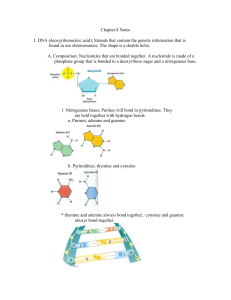
Chapter 2: Chemistry of Life 2.1: Atoms, Ions, and Molecules
... chemical reaction in living things. • Enzymes lower the activation energy needed to start chemical reactions. • Enzymes are involved in almost every process in living things. • Conditions such as temperature and pH can effect how well enzymes work. • Enzyme structure is important because each enzyme ...
... chemical reaction in living things. • Enzymes lower the activation energy needed to start chemical reactions. • Enzymes are involved in almost every process in living things. • Conditions such as temperature and pH can effect how well enzymes work. • Enzyme structure is important because each enzyme ...
Cellular Respiration
... adenine and the sugar ribose Main energy carrier in cells Formed during reactions that breakdown organic compounds to CO2 and water ...
... adenine and the sugar ribose Main energy carrier in cells Formed during reactions that breakdown organic compounds to CO2 and water ...
OCHeM.com ©1999 Thomas Poon Amino Acids, Peptides, and
... Abbr. Abbr. Side Chain (protonated form) α-COOH ...
... Abbr. Abbr. Side Chain (protonated form) α-COOH ...
Part 1B: Understanding Biochemical Testing for Bacterial
... Carbohydrates are complex chemical substrates which serve as energy sources when broken down by bacteria and other cells. They are composed of carbon, hydrogen, and oxygen (with ...
... Carbohydrates are complex chemical substrates which serve as energy sources when broken down by bacteria and other cells. They are composed of carbon, hydrogen, and oxygen (with ...
Cellular Respiration
... we see how energy enters food chains (via autotrophs) we can look at how organisms use that energy to fuel their bodies. Plants and animals both use products of photosynthesis (glucose) for metabolic fuel Heterotrophs: must take in energy from outside sources, cannot make their own e.g. animals ...
... we see how energy enters food chains (via autotrophs) we can look at how organisms use that energy to fuel their bodies. Plants and animals both use products of photosynthesis (glucose) for metabolic fuel Heterotrophs: must take in energy from outside sources, cannot make their own e.g. animals ...
The Origin and Chemistry of Life
... First cells would have been autonomous, membrane-bound units capable of selfreplication requiring: Nucleic Acids This causes a biological paradox. How could nucleic acids appear without the enzymes to synthesize them? How could enzymes exist without nucleic acids to ...
... First cells would have been autonomous, membrane-bound units capable of selfreplication requiring: Nucleic Acids This causes a biological paradox. How could nucleic acids appear without the enzymes to synthesize them? How could enzymes exist without nucleic acids to ...
File
... Disaccharide- two monosaccharides linked together, a polymer of glucose & fructose make sucrose Polysaccharide- the largest carbohydrate polymer made of many monosaccharide subunits. Starch, glycogen, & cellulose are polysaccharides that store energy for organisms. ...
... Disaccharide- two monosaccharides linked together, a polymer of glucose & fructose make sucrose Polysaccharide- the largest carbohydrate polymer made of many monosaccharide subunits. Starch, glycogen, & cellulose are polysaccharides that store energy for organisms. ...
Amino Acids and Dipeptides - Chemical Minds
... ii) Explain your answer in terms of the structure and functional groups present in the amino acids and in the dipeptide(s). ...
... ii) Explain your answer in terms of the structure and functional groups present in the amino acids and in the dipeptide(s). ...
Document
... i. Dietary fiber (roughage) is indigestible complex carbohydrates but good because it helps keep good cholesterol levels & may reduce risk of cancers. 4. Proteins COOa. made of amino acids: 20+ (R-CH-NH3+; R= side chain/ specific properties) i. essential ones = PVT TIM HALL: phenylalanine, valine, t ...
... i. Dietary fiber (roughage) is indigestible complex carbohydrates but good because it helps keep good cholesterol levels & may reduce risk of cancers. 4. Proteins COOa. made of amino acids: 20+ (R-CH-NH3+; R= side chain/ specific properties) i. essential ones = PVT TIM HALL: phenylalanine, valine, t ...
• Microbial Metabolism • What is metabolism? • All chemical
... Oxidation is the removal of electrons. Reduction is the gain of electrons. Redox reaction is an oxidation reaction paired with a reduction reaction. ...
... Oxidation is the removal of electrons. Reduction is the gain of electrons. Redox reaction is an oxidation reaction paired with a reduction reaction. ...
PowerPoint Presentation - Nerve activates contraction
... Consists of amino acids connected by peptide bonds Unique properties of each protein are determined by ◦ Type of amino acid ◦ Sequence of amino acid ...
... Consists of amino acids connected by peptide bonds Unique properties of each protein are determined by ◦ Type of amino acid ◦ Sequence of amino acid ...
Chapter 1 Study Questions
... 4. Compare the chemical structure of a small non-polar amino acid (such as alanine) to one with a bulky hydrocarbon side chain (such as isoleucine). What kind of chemical interactions are non-polar side chains involved in? 5. Which amino acids contain sulfur? Which contain hydroxyl (-OH) groups? Whi ...
... 4. Compare the chemical structure of a small non-polar amino acid (such as alanine) to one with a bulky hydrocarbon side chain (such as isoleucine). What kind of chemical interactions are non-polar side chains involved in? 5. Which amino acids contain sulfur? Which contain hydroxyl (-OH) groups? Whi ...
Unit 5 Proteins PPT
... Vegans do not eat meat of any kind and also do not eat eggs, dairy products, or processed foods containing these or other animal-derived ingredients such as gelatin. Many vegans also refrain from eating foods that are made using animal products that may not contain animal products in the finished pr ...
... Vegans do not eat meat of any kind and also do not eat eggs, dairy products, or processed foods containing these or other animal-derived ingredients such as gelatin. Many vegans also refrain from eating foods that are made using animal products that may not contain animal products in the finished pr ...
BioIIch17notesRNAfilled.p pt
... -one gene codes for one polypeptide chain -polypeptide chains may combine to form 1 protein -Hemoglobin made from 2 kinds of polypeptides—thus 2 genes code for this protein -ok to say one gene codes for one protein -DNA contains instructions for making proteins, RNA actually Makes the proteins -3 ma ...
... -one gene codes for one polypeptide chain -polypeptide chains may combine to form 1 protein -Hemoglobin made from 2 kinds of polypeptides—thus 2 genes code for this protein -ok to say one gene codes for one protein -DNA contains instructions for making proteins, RNA actually Makes the proteins -3 ma ...
Module 10: Catabolism of Amino Acids
... Module 9: Catabolism of Lipids 12. What is the structure of the partially oxidized fatty acyl group that is formed when oleic acid, 18:1(Δ9), has undergone three cycles of β oxidation? 13. Below is list of events that occur during fatty acid oxidation. For each fatty acid drawn below, fill in the bl ...
... Module 9: Catabolism of Lipids 12. What is the structure of the partially oxidized fatty acyl group that is formed when oleic acid, 18:1(Δ9), has undergone three cycles of β oxidation? 13. Below is list of events that occur during fatty acid oxidation. For each fatty acid drawn below, fill in the bl ...
Sample exam questions Chapter 11 Carbohydrates
... 14) During anaerobic metabolism of glucose in muscle, there is a net production in the cytosol of lactate and A. NAD+ B. NADH C. CO2 D. ATP * E. FADH2 15) The committed step of the glycolytic pathway is catalyzed by A. hexokinase B. phosphofructokinase * C. aldolase D. glycerol 6-phosphate dehydrog ...
... 14) During anaerobic metabolism of glucose in muscle, there is a net production in the cytosol of lactate and A. NAD+ B. NADH C. CO2 D. ATP * E. FADH2 15) The committed step of the glycolytic pathway is catalyzed by A. hexokinase B. phosphofructokinase * C. aldolase D. glycerol 6-phosphate dehydrog ...
Complete Protein - Kelloggs Nutrition
... Get Smart with Your Proteins Aim to get about 1 gram of protein for every 3 pounds of body weight daily depending on your needs and activity level. Follow these tips to be sure you're getting enough: Here are a few combinations to try but get creative and find your favorite. Barley and lentils Oatme ...
... Get Smart with Your Proteins Aim to get about 1 gram of protein for every 3 pounds of body weight daily depending on your needs and activity level. Follow these tips to be sure you're getting enough: Here are a few combinations to try but get creative and find your favorite. Barley and lentils Oatme ...
H &
... The names and structures of some of the intermediate compounds in metabolism are complex. You do not need to memorize them, but they wi[ be used in the text to make it easierto followwhat is happening. Remembei also that all the steps ofthese reactions are catalyzedby enzyrnes. The phosphoryl group ...
... The names and structures of some of the intermediate compounds in metabolism are complex. You do not need to memorize them, but they wi[ be used in the text to make it easierto followwhat is happening. Remembei also that all the steps ofthese reactions are catalyzedby enzyrnes. The phosphoryl group ...
Chapter 11 Notes
... major differences in DNA and RNA 1. ribose sugar instead of deoxyribose 2. uracil instead of thymine B. Types; 1. r-RNA; Ribosomal RNA and protein make up the beadlike structures known as ribosomes. 2. m-RNA; Messenger RNA takes instructions from DNA in the nucleus to the ribosomes in the cytoplasm ...
... major differences in DNA and RNA 1. ribose sugar instead of deoxyribose 2. uracil instead of thymine B. Types; 1. r-RNA; Ribosomal RNA and protein make up the beadlike structures known as ribosomes. 2. m-RNA; Messenger RNA takes instructions from DNA in the nucleus to the ribosomes in the cytoplasm ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.