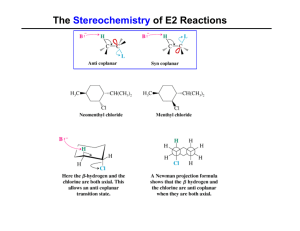
Year 13 Organic Chemistry Test
... Tollen's reagent. What would be seen in a positive test with Tollen's reagent? _____________________________________________________ _____________________________________________________ A second alcohol, 2-methyl propan-2-ol, will not give a positive breathalyzer test. Why not? ____________________ ...
... Tollen's reagent. What would be seen in a positive test with Tollen's reagent? _____________________________________________________ _____________________________________________________ A second alcohol, 2-methyl propan-2-ol, will not give a positive breathalyzer test. Why not? ____________________ ...
Chapter 18 Reactions of aromatics
... Mechanism of NBS (Radical) Reaction • Abstraction of a benzylic hydrogen atom generates an intermediate benzylic radical • Reacts with Br2 to yield product • Br· radical cycles back into reaction to carry chain • Br2 produced from reaction of HBr with NBS ...
... Mechanism of NBS (Radical) Reaction • Abstraction of a benzylic hydrogen atom generates an intermediate benzylic radical • Reacts with Br2 to yield product • Br· radical cycles back into reaction to carry chain • Br2 produced from reaction of HBr with NBS ...
Nucleophilic Substitution
... Let's look at how the various components of the reaction influence the reaction pathway: RReactivity order : CH3- > CH3CH2- > (CH3)2CH- > (CH3)3CIn an SN2 reaction, the transition state has 5 groups around the central C atom. As a consequence of the steric requirements at this center, less highly su ...
... Let's look at how the various components of the reaction influence the reaction pathway: RReactivity order : CH3- > CH3CH2- > (CH3)2CH- > (CH3)3CIn an SN2 reaction, the transition state has 5 groups around the central C atom. As a consequence of the steric requirements at this center, less highly su ...
IOSR Journal of Applied Chemistry (IOSR-JAC) ISSN: 2278-5736.
... Abstract: The selective introduction of acetonide as a protecting group for various diols has been achieved in an excellent yield under mild reaction conditions. The diol is transformed into acetonide with acetone and a cation exchange resin in solvent free condition. Keywords: Acetonide, diols, res ...
... Abstract: The selective introduction of acetonide as a protecting group for various diols has been achieved in an excellent yield under mild reaction conditions. The diol is transformed into acetonide with acetone and a cation exchange resin in solvent free condition. Keywords: Acetonide, diols, res ...
Alkanes CH4 + Cl2 → CH3Cl + HCl CH3CH3 + Cl2 → CH3CH2Cl +
... When the substance attacking the double bond is unsymmetrical (like HBr), the hydrogen atom goes on the atom with more H atoms to begin with. This will be studied more in a later unit, just remember it for now. It is Markovnikov’s rule. Substances that add to C=C bonds, such as H2, Br2, and HBr are ...
... When the substance attacking the double bond is unsymmetrical (like HBr), the hydrogen atom goes on the atom with more H atoms to begin with. This will be studied more in a later unit, just remember it for now. It is Markovnikov’s rule. Substances that add to C=C bonds, such as H2, Br2, and HBr are ...
ppt
... Conclusion • Very simple strategy for engaging CO2 in C–C bond formation that does not require synthetic or biological catalysts. • The ability to deprotonate unactivated C–H bonds opens the possibility of using this approach to prepare numerous high-volume targets. ...
... Conclusion • Very simple strategy for engaging CO2 in C–C bond formation that does not require synthetic or biological catalysts. • The ability to deprotonate unactivated C–H bonds opens the possibility of using this approach to prepare numerous high-volume targets. ...
A2 LEVEL CHEMISTRY 4.1.1 ARENES TEST Answer all questions
... A chemist was investigating the reactions of benzene, phenol and cyclohexene with bromine. She found that they all reacted with bromine but under different conditions. (a) ...
... A chemist was investigating the reactions of benzene, phenol and cyclohexene with bromine. She found that they all reacted with bromine but under different conditions. (a) ...
Lecture 7a
... The carboxylic acid is a poor electrophile (neutral) The alcohol is a poor nucleophile (neutral) The reaction is very slow at room temperature Any water in the reaction mixture lowers the yield significantly ...
... The carboxylic acid is a poor electrophile (neutral) The alcohol is a poor nucleophile (neutral) The reaction is very slow at room temperature Any water in the reaction mixture lowers the yield significantly ...
Organometallic Catalysts
... The establishment of a reaction mechanism is always a difficult task. It is even harder to definitively establish a catalytic cycle as all the reactions are going on in parallel! ...
... The establishment of a reaction mechanism is always a difficult task. It is even harder to definitively establish a catalytic cycle as all the reactions are going on in parallel! ...
Yearly Lesson Plan 2007
... 3.3 Understanding the reactivity series of metals and its application compare the differences in the vigour of the reactions of some metals with oxygen, deduce the reactivity series of metals, determine the position of carbon and hydrogen in the reactivity series of metals, state what the re ...
... 3.3 Understanding the reactivity series of metals and its application compare the differences in the vigour of the reactions of some metals with oxygen, deduce the reactivity series of metals, determine the position of carbon and hydrogen in the reactivity series of metals, state what the re ...
Pyrrolidine-2-carboxylic Acid (l
... involving Mannich reaction of acetone aldehydes and aryl amines to give β-amino ketones. In case of hydoxyacetone it gives α-hydroxy β-amino ketones in good to excellent ee. This reaction complements the Sharpless asymmetric aminohydroxylation.3–5,10 ...
... involving Mannich reaction of acetone aldehydes and aryl amines to give β-amino ketones. In case of hydoxyacetone it gives α-hydroxy β-amino ketones in good to excellent ee. This reaction complements the Sharpless asymmetric aminohydroxylation.3–5,10 ...
top 5 organic - No Brain Too Small
... Preparation method (alc + c.acid); heat under reflux; add carbonate to neutralise acids, separate ester from impurities by distillation Breaking – reaction with H2O - hydrolysis (break where you make – i.e between O and C=O) ...
... Preparation method (alc + c.acid); heat under reflux; add carbonate to neutralise acids, separate ester from impurities by distillation Breaking – reaction with H2O - hydrolysis (break where you make – i.e between O and C=O) ...
C h e m g u id e –... ACID ANHYDRIDES: REACTIONS WITH WATER, ALCOHOLS AND PHENOLS
... and so the top group in your target molecule must come from an acid anhydride, but a bigger one than ethanoic anhydride. You can ignore the other group on the ring as just a distraction. You haven’t come across any reaction which would attach a group like this to a benzene ring, so it must have been ...
... and so the top group in your target molecule must come from an acid anhydride, but a bigger one than ethanoic anhydride. You can ignore the other group on the ring as just a distraction. You haven’t come across any reaction which would attach a group like this to a benzene ring, so it must have been ...
Organic Synthesis Part 2
... LiAlH4, NaCNBH3 and LiEt3BH (Super hydride!) can all displace halides, tosylates and mesylates from primary and secondary alkyl positions. Since the reactions are SN2 in nature, this is a useful way of making stereospecifically deuterated compounds through the corresponding deutero reducing agents. ...
... LiAlH4, NaCNBH3 and LiEt3BH (Super hydride!) can all displace halides, tosylates and mesylates from primary and secondary alkyl positions. Since the reactions are SN2 in nature, this is a useful way of making stereospecifically deuterated compounds through the corresponding deutero reducing agents. ...
Background Information
... When condensation products (β-hydroxycarbonyl compounds) undergo E2 elimination, the resulting product is referred to as an α,β-unsaturated carbonyl. In the case of the dibenzalacetone reaction, the product is referred to as an α,β-unsaturated ketone. These products are very stable (due to resonance ...
... When condensation products (β-hydroxycarbonyl compounds) undergo E2 elimination, the resulting product is referred to as an α,β-unsaturated carbonyl. In the case of the dibenzalacetone reaction, the product is referred to as an α,β-unsaturated ketone. These products are very stable (due to resonance ...
Discuss on Reactions of Alcohols
... (K a value of 1 × 10 −15). Likewise, similar reactions occur with potassium metal. The acidity of alcohols decreases while going from primary to secondary to tertiary. This decrease in acidity is due to two factors: an increase of electron density on the oxygen atom of the more highly‐substituted al ...
... (K a value of 1 × 10 −15). Likewise, similar reactions occur with potassium metal. The acidity of alcohols decreases while going from primary to secondary to tertiary. This decrease in acidity is due to two factors: an increase of electron density on the oxygen atom of the more highly‐substituted al ...
Microsoft Word
... anti-Markovnikov fashion by a free radical mechanism.17 We have developed a new and catalytic method for the thiol addition across double bond using H-Rho-zeolite (Scheme 11). Figure ...
... anti-Markovnikov fashion by a free radical mechanism.17 We have developed a new and catalytic method for the thiol addition across double bond using H-Rho-zeolite (Scheme 11). Figure ...
Organic Chemistry I Laboratory
... More than a century ago at the University of Kazan, Vladimir Vasilevich Markovnikov and Alexander Zaitzev were investigating a chemical reaction both backward and forward. Markovnikov was adding hydrogen iodide to alkenes to prepare alkyl iodides, and Zaitzev was removing hydrogen iodide from alkyl ...
... More than a century ago at the University of Kazan, Vladimir Vasilevich Markovnikov and Alexander Zaitzev were investigating a chemical reaction both backward and forward. Markovnikov was adding hydrogen iodide to alkenes to prepare alkyl iodides, and Zaitzev was removing hydrogen iodide from alkyl ...
Organic Chemistry
... Synthesis of Single Enantiomers • In a second strategy, asymmetric induction, the achiral starting material is placed in a chiral environment by reacting it with a chiral auxiliary. Later it will be removed. • E. J. Corey used this chiral auxiliary to direct an asymmetric Diels-Alder reaction. ...
... Synthesis of Single Enantiomers • In a second strategy, asymmetric induction, the achiral starting material is placed in a chiral environment by reacting it with a chiral auxiliary. Later it will be removed. • E. J. Corey used this chiral auxiliary to direct an asymmetric Diels-Alder reaction. ...
وزارة التربية و التعليم العالي امتحانات شهادة الثانوية الع
... II- Chlorination Agent in Organic Chemistry Phosphorous pentachloride reacts, at room temperature, with a carboxylic acid A, having a saturated carbon chain, according to the reaction of the following equation: A + PCl 5 → B + HCl + POCl 3 . 1- Determine the molecular formula of A. Write the condens ...
... II- Chlorination Agent in Organic Chemistry Phosphorous pentachloride reacts, at room temperature, with a carboxylic acid A, having a saturated carbon chain, according to the reaction of the following equation: A + PCl 5 → B + HCl + POCl 3 . 1- Determine the molecular formula of A. Write the condens ...
Demonstrate skill in organic chemistry techniques.
... Analyze chemical reactions or processes to determine greenness. Learning Objectives Compare methods using parameters such as relative chemical hazards of reagents, environmental impact of materials and waste, transportation hazards, difficulty of synthetic method, and reaction efficiency. Perform an ...
... Analyze chemical reactions or processes to determine greenness. Learning Objectives Compare methods using parameters such as relative chemical hazards of reagents, environmental impact of materials and waste, transportation hazards, difficulty of synthetic method, and reaction efficiency. Perform an ...
Experiment 7 — Nucleophilic Substitution
... compound names, make predictions first. Heat or cool as necessary. (3) In your notebook, write a brief discussion of the results that addresses the following points — (a) What was the effect of substitution at the C undergoing nucleophilic attack, in particular, 1° vs 2° vs 3° alkyl. (b) Do the ally ...
... compound names, make predictions first. Heat or cool as necessary. (3) In your notebook, write a brief discussion of the results that addresses the following points — (a) What was the effect of substitution at the C undergoing nucleophilic attack, in particular, 1° vs 2° vs 3° alkyl. (b) Do the ally ...
Ene reaction

The ene reaction (also known as the Alder-ene reaction) is a chemical reaction between an alkene with an allylic hydrogen (the ene) and a compound containing a multiple bond (the enophile), in order to form a new σ-bond with migration of the ene double bond and 1,5 hydrogen shift. The product is a substituted alkene with the double bond shifted to the allylic position.This transformation is a group transfer pericyclic reaction, and therefore, usually requires highly activated substrates and/or high temperatures. Nonetheless, the reaction is compatible with a wide variety of functional groups that can be appended to the ene and enophile moieties. Also,many useful Lewis acid-catalyzed ene reactions have been developed which can afford high yields and selectivities at significantly lower temperatures, making the ene reaction a useful C–C forming tool for the synthesis of complex molecules and natural products.