Synthesis and Structure of Alcohols
... They are made via the reaction of metallic Mg with the corresponding alkyl halide, usually in ether type solvents. This reaction is versatile; primary, secondary and tertiary alkyl halides can be used, and also vinyl, allyl and aryl halides. ...
... They are made via the reaction of metallic Mg with the corresponding alkyl halide, usually in ether type solvents. This reaction is versatile; primary, secondary and tertiary alkyl halides can be used, and also vinyl, allyl and aryl halides. ...
Dicyanomethylenedihydrofuran photorefractive materials
... by the heterocycle bearing three cyano groups. Intermediate 25 itself was made from aromatic -ketol 24, which could be obtained conveniently from the attack of trimethylsilyl protected acetone cyanohydrin 22 by a Grignard Reagent 23. The condensation between 24 and malononitrile was found to procee ...
... by the heterocycle bearing three cyano groups. Intermediate 25 itself was made from aromatic -ketol 24, which could be obtained conveniently from the attack of trimethylsilyl protected acetone cyanohydrin 22 by a Grignard Reagent 23. The condensation between 24 and malononitrile was found to procee ...
17: Oxidation and Reduction
... We can imagine that chromate ion (CrO4-2) forms from dichromate (Cr2O7-2) by loss of chromium trioxide (CrO3), or that it forms from addition of H2O to CrO3 followed by deprotonation. The three "chromate" species, or the three "dichromate" species, are simply differently protonated froms of CrO4-2 o ...
... We can imagine that chromate ion (CrO4-2) forms from dichromate (Cr2O7-2) by loss of chromium trioxide (CrO3), or that it forms from addition of H2O to CrO3 followed by deprotonation. The three "chromate" species, or the three "dichromate" species, are simply differently protonated froms of CrO4-2 o ...
4.7 Preparation of Alkyl Halides from Alcohols and Hydrogen
... A) the alkyloxonium ion intermediate B) the transition step of the bimolecular proton transfer C) the transition state of the attack of the nucleophile on the carbocation D) the transition state of the unimolecular ...
... A) the alkyloxonium ion intermediate B) the transition step of the bimolecular proton transfer C) the transition state of the attack of the nucleophile on the carbocation D) the transition state of the unimolecular ...
Organic Compounds - 2012 Book Archive
... aryl halides. Oxygen-containing organic compounds, a third family, may be divided into two main types: those that contain at least one C–O bond, which include alcohols, phenols (derivatives of benzene), and ethers, and those that contain a carbonyl group (C=O), which include aldehydes, ketones, and ...
... aryl halides. Oxygen-containing organic compounds, a third family, may be divided into two main types: those that contain at least one C–O bond, which include alcohols, phenols (derivatives of benzene), and ethers, and those that contain a carbonyl group (C=O), which include aldehydes, ketones, and ...
Chemical Aspects of Distilling Wines into Brandy
... Peynaud and Guimberteau (8) estimated that no more than one-sixth of the leucine and valine in grape musts assimilated during fermentation gave rise to isoamyl and isobutyl alcohols, and since these amino acids are low i n grape musts, the formation of higher alcohols by amino acid degradation is ne ...
... Peynaud and Guimberteau (8) estimated that no more than one-sixth of the leucine and valine in grape musts assimilated during fermentation gave rise to isoamyl and isobutyl alcohols, and since these amino acids are low i n grape musts, the formation of higher alcohols by amino acid degradation is ne ...
Novel Transition Metal-Catalysed Syntheses of Carboxylic Acid
... The largest category of such reactions features the ruthenium metathesis catalysts developed by Grubbs and co-workers.[14] These alkylidene complexes are truly remarkable in terms of scope of their activity, so much so that an account of their nonmetathetic reactions has been recently published.[15] ...
... The largest category of such reactions features the ruthenium metathesis catalysts developed by Grubbs and co-workers.[14] These alkylidene complexes are truly remarkable in terms of scope of their activity, so much so that an account of their nonmetathetic reactions has been recently published.[15] ...
C 1 hapter
... adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through transition metal complexes. There are several challenges involved in applying chiral catalysts in the hydroboration reaction: the catalytic performance must be excellent, the dev ...
... adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through transition metal complexes. There are several challenges involved in applying chiral catalysts in the hydroboration reaction: the catalytic performance must be excellent, the dev ...
Organic Chemistry - UCR Chemistry
... deprotonation. The three "chromate" species, or the three "dichromate" species, are simply differently protonated froms of CrO4-2 or Cr2O7-2, respectively Unwanted Oxidation of Aldehydes. Cr(VI) reagents are powerful oxidizing agents useful for oxidizing 2° alcohols to ketones (Figure 17.005) becaus ...
... deprotonation. The three "chromate" species, or the three "dichromate" species, are simply differently protonated froms of CrO4-2 or Cr2O7-2, respectively Unwanted Oxidation of Aldehydes. Cr(VI) reagents are powerful oxidizing agents useful for oxidizing 2° alcohols to ketones (Figure 17.005) becaus ...
Full-Text PDF
... science-oriented research. In particular, commercial pharmaceuticals and agrochemicals frequently contain single fluorine atoms or trifluoromethyl groups [1–4]. Nevertheless, growing interest in emergent fluorinated substituents, like α-fluorinated ethers, has been observed within recent years [5–8] ...
... science-oriented research. In particular, commercial pharmaceuticals and agrochemicals frequently contain single fluorine atoms or trifluoromethyl groups [1–4]. Nevertheless, growing interest in emergent fluorinated substituents, like α-fluorinated ethers, has been observed within recent years [5–8] ...
15. Thermosets from Renewable Sources
... content as the amount of bio-based carbon in the material or product as a percent of the weight (mass) of the total organic carbon in the product. This standard utilizes two methods to quantify the bio-based content of a given product: (a) Accelerator Mass Spectrometry (AMS) along with Isotope Ratio ...
... content as the amount of bio-based carbon in the material or product as a percent of the weight (mass) of the total organic carbon in the product. This standard utilizes two methods to quantify the bio-based content of a given product: (a) Accelerator Mass Spectrometry (AMS) along with Isotope Ratio ...
Substitution Reactions of Specifically Ortho
... conversion is n o t as effective. In t h e particular case of 2-allylpiperonal (Xi),t h e reaction (condition B) is only 11%complete. When t h e ortho-lithiated imines were treated with CuIs at -78 "C, a green suspension was formed which showed a more pronounced effect o n the allyl bromide alkylati ...
... conversion is n o t as effective. In t h e particular case of 2-allylpiperonal (Xi),t h e reaction (condition B) is only 11%complete. When t h e ortho-lithiated imines were treated with CuIs at -78 "C, a green suspension was formed which showed a more pronounced effect o n the allyl bromide alkylati ...
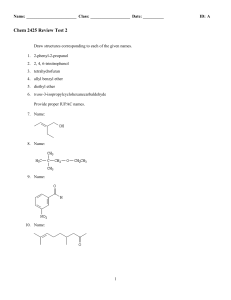
Chem 2425-Test 2 Review
... Consider the data below to answer the following question(s). Cyanohydrins are important intermediates in the synthesis of α-hydroxycarboxylic acids from ketones and aldehydes. The nitrile functional group can be hydrolyzed by aqueous acid to yield a carboxylic acid. Nitriles can also be hydrolyzed t ...
... Consider the data below to answer the following question(s). Cyanohydrins are important intermediates in the synthesis of α-hydroxycarboxylic acids from ketones and aldehydes. The nitrile functional group can be hydrolyzed by aqueous acid to yield a carboxylic acid. Nitriles can also be hydrolyzed t ...
CH - UTH e
... • Can’t make Grignards with acidic or electro‐philic f functional groups present in the molecule: i l i h l l ...
... • Can’t make Grignards with acidic or electro‐philic f functional groups present in the molecule: i l i h l l ...
Synthesis of Alcohols Using Grignard Reagents Grignard reagents
... Retrosynthetic analysis is the process by which we plan a synthesis by reasoning backward from the desired product (the "target molecule"). ...
... Retrosynthetic analysis is the process by which we plan a synthesis by reasoning backward from the desired product (the "target molecule"). ...
New insights into the mechanism of sorbitol transformation
... [25,28]. In this paper, we propose a focus on the reaction mechanisms involved in sorbitol transformation over this original bifunctional catalytic system using the following strategy. First, we extracted preliminary information on the C ...
... [25,28]. In this paper, we propose a focus on the reaction mechanisms involved in sorbitol transformation over this original bifunctional catalytic system using the following strategy. First, we extracted preliminary information on the C ...
unit 6 alcohols
... Esters: Now the LG is RO-, not usually considered “good,” but the reaction takes place by nucleophilic acyl substitution, not by SN2. In this mechanism, RO- leaving is exothermic and therefore favorable. ...
... Esters: Now the LG is RO-, not usually considered “good,” but the reaction takes place by nucleophilic acyl substitution, not by SN2. In this mechanism, RO- leaving is exothermic and therefore favorable. ...
OC 2/e Ch 15
... functional group • for an aldehyde, change the suffix from -e to -al • for an unsaturated aldehyde, show the carbon-carbon double bond by changing the infix from -an- to -en-; the location of the suffix determines the numbering ...
... functional group • for an aldehyde, change the suffix from -e to -al • for an unsaturated aldehyde, show the carbon-carbon double bond by changing the infix from -an- to -en-; the location of the suffix determines the numbering ...
Petasis reaction

The Petasis reaction (alternatively called the Petasis borono–Mannich (PBM) reaction) is the chemical reaction of an amine, aldehyde, and vinyl- or aryl-boronic acid to form substituted amines.Reported in 1993 by Nicos Petasis as a practical method towards the synthesis of a geometrically pure antifungal agent, naftifine, the Petasis reaction can be described as a variation of the Mannich reaction. Rather than generating an enolate to form the substituted amine product, in the Petasis reaction, the vinyl group of the organoboronic acid serves as the nucleophile. In comparison to other methods of generating allyl amines, the Petasis reaction tolerates a multifunctional scaffold, with a variety of amines and organoboronic acids as potential starting materials. Additionally, the reaction does not require anhydrous or inert conditions. As a mild, selective synthesis, the Petasis reaction is useful in generating α-amino acids, and is utilized in combinatorial chemistry and drug discovery.