Organic Chemistry for Biology
... 4 groups of carbon compounds found in living things are carbohydrates, lipids, nucleic acids, and protein. Living things use carbohydrates as their main source of energy. Plants and some animals also use carbohydrates for structural purposes. Lipids can be used to store energy. Some lipids are impor ...
... 4 groups of carbon compounds found in living things are carbohydrates, lipids, nucleic acids, and protein. Living things use carbohydrates as their main source of energy. Plants and some animals also use carbohydrates for structural purposes. Lipids can be used to store energy. Some lipids are impor ...
CARBOHYDRATES Carbohydrates are made up of carbon
... 2. They help in maintaining the composition of protoplasm. 3. They are used in the formation of various structures in the body eg keratin in the hair, nails, hooves, horns, feathers etc. 4. They are important in the formation of enzymes, which speed up chemical reactions in organisms.. 5. They form ...
... 2. They help in maintaining the composition of protoplasm. 3. They are used in the formation of various structures in the body eg keratin in the hair, nails, hooves, horns, feathers etc. 4. They are important in the formation of enzymes, which speed up chemical reactions in organisms.. 5. They form ...
This is Most of an Old Exam
... C. NAD+ and FAD+ are hydrogen carrier molecules. NAD+ can carry one hydrogen and electron, FAD+ can carry two. D. Hexokinase, the enzyme that catalyzes the first reaction in glycolysis, is highly regulated via allosteric mechanisms. _____ 2.Synthesis of glucose from non-carbohydrate precursors is ca ...
... C. NAD+ and FAD+ are hydrogen carrier molecules. NAD+ can carry one hydrogen and electron, FAD+ can carry two. D. Hexokinase, the enzyme that catalyzes the first reaction in glycolysis, is highly regulated via allosteric mechanisms. _____ 2.Synthesis of glucose from non-carbohydrate precursors is ca ...
How many nucleotides are in 12 mRNA codons?
... A particular DNA sequence reads TCGAGGTCACCG. A mutation occurs in which the first "A" in the sequence is deleted. What will happen to the protein produced? A ...
... A particular DNA sequence reads TCGAGGTCACCG. A mutation occurs in which the first "A" in the sequence is deleted. What will happen to the protein produced? A ...
FAD
... protein—ester linkage E. steroid—peptide bond 27. When a nucleic acid undergoes hydrolysis, the resulting subunits are: A. amino acids B. monosaccharides C. nucleotides D. fatty acids E. carotenoids 28. ATP is important in living organisms because: A. like all other nucleic acids, it stores heredita ...
... protein—ester linkage E. steroid—peptide bond 27. When a nucleic acid undergoes hydrolysis, the resulting subunits are: A. amino acids B. monosaccharides C. nucleotides D. fatty acids E. carotenoids 28. ATP is important in living organisms because: A. like all other nucleic acids, it stores heredita ...
chapter 3 - Fullfrontalanatomy.com
... Key Terms amino acid atherosclerosis carbohydrates cellulose dehydration reaction denaturation disaccharides DNA double helix fat functional groups gene glycogen hydrocarbons hydrogenation hydrolysis hydrophilic hydrophobic isomers lipids macromolecules monomers monosaccharides nucleic acids nucleot ...
... Key Terms amino acid atherosclerosis carbohydrates cellulose dehydration reaction denaturation disaccharides DNA double helix fat functional groups gene glycogen hydrocarbons hydrogenation hydrolysis hydrophilic hydrophobic isomers lipids macromolecules monomers monosaccharides nucleic acids nucleot ...
Chemical Basis of Life
... a. It creates the right pH needed for the reaction. b. It decreases the amount of energy needed for the reaction. c. It provides the extra energy needed for the reaction. d. It maintains the proper temperature needed for the reaction. Examine the structural formula below. ...
... a. It creates the right pH needed for the reaction. b. It decreases the amount of energy needed for the reaction. c. It provides the extra energy needed for the reaction. d. It maintains the proper temperature needed for the reaction. Examine the structural formula below. ...
Unit 2 Biochemistry Chp 4 and 5 Organic and
... 2. How many valence electrons (outer energy level) does carbon have? ...
... 2. How many valence electrons (outer energy level) does carbon have? ...
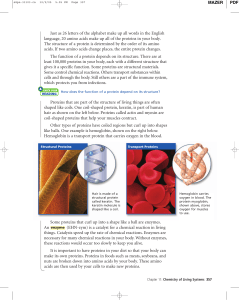
Just as 26 letters of the alphabet make up all words in the English
... acids are then used by your cells to make new proteins. Chapter 11: Chemistry of Living Systems 357 ...
... acids are then used by your cells to make new proteins. Chapter 11: Chemistry of Living Systems 357 ...
2008 CELL BIOLOGY – TRAINING HANDOUT
... has a pH of 7 but when it mixes with air the suspended materials will either raise or lower its pH. Acid Rain is an example of this type of reaction. Enzymes ...
... has a pH of 7 but when it mixes with air the suspended materials will either raise or lower its pH. Acid Rain is an example of this type of reaction. Enzymes ...
Download PDF
... electron capture, proton and ion gradients, and conversion to mechanical energy. In particular, we will explore the thermodynamics of electron transport, proton pumping, and ATP biosynthesis. 3. Molecular biosynthesis. Most organisms can biosynthesize amino acids, lipids, vitamins, and cofactors usi ...
... electron capture, proton and ion gradients, and conversion to mechanical energy. In particular, we will explore the thermodynamics of electron transport, proton pumping, and ATP biosynthesis. 3. Molecular biosynthesis. Most organisms can biosynthesize amino acids, lipids, vitamins, and cofactors usi ...
Metabolic Adaptation - Washington State University
... versus protein • Amino acids and carbohydrates are at about the same oxidation state – so although the exact pathways may vary from one amino acid to another, the yield of ATP from a gram of amino acid is about the same as the yield from a gram of glucose. • However, as in the example of the fly, am ...
... versus protein • Amino acids and carbohydrates are at about the same oxidation state – so although the exact pathways may vary from one amino acid to another, the yield of ATP from a gram of amino acid is about the same as the yield from a gram of glucose. • However, as in the example of the fly, am ...
METABOLIC PATHWAYS Section 6 SOME BASIC CONCEPTS OF
... Instead, this reaction is slowed down so that 3 ATP molecules are formed: 52 - 21 = 3 ATP molecules + 31 kcal heat, efficiency is 40%. If O2 is not available to the cytochrome system, ATP will not be formed by the mitochondria. FAD flavine adenine dinucleotide is similar to NAD I. Carbohydrate metab ...
... Instead, this reaction is slowed down so that 3 ATP molecules are formed: 52 - 21 = 3 ATP molecules + 31 kcal heat, efficiency is 40%. If O2 is not available to the cytochrome system, ATP will not be formed by the mitochondria. FAD flavine adenine dinucleotide is similar to NAD I. Carbohydrate metab ...
Biology 20 Lecture Quiz #3 – Take Home Cellular Respiration
... Cellular Respiration – DUE 23 June 2010 at 7:50 AM – I do not want any late quizzes! 1. The main function of cellular respiration is _____. a) breaking down toxic molecules; b) making ATP to power cell activities; c) making food; d) producing cell structures from chemical building blocks; e) breakin ...
... Cellular Respiration – DUE 23 June 2010 at 7:50 AM – I do not want any late quizzes! 1. The main function of cellular respiration is _____. a) breaking down toxic molecules; b) making ATP to power cell activities; c) making food; d) producing cell structures from chemical building blocks; e) breakin ...
Medical Biochemistry at a Glance. 3rd Edition. At a Glance Brochure
... Order Online - http://www.researchandmarkets.com/reports/2251746/ Order by Fax - using the form below Order by Post - print the order form below and send to Research and Markets, ...
... Order Online - http://www.researchandmarkets.com/reports/2251746/ Order by Fax - using the form below Order by Post - print the order form below and send to Research and Markets, ...
Physical Science EOC Review Name
... 9. Energy enters an ecosystem from the ________________. 10. Each level in the transfer of energy through an ecosystem is called a __________________________. a. The first trophic level consists of primary (producers or consumers). i. Plants or other photosynthetic organisms are also called ________ ...
... 9. Energy enters an ecosystem from the ________________. 10. Each level in the transfer of energy through an ecosystem is called a __________________________. a. The first trophic level consists of primary (producers or consumers). i. Plants or other photosynthetic organisms are also called ________ ...
RNA and protein synthesis
... 4. A sequence of three mRNA nucleotides is called a codon. 5. One codon codes for one amino acid. 6. tRNA molecules enter the ribosome carrying the correct amino acid. The tRNA has an anticodon that matches the codon on the mRNA. 7. Amino acids are linked together to form a protein! ...
... 4. A sequence of three mRNA nucleotides is called a codon. 5. One codon codes for one amino acid. 6. tRNA molecules enter the ribosome carrying the correct amino acid. The tRNA has an anticodon that matches the codon on the mRNA. 7. Amino acids are linked together to form a protein! ...
Proteins - Downtown Magnets High School
... between molecules affect their structure and function. • a. Change in the structure of a molecular system may result in a change of the function of the system. • b. The shape of enzymes, active sites, and interaction with specific molecules are essential for basic functioning of the enzyme. ...
... between molecules affect their structure and function. • a. Change in the structure of a molecular system may result in a change of the function of the system. • b. The shape of enzymes, active sites, and interaction with specific molecules are essential for basic functioning of the enzyme. ...
Chapter 6: Chemistry in Biology
... __________ are the starting substances, on the left side of the arrow. __________ are the substances formed during the reaction, on the right side of the ...
... __________ are the starting substances, on the left side of the arrow. __________ are the substances formed during the reaction, on the right side of the ...
- thevignanam
... Secondary structure • Secondary structure in a protein refers to the regular folding of regions of the polypeptide chain. • The two most common types of secondary structure are the -helix and the -pleated sheet. • The -helix is a cylindrical, rod-like helical arrangement of the amino acids in the p ...
... Secondary structure • Secondary structure in a protein refers to the regular folding of regions of the polypeptide chain. • The two most common types of secondary structure are the -helix and the -pleated sheet. • The -helix is a cylindrical, rod-like helical arrangement of the amino acids in the p ...
Chapter 5 The Structure and Function of Macromolecules
... monomers Three of the four classes of macromolecules—carbohydrates, proteins, and nucleic acids—form chainlike molecules called polymers. A polymer is a long molecule consisting of many similar or identical building blocks linked by covalent bonds. The repeated units are small molecules called ...
... monomers Three of the four classes of macromolecules—carbohydrates, proteins, and nucleic acids—form chainlike molecules called polymers. A polymer is a long molecule consisting of many similar or identical building blocks linked by covalent bonds. The repeated units are small molecules called ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.