* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Preparation of Alcohols
Discodermolide wikipedia , lookup
Elias James Corey wikipedia , lookup
Kinetic resolution wikipedia , lookup
Asymmetric induction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Hydroformylation wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Preparation of Alcohols
Alcohols can be formed from many, many different sources. More than 7
different functional groups can be converted to an alcohol.
Ethers
R O
R
Alkenes
CH CH2
Alkyl Halides
R—X
B
A
Ketones
Aldehydes
O
R C
R'
R'
C
D
Epoxides
O
R
E, F
Carboxylic Acids
O
R C
O H
G
Alcohols
R—OH
Diols
HO
OH
R2 C CR2
Esters
O
R C
O R'
H
Legend:
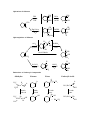
A: Substitution only. Hydroxide nucleophile; difficult reaction because of competing elimination (E2).
See Carey Chpt. 8.
B: Hydration can go in two ways to give different regioselectivities. Oxymercuration-demercuration gives
Markovnikov product, hydroboration/oxidation gives opposite. Generally clean and high-yielding.
Carey 6.10-11.
D i o l s ("vicinal" diols) also can be formed by two complementary sequences. Hydroxylation (KMnO4
or OsO4 with reductive cleavage) yields vicinal diols with syn addition (Carey 15.5). Epoxidation
followed by cleavage (peracid, then acid or basic cleavage) yields anti- or trans addition.
C: Benzyl ethers can be cleaved by hydrogenolysis (H2/Pt or Pd) to give the alcohol.
R O
H2
H
R O
+ CH
CH2
2
H
Pt or Pd
D: Addition of hydride reagents (LiAlH4, or NaBH4) or organometallic reagents (RMgBr or R-Li) yield
alcohols. The process converts the carbonyl carbon to a carbinol carbon (1° from reduction of
aldehyde; 2° from reduction of ketone or alkylation of aldehyde; 3° from alkylation of ketone). Carey
15.2.
E: Addition of hydride reagents (LiAlH4, or NaBH4) or organometallic reagents (RMgBr or R-Li) yield
alcohols. Attack of the nucleophile is usually on the less hindered carbon, and trans geometry
usually is seen. Carey 15.4.
F: Hydroxide attacks the less hindered carbon to give the trans diol. Carey 16.12-13.
G: Alcohols are formed by the use of hydride reagents: LiAlH4, BH3. NaBH4 doesn't work (not
reactive enough), even though BH3 does. Go figure. Carey Table 15.3.
H: Two ways to do this, depending on which part of the
R'
H
ester should yield the alcohol. First, simple
H2O
O O
hydrolysis of an ester gives an alcohol and an acid.
O O + HO
C
R
Second, reduction of an ester gives two alcohol
products. Often, with simple esters, one of the
alcohols is small, water soluble or volatile, and can
be separated easily from the other (Carey Table
15.3.):
+
-
H or OH
C
R
R'
O
R C
O R'
R CH2 O H
+
HO R'
Hydration of Alkenes
CH3
H
BH3
THF
H2 O2
BR2
NaOH
Hg(OAc)2
CH3
OH
NaBH4
H2 O
H
HgOAc
CH3
H
H
CH3
H
OH
H
CH3
OH
H
H
Hydroxylation of Alkenes
H
OsO4
Pyridine
O
O Os
NaHSO3
O
O
H2 O
CH3
H
H
Basic KMnO 4
OH
OH
CH3
CH3
H
RCO3 H
H
NaOH
O
CH2 Cl2
H2 O
CH3
OH
OH
CH3
Reduction of Carbonyl Compounds
Aldehydes
Ketones
Esters
Carboxylic Acids
O
H3C
CH
H2
C
H3C
O
O
H3C
CH
H3C
OH
NaBH4
CH3 OH
OH
C
H2
OH
O
H3 C(H2 C)8 C
C
H
NaBH4
CH3 OH
H2
C
C O CH3
LiAlH4
ether
H2
C OH
+ HO CH3
LiAlH4
ether
H3 C(H2 C)8 CH2
OH