because it is a polar molecule. D. Water can absorb large amounts of energy without significant changes in temperature. Ans ...
... A. Water sticks to things it cannot dissolve. B. Water is able to exist in three states of matter at room temperature. C. Water is able to dissolve a large variety of chemicals because it is a polar molecule. D. Water can absorb large amounts of energy without significant changes in temperature. Ans ...
2-Biochemistry
... A. Water sticks to things it cannot dissolve. B. Water is able to exist in three states of matter at room temperature. C. Water is able to dissolve a large variety of chemicals because it is a polar molecule. D. Water can absorb large amounts of energy without significant changes in temperature. Ans ...
... A. Water sticks to things it cannot dissolve. B. Water is able to exist in three states of matter at room temperature. C. Water is able to dissolve a large variety of chemicals because it is a polar molecule. D. Water can absorb large amounts of energy without significant changes in temperature. Ans ...
Unit 3 Review Sheet – Biochemistry
... What is the difference in a monosaccharide, disaccharide, and polysaccharide? Monosaccharide is a single sugar, disaccharide is made of two monosaccharides and a polysaccharide is made of many monosaccharides What are some examples of each of the 4 macromolecules? 1. Carbohydrate Glucose, fructose ( ...
... What is the difference in a monosaccharide, disaccharide, and polysaccharide? Monosaccharide is a single sugar, disaccharide is made of two monosaccharides and a polysaccharide is made of many monosaccharides What are some examples of each of the 4 macromolecules? 1. Carbohydrate Glucose, fructose ( ...
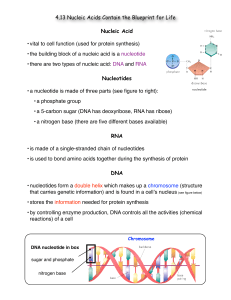
4.13 notes
... • there are two types of nucleic acid: DNA and RNA Nucleotides • a nucleotide is made of three parts (see figure to right): • a phosphate group • a 5-carbon sugar (DNA has deoxyribose, RNA has ribose) • a nitrogen base (there are five different bases available) RNA • is made of a single-stranded cha ...
... • there are two types of nucleic acid: DNA and RNA Nucleotides • a nucleotide is made of three parts (see figure to right): • a phosphate group • a 5-carbon sugar (DNA has deoxyribose, RNA has ribose) • a nitrogen base (there are five different bases available) RNA • is made of a single-stranded cha ...
( 2 points each).
... Multiple Choice. Choose the one alternative that best completes the statement or answers the question. ( 2 points each). ...
... Multiple Choice. Choose the one alternative that best completes the statement or answers the question. ( 2 points each). ...
Complex carbohydrates
... Compounds: molecules made of different kinds of atoms. Molecules found in living things are most often combinations of six elements: carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur. These elements join together to form proteins, carbohydrates, lipids, ATP, and nucleic acids. ...
... Compounds: molecules made of different kinds of atoms. Molecules found in living things are most often combinations of six elements: carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur. These elements join together to form proteins, carbohydrates, lipids, ATP, and nucleic acids. ...
THE MOLECULES OF LIFE - Christian Heritage School
... Glycogen, cellulose, and starch are three types of polysaccharides found in food. Though all three polymers are composed of the same monomer, glucose, the way the glucose monomers link together is different for each. ...
... Glycogen, cellulose, and starch are three types of polysaccharides found in food. Though all three polymers are composed of the same monomer, glucose, the way the glucose monomers link together is different for each. ...
Organic Molecules
... • Carbon bonds to hydrogen • Simplest hydrocarbon is when 4 hydrogen atoms bond to one ...
... • Carbon bonds to hydrogen • Simplest hydrocarbon is when 4 hydrogen atoms bond to one ...
Proteins
... parts towards the centre and hydrophilic parts towards the edges – therefore water soluble – metabolic Fibrous – long fibres of repeated sequences of AA’s so therefore they are insoluble in water – and these are structural – Keratin and Collagen ...
... parts towards the centre and hydrophilic parts towards the edges – therefore water soluble – metabolic Fibrous – long fibres of repeated sequences of AA’s so therefore they are insoluble in water – and these are structural – Keratin and Collagen ...
Biochemistry Review Sheet Chemical Reactions and Properties of
... Why are lipids and carbohydrates important in animal cells? List the seven functions of proteins. Know examples for each. What is the chemical formula for glucose? How many amino acids are there total? How many does your body make naturally? How many do you have to eat? 5. What is the difference bet ...
... Why are lipids and carbohydrates important in animal cells? List the seven functions of proteins. Know examples for each. What is the chemical formula for glucose? How many amino acids are there total? How many does your body make naturally? How many do you have to eat? 5. What is the difference bet ...
Catabolic Pathways and Glycolysis
... molecules. The 2 catabolic processes that occur in organisms are fermentation (breakdown without O2)and cellular respiration (breakdown with O2). ...
... molecules. The 2 catabolic processes that occur in organisms are fermentation (breakdown without O2)and cellular respiration (breakdown with O2). ...
VGCSE Health and Social Care Unit 2
... polypeptide chains held together by various bonds. The quaternary structure is the way these different parts are assembled together. Types of bonds: The shape of the protein is held together by Hydrogen bonds between some of the R groups (side chains) and Ionic bonds between positively and negativel ...
... polypeptide chains held together by various bonds. The quaternary structure is the way these different parts are assembled together. Types of bonds: The shape of the protein is held together by Hydrogen bonds between some of the R groups (side chains) and Ionic bonds between positively and negativel ...
Document
... Proteins: Composed of a long chain of subunits called AMINO ACIDS. Proteins function as Structural molecules – Hair for example And Proteins function as ENZYMES. Nucleic Acids: Are made up of Nucleotides. The Nucleotides are the ...
... Proteins: Composed of a long chain of subunits called AMINO ACIDS. Proteins function as Structural molecules – Hair for example And Proteins function as ENZYMES. Nucleic Acids: Are made up of Nucleotides. The Nucleotides are the ...
The Necessities of Life
... Molecules and Nutrients • All organisms must break down food in order to use the nutrients in it. • Nutrients are made up of molecules. • A molecule is a substance made when two or more atoms combine • Molecules made of two or more different atoms are compounds. • Molecules in living things are mad ...
... Molecules and Nutrients • All organisms must break down food in order to use the nutrients in it. • Nutrients are made up of molecules. • A molecule is a substance made when two or more atoms combine • Molecules made of two or more different atoms are compounds. • Molecules in living things are mad ...
Review Problems #2 (Enzyme Review, Phosphatases
... We will definitely not get through all of these, but it is useful to have them in one place. 1) Outline the chemical intermediates in the degradation of the following amino acids: Asn, Asp. What cofactor(s) play a role in this process? What other end product may be formed from Asp. What cycle does t ...
... We will definitely not get through all of these, but it is useful to have them in one place. 1) Outline the chemical intermediates in the degradation of the following amino acids: Asn, Asp. What cofactor(s) play a role in this process? What other end product may be formed from Asp. What cycle does t ...
Organic Macromolecules
... molecules (monomers) into long chains (polymers) Monomers combine by disconnecting from some of the hydrogen and oxygen atoms between them After the monomers bond, the excess oxygen and hydrogen atoms combine forming a water molecule Since the bonding process releases water, we call it a dehydration ...
... molecules (monomers) into long chains (polymers) Monomers combine by disconnecting from some of the hydrogen and oxygen atoms between them After the monomers bond, the excess oxygen and hydrogen atoms combine forming a water molecule Since the bonding process releases water, we call it a dehydration ...
Document
... B) Oxaloacetate C) a-ketoglutarate D) 3-phosphoglycerate 2. A Roundup Ready plant is one that has been genetically modified so that an enzyme (EPSP synthase) can no longer bind to the active ingredient (glyphosphate) which is a competitive inhibitor of A) shikimate (in the aromatic amino acid pathwa ...
... B) Oxaloacetate C) a-ketoglutarate D) 3-phosphoglycerate 2. A Roundup Ready plant is one that has been genetically modified so that an enzyme (EPSP synthase) can no longer bind to the active ingredient (glyphosphate) which is a competitive inhibitor of A) shikimate (in the aromatic amino acid pathwa ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.