Metabolism and Energetics
... Beta oxidation is a repeating 4 step process in which sequential 2-C groups (“acetyl groups”) are cut from the long chain; they are attached to a carrier (CoA) and then shuttled into the mitochondria These 2-C groups are then burned in the ...
... Beta oxidation is a repeating 4 step process in which sequential 2-C groups (“acetyl groups”) are cut from the long chain; they are attached to a carrier (CoA) and then shuttled into the mitochondria These 2-C groups are then burned in the ...
File - Craftsbury Science
... larger polymer. 3. I can understand and interpret the form and function of polysaccharides. 4. I can interpret how the number of saturated or unsaturated fatty acids can influence polysaccharides’ appearance and function. 5. I can explain the interacting forces in determining the form and function o ...
... larger polymer. 3. I can understand and interpret the form and function of polysaccharides. 4. I can interpret how the number of saturated or unsaturated fatty acids can influence polysaccharides’ appearance and function. 5. I can explain the interacting forces in determining the form and function o ...
Quiz 7 Name: 1. After ATP fuels the Na+/K+ pump at the cell
... 3. Which molecules can be utilized (directly or after some breakdown) in the cellular respiration pathway to generate ATP energy? A) amino acids and proteins B) glycerol and fatty acids C) glucose and sucrose D) starch and glycogen E) all of the above 4. What is needed to keep glycolysis reaction ru ...
... 3. Which molecules can be utilized (directly or after some breakdown) in the cellular respiration pathway to generate ATP energy? A) amino acids and proteins B) glycerol and fatty acids C) glucose and sucrose D) starch and glycogen E) all of the above 4. What is needed to keep glycolysis reaction ru ...
Amino Acids - Newcastle University
... Amino acids are often referred to as ‘the building blocks of life’. This is because they combine in different sequences to form proteins, which are fundamental to all living organisms. There are 21 amino acids, 9 of which are called ‘essential’ because they cannot be naturally found in the body. Thi ...
... Amino acids are often referred to as ‘the building blocks of life’. This is because they combine in different sequences to form proteins, which are fundamental to all living organisms. There are 21 amino acids, 9 of which are called ‘essential’ because they cannot be naturally found in the body. Thi ...
Fibers, Proteins and Membranes
... Clearly, packing the hydrocarbon tails is easier when they are straight and therefore one way to lower the temperature is to have tails with kinks. Kinks are due to double bonds. Another way is the insertion of other suitable molecules that disrupt the packing of the tails. The main such molecule is ...
... Clearly, packing the hydrocarbon tails is easier when they are straight and therefore one way to lower the temperature is to have tails with kinks. Kinks are due to double bonds. Another way is the insertion of other suitable molecules that disrupt the packing of the tails. The main such molecule is ...
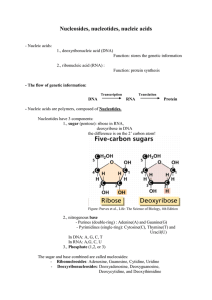
Nucleosides, nucleotides, nucleic acids
... The nucleotide sequence (=base sequence) carries the genetic information, this information will be translated into amino-acid sequence during protein synthesis. - Types and structure of RNA: - messenger RNA = mRNA: carries the information from the DNA to the site of protein synthesis. Single strande ...
... The nucleotide sequence (=base sequence) carries the genetic information, this information will be translated into amino-acid sequence during protein synthesis. - Types and structure of RNA: - messenger RNA = mRNA: carries the information from the DNA to the site of protein synthesis. Single strande ...
Carbon Compounds 2-3 Foldable Instructions
... There are four groups of organic compounds found in living things: ...
... There are four groups of organic compounds found in living things: ...
Unit 4 Test Review-Biomolecules Name Period ______ 1. Complete
... Macromolecule/ Organic Compound Carbohydrate ...
... Macromolecule/ Organic Compound Carbohydrate ...
Chemical Basis of Life
... Introduction to Lipids • Lipids are non-polar molecules that include fats, oils, and cholesterol. • Lipids, similar to carbohydrates, contain carbon atoms bonded to oxygen and hydrogen atoms. • Lipids have several different functions in living organisms. Some lipids are broken down for usable energ ...
... Introduction to Lipids • Lipids are non-polar molecules that include fats, oils, and cholesterol. • Lipids, similar to carbohydrates, contain carbon atoms bonded to oxygen and hydrogen atoms. • Lipids have several different functions in living organisms. Some lipids are broken down for usable energ ...
Unit 1 Practice Test
... 31. Which organic compound is correctly matched with the subunit that composes it? (a) maltose amino acid (b) starchglucose (c) proteinfatty acid (d) lipidsucrose 32. A chemical bond in which two atoms share a pair of electrons is referred to as (a) covalent (b) acidic (c) ionic (d) double 33. ...
... 31. Which organic compound is correctly matched with the subunit that composes it? (a) maltose amino acid (b) starchglucose (c) proteinfatty acid (d) lipidsucrose 32. A chemical bond in which two atoms share a pair of electrons is referred to as (a) covalent (b) acidic (c) ionic (d) double 33. ...
Atomic Structure (Bohr or Planetary Model)
... • decomposition reactions that release energy (due to bonds breaking) in the form of HEAT into the environment of the reaction • reactants contain more energy than the products ...
... • decomposition reactions that release energy (due to bonds breaking) in the form of HEAT into the environment of the reaction • reactants contain more energy than the products ...
Slide 1
... Cohesion and adhesion contribute to the rise of water molecules within a tree's water transport system. The dotted lines in the diagram indicate hydrogen bonds. ...
... Cohesion and adhesion contribute to the rise of water molecules within a tree's water transport system. The dotted lines in the diagram indicate hydrogen bonds. ...
Proteins - Westgate Mennonite Collegiate
... 2. secondary structure (2) of a protein results from hydrogen bonds involving the backbone, where the peptide chain is held in structures, either a coiled α-helix or folded β-pleated sheet; proteins often have both types of secondary structure in different regions of the chain ...
... 2. secondary structure (2) of a protein results from hydrogen bonds involving the backbone, where the peptide chain is held in structures, either a coiled α-helix or folded β-pleated sheet; proteins often have both types of secondary structure in different regions of the chain ...
Atoms
... Excess OH- combines with H+ to form H2O Together these reactions keep the blood at a pH between 7.3 and 7.5 ...
... Excess OH- combines with H+ to form H2O Together these reactions keep the blood at a pH between 7.3 and 7.5 ...
Lorem Ipsum - Tri-County Technical College
... live without it Obligate anaerobe is organism that cannot use oxygen and is poisoned by it Facultative anaerobe is organism that uses oxygen if available (in fact, prefers it) but can switch to alternate pathway if oxygen is unavailable ...
... live without it Obligate anaerobe is organism that cannot use oxygen and is poisoned by it Facultative anaerobe is organism that uses oxygen if available (in fact, prefers it) but can switch to alternate pathway if oxygen is unavailable ...
4NucleicAcidsProteins - San Elijo Elementary School
... Enzymatic proteins regulate chemical Rxs Structural proteins support (ex. Muscles, cartilage) Storage proteins store amino acids Transport proteins move substances Hormonal proteins coordinate multicellular ...
... Enzymatic proteins regulate chemical Rxs Structural proteins support (ex. Muscles, cartilage) Storage proteins store amino acids Transport proteins move substances Hormonal proteins coordinate multicellular ...
McMush
... 2) Disaccharides two sugar monomers = Structure (joined by a glycosidic linkage via. dehydration synthesis) ...
... 2) Disaccharides two sugar monomers = Structure (joined by a glycosidic linkage via. dehydration synthesis) ...
Chapter 3 Lecture notes
... differences in the covalent bonds that lead to different overall structures and functions. Review: Covalent bonds (Module 2.8). C. Starch is used for long-term energy storage only in plants. Starch molecules are helical and may be either unbranched or branched. Animals can hydrolyze this polymer to ...
... differences in the covalent bonds that lead to different overall structures and functions. Review: Covalent bonds (Module 2.8). C. Starch is used for long-term energy storage only in plants. Starch molecules are helical and may be either unbranched or branched. Animals can hydrolyze this polymer to ...
Cellular_Respiration_overviewap
... generation is via substrate level phosphorylation. Below is the general reaction per one glucose molecule: 2 acetylCoA + 2 oxaloacetate 2 oxaloacetate + 2ATP + 6NADH + 2FADH2 + 4CO2 Electron Transport Chain: Along the inner membrane of the mitochondria The final step of aerobic cellular respiratio ...
... generation is via substrate level phosphorylation. Below is the general reaction per one glucose molecule: 2 acetylCoA + 2 oxaloacetate 2 oxaloacetate + 2ATP + 6NADH + 2FADH2 + 4CO2 Electron Transport Chain: Along the inner membrane of the mitochondria The final step of aerobic cellular respiratio ...
SURFIN` THROUGH STAAR
... a. composed of building blocks called amino acids b. insoluble in water and are used by the body for energy storage and insulation c. complex biomolecules that store genetic information d. organic compounds used by cells to store and release energy ...
... a. composed of building blocks called amino acids b. insoluble in water and are used by the body for energy storage and insulation c. complex biomolecules that store genetic information d. organic compounds used by cells to store and release energy ...
Chemistry of Life 3a Puzzle Paragraph
... Enzymes are globular proteins which act as catalysts of chemical reactions. Without enzymes to ____________ them, many chemical processes happen at a very slow rate in living organisms. By making some enzymes and not others, cells can control what chemical reactions happen in their cytoplasm. Denatu ...
... Enzymes are globular proteins which act as catalysts of chemical reactions. Without enzymes to ____________ them, many chemical processes happen at a very slow rate in living organisms. By making some enzymes and not others, cells can control what chemical reactions happen in their cytoplasm. Denatu ...
Organic Compounds Powerpoint
... – each can form a strong covalent bond – bonds with many elements including other carbon atoms ...
... – each can form a strong covalent bond – bonds with many elements including other carbon atoms ...
biochemistry - Kuliah FTSL
... • Cells break down macromolecules by a process called hydrolysis (adding a molecule of water) ...
... • Cells break down macromolecules by a process called hydrolysis (adding a molecule of water) ...
Organic Chemistry & Carbohydrates: Structure & Function
... Aldehydes if the carbonyl group is at the end of the carbon skeleton ...
... Aldehydes if the carbonyl group is at the end of the carbon skeleton ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.