The Hydroxylation of Aromatic Nitro Compounds by Alkalies
... should diminish the yield; but no such diminution occurs. The only remaining product is water; and this is now believed to render the potassium hydroxide incapable of further reaction by coating the surface. Wohl's statement that the hydroxylation proceeds in the absence of air Is true. but then the ...
... should diminish the yield; but no such diminution occurs. The only remaining product is water; and this is now believed to render the potassium hydroxide incapable of further reaction by coating the surface. Wohl's statement that the hydroxylation proceeds in the absence of air Is true. but then the ...
Chapter 12: Aldehydes, Ketones and Carboxylic acids
... (ii) Esterification: Carboxylic acids are esterified with alcohols in the presence of a mineral acid such as concentrated H SO or HCl gas as a ...
... (ii) Esterification: Carboxylic acids are esterified with alcohols in the presence of a mineral acid such as concentrated H SO or HCl gas as a ...
Investigating Esters
... The reaction is reversible and the reaction proceeds very slowly towards an equilibrium. It is difficult to achieve 100% conversion and the yield of the ester will not be high. It may possible to improve the yield by changing the reaction conditions. The methods of changing the position of the equil ...
... The reaction is reversible and the reaction proceeds very slowly towards an equilibrium. It is difficult to achieve 100% conversion and the yield of the ester will not be high. It may possible to improve the yield by changing the reaction conditions. The methods of changing the position of the equil ...
Investigating Esters
... The reaction is reversible and the reaction proceeds very slowly towards an equilibrium. It is difficult to achieve 100% conversion and the yield of the ester will not be high. It may possible to improve the yield by changing the reaction conditions. The methods of changing the position of the equil ...
... The reaction is reversible and the reaction proceeds very slowly towards an equilibrium. It is difficult to achieve 100% conversion and the yield of the ester will not be high. It may possible to improve the yield by changing the reaction conditions. The methods of changing the position of the equil ...
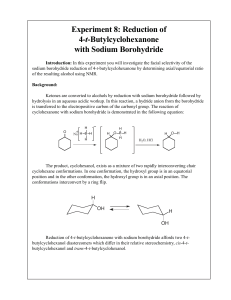
Experiment 8: Reduction of 4-t-Butylcyclohexanone with Sodium
... solubilities of the compound in the two solvents. Organic compounds are usually relatively more soluble in organic solvents than in water, hence they may be extracted from aqueous solutions. Several extractions may be needed to remove “all” of the organic compound from the aqueous layer, depending o ...
... solubilities of the compound in the two solvents. Organic compounds are usually relatively more soluble in organic solvents than in water, hence they may be extracted from aqueous solutions. Several extractions may be needed to remove “all” of the organic compound from the aqueous layer, depending o ...
Lecture 2 - Bonding in Organic Compounds
... • Alkanes are usually joined together by Van der Waals forces, which increase when the size of the alkane increases. • Their melting and boiling point also increases with size. • Alkanes are generally inert and liberate a lot of energy in combustion. (reaction with oxygen) • They are commonl ...
... • Alkanes are usually joined together by Van der Waals forces, which increase when the size of the alkane increases. • Their melting and boiling point also increases with size. • Alkanes are generally inert and liberate a lot of energy in combustion. (reaction with oxygen) • They are commonl ...
7. Alkenes: Reactions and Synthesis
... Osmium Tetroxide Catalyzed Formation of Diols Hydroxylation - converts to syn-diol Osmium tetroxide, then sodium bisulfate ...
... Osmium Tetroxide Catalyzed Formation of Diols Hydroxylation - converts to syn-diol Osmium tetroxide, then sodium bisulfate ...
Chem 30CL - Lecture 1c - UCLA Chemistry and Biochemistry
... and can be used as reactants or as part of a chiral catalyst or a chiral auxiliary • The TADDOL, DIOP and the Chiraphos ligand have tartaric acid as chiral backbone ...
... and can be used as reactants or as part of a chiral catalyst or a chiral auxiliary • The TADDOL, DIOP and the Chiraphos ligand have tartaric acid as chiral backbone ...
Introduction to Chemical Reactions
... 2 Mg + O2 → 2 MgO Magnesium atoms and oxygen gas molecules combine to form a single new product Magnesium oxide is the product of the reaction In a moment, we will also see that this reaction can also be classified as a combustion reaction ...
... 2 Mg + O2 → 2 MgO Magnesium atoms and oxygen gas molecules combine to form a single new product Magnesium oxide is the product of the reaction In a moment, we will also see that this reaction can also be classified as a combustion reaction ...
Preparation of Alcohols
... E: Addition of hydride reagents (LiAlH4, or NaBH4) or organometallic reagents (RMgBr or R-Li) yield alcohols. Attack of the nucleophile is usually on the less hindered carbon, and trans geometry usually is seen. Carey 15.4. F: Hydroxide attacks the less hindered carbon to give the trans diol. Carey ...
... E: Addition of hydride reagents (LiAlH4, or NaBH4) or organometallic reagents (RMgBr or R-Li) yield alcohols. Attack of the nucleophile is usually on the less hindered carbon, and trans geometry usually is seen. Carey 15.4. F: Hydroxide attacks the less hindered carbon to give the trans diol. Carey ...
論 文 の 内 容 の 要 旨 論文題目 Synthesis and Property of Low
... Since boron is group 13 element and it has three valence electrons, the oxidation state of most boron compounds is three. Boron(III) compounds are known as useful reagents in synthetic organic chemistry, for instance, hydroboranes for hydroboration and boronic acids for Suzuki-Miyaura cross-coupling ...
... Since boron is group 13 element and it has three valence electrons, the oxidation state of most boron compounds is three. Boron(III) compounds are known as useful reagents in synthetic organic chemistry, for instance, hydroboranes for hydroboration and boronic acids for Suzuki-Miyaura cross-coupling ...
Alkanes CH4 + Cl2 → CH3Cl + HCl CH3CH3 + Cl2 → CH3CH2Cl +
... atom goes on the atom with more H atoms to begin with. This will be studied more in a later unit, just remember it for now. It is Markovnikov’s rule. Substances that add to C=C bonds, such as H2, Br2, and HBr are termed electrophiles. Definition: An electrophile is an electron deficient species that ...
... atom goes on the atom with more H atoms to begin with. This will be studied more in a later unit, just remember it for now. It is Markovnikov’s rule. Substances that add to C=C bonds, such as H2, Br2, and HBr are termed electrophiles. Definition: An electrophile is an electron deficient species that ...
CHAPTER 12 Solid-Phase Synthesis of Peptides Containing the
... the peptide. These changes can help stabilize the peptide against proteases, disrupt or induce secondary structural elements, and eliminate hydrogen bonds. In addition to these approaches, replacement of peptide bonds by peptide bond surrogates, such as a CH,NH reduced peptide bond, can also be perf ...
... the peptide. These changes can help stabilize the peptide against proteases, disrupt or induce secondary structural elements, and eliminate hydrogen bonds. In addition to these approaches, replacement of peptide bonds by peptide bond surrogates, such as a CH,NH reduced peptide bond, can also be perf ...
000217986-Tajbakhsh_et_al_
... Yields refer to isolated products. c The reaction was conducted at reflux temperature. b ...
... Yields refer to isolated products. c The reaction was conducted at reflux temperature. b ...
Chem 30 Diploma Review
... • Monomers combine to form a polymer and a bi-product. Each time a bond forms between monomers, small molecules, such as water, ammonia, or HCl are “condensed” out. • The polymerization of nylon: ...
... • Monomers combine to form a polymer and a bi-product. Each time a bond forms between monomers, small molecules, such as water, ammonia, or HCl are “condensed” out. • The polymerization of nylon: ...
No Slide Title
... • Primary amines and carbonyls form Imines (also called Schiff base) – examples include, hydrazones, phenylhydrazones, 2,4dinitrophenylhydrazones, semicarbazones, tosylhydrazones, oximes ...
... • Primary amines and carbonyls form Imines (also called Schiff base) – examples include, hydrazones, phenylhydrazones, 2,4dinitrophenylhydrazones, semicarbazones, tosylhydrazones, oximes ...
biological sulfate reduction in alkaline waters for the reprocessing of
... purge streams are disposed of in large tailings ponds. The sodium carbonate levels in these ponds are quite high (8 to 14 weight percent); thus, after 50 years of operation, these ponds represent a significant potential source of sodium carbonate. Unfortunately, moderate levels of sodium sulfate (1 ...
... purge streams are disposed of in large tailings ponds. The sodium carbonate levels in these ponds are quite high (8 to 14 weight percent); thus, after 50 years of operation, these ponds represent a significant potential source of sodium carbonate. Unfortunately, moderate levels of sodium sulfate (1 ...
A Floral Fragrance, Methyl Benzoate
... Cool the solution to room temperature, then decant it into a separatory funnel containing 25 mL of water, and rinse the flask with 25 mL of diethyl ether (use wet ether found in a supply bottle in each hood). Add this ether to the separatory funnel, shake thoroughly, and drain off the water layer, w ...
... Cool the solution to room temperature, then decant it into a separatory funnel containing 25 mL of water, and rinse the flask with 25 mL of diethyl ether (use wet ether found in a supply bottle in each hood). Add this ether to the separatory funnel, shake thoroughly, and drain off the water layer, w ...
Correlation Between Acidity, Basicity and Catalytic Performance of
... Results and discussion The sol-gel magnesia-alumina oxides show a surface area ranging from 237 to 323 m2/g with bimodel pore size distribution. Acid and basic sites coexist on the surface of the samples, depending on the molar ratio of MgO/Al2O3. When isopropanol was decomposed over the magnesia-al ...
... Results and discussion The sol-gel magnesia-alumina oxides show a surface area ranging from 237 to 323 m2/g with bimodel pore size distribution. Acid and basic sites coexist on the surface of the samples, depending on the molar ratio of MgO/Al2O3. When isopropanol was decomposed over the magnesia-al ...
Document
... Elimination occurs instead of substitution when pathway of nucleophile is blocked, hence the Lewis base will remove an electrophile from the haloalkane instead ...
... Elimination occurs instead of substitution when pathway of nucleophile is blocked, hence the Lewis base will remove an electrophile from the haloalkane instead ...
EXPERIMENT 5 (Organic Chemistry II) Pahlavan/Cherif
... Continue the distillation until only a few milliliters (< 2 ml) of high-boiling residue remain in the flask. If white fumes appear near the end of the distillation, stop heating a once by lowering the heating mantle. (NOTE - these fumes are oxides of sulfur, SO2 , if sulfuric acid is being used). No ...
... Continue the distillation until only a few milliliters (< 2 ml) of high-boiling residue remain in the flask. If white fumes appear near the end of the distillation, stop heating a once by lowering the heating mantle. (NOTE - these fumes are oxides of sulfur, SO2 , if sulfuric acid is being used). No ...
Dehydration of Alcohols - Dehydration of Cyclohexanol
... of cyclohexanol as starting material is based on the following considerations: a) Because of its structure, cyclohexene can give only one alkene upon dehydration, normally cyclohexene. b) The rate of dehydration of cyclohexanol using 85% phosphoric acid is conveniently fast. c) The product is easily ...
... of cyclohexanol as starting material is based on the following considerations: a) Because of its structure, cyclohexene can give only one alkene upon dehydration, normally cyclohexene. b) The rate of dehydration of cyclohexanol using 85% phosphoric acid is conveniently fast. c) The product is easily ...
SOLVENT-FREE SYNTHESIS OF CHALCONE BY ALDOL
... Chalcones represent a group of compounds with interesting biological activities that are formed from an aldol condensation between a benzaldehyde and an acetophenone in the presence of NaOH as a catalyst. Although traditionally synthesized using aqueous sodium hydroxide in organic solvents, in this ...
... Chalcones represent a group of compounds with interesting biological activities that are formed from an aldol condensation between a benzaldehyde and an acetophenone in the presence of NaOH as a catalyst. Although traditionally synthesized using aqueous sodium hydroxide in organic solvents, in this ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.