Organic Chemistry
... selectivity with the result that enzyme-catalyzed reactions invariably give only one of all possible stereoisomers. ...
... selectivity with the result that enzyme-catalyzed reactions invariably give only one of all possible stereoisomers. ...
Alcohol oxidation
... The regiospecificity of the reaction depends on the relative migratory ability of the substituents attached to the carbonyl. Substituents which are able to stabilize a positive charge migrate more readily, so that the order of preference is: tert. alkyl > cyclohexyl > sec. alkyl > phenyl > prim. alk ...
... The regiospecificity of the reaction depends on the relative migratory ability of the substituents attached to the carbonyl. Substituents which are able to stabilize a positive charge migrate more readily, so that the order of preference is: tert. alkyl > cyclohexyl > sec. alkyl > phenyl > prim. alk ...
-23- ORGANIC CHEMISTRY A. STRUCTURE AND ISOMERISM 1
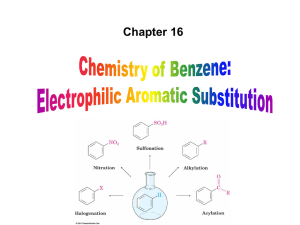
... (b) halogenation (addition) - Br2 or Cl2 (c) hydrohalogenation (addition) - HBr or HCl Aromatic compounds (a) halogenation (substitution)- Br2 or Cl2/Fe (b) conversion of alkyl substitutents to COOH (oxidation) - hot KMnO4 ...
... (b) halogenation (addition) - Br2 or Cl2 (c) hydrohalogenation (addition) - HBr or HCl Aromatic compounds (a) halogenation (substitution)- Br2 or Cl2/Fe (b) conversion of alkyl substitutents to COOH (oxidation) - hot KMnO4 ...
Haloalkanes
... The Environmental problems associated with CFC Because of the lack of reactivity and insolubility in water, there is no natural process for removing CFC. In fact they drift up into the stratosphere (the upper atmosphere), where they receive sufficient ultraviolet Light to cause photolysis. The chlor ...
... The Environmental problems associated with CFC Because of the lack of reactivity and insolubility in water, there is no natural process for removing CFC. In fact they drift up into the stratosphere (the upper atmosphere), where they receive sufficient ultraviolet Light to cause photolysis. The chlor ...
Alcohols , Phenols and Ethers
... phenoxide ion. But alcohols can be converted into sodium alkoxide ion by reaction with sodium metal only. [Alkyl halide used here can not be aryl halide as it does not undergo nucleophilic substitution easily] R – X can be X = –Cl, –Br, I, (b) ...
... phenoxide ion. But alcohols can be converted into sodium alkoxide ion by reaction with sodium metal only. [Alkyl halide used here can not be aryl halide as it does not undergo nucleophilic substitution easily] R – X can be X = –Cl, –Br, I, (b) ...
3.8 Aldehydes and ketones
... In practise, KCN is added. This is because HCN is a weak acid (partially dissociates) giving a low [CN-] ...
... In practise, KCN is added. This is because HCN is a weak acid (partially dissociates) giving a low [CN-] ...
Answers - Final Exam 2013
... a. An aqueous solution of H2CrO4 and H2SO4 oxidizes primary alcohols to aldehydes and secondary alcohols to ketones b. An alkene will form the same epoxide upon treatment with either Br2 and H2O followed by base, or with MCPBA c. Rearrangements may occur during SN1 reactions, but not during SN2 reac ...
... a. An aqueous solution of H2CrO4 and H2SO4 oxidizes primary alcohols to aldehydes and secondary alcohols to ketones b. An alkene will form the same epoxide upon treatment with either Br2 and H2O followed by base, or with MCPBA c. Rearrangements may occur during SN1 reactions, but not during SN2 reac ...
THE CARBON-CARBON DOUBLE BOND
... nitrogen - amides with alkyl or other substituents on N cannot be dehydrated. (2) SOCl2 is thionyl chloride and POCl3 is phosphorus oxychloride. Both are powerful dehydrating agents. Reactivity of Nitriles: ...
... nitrogen - amides with alkyl or other substituents on N cannot be dehydrated. (2) SOCl2 is thionyl chloride and POCl3 is phosphorus oxychloride. Both are powerful dehydrating agents. Reactivity of Nitriles: ...
ch15 by dr Dina
... Chloro and bromo groups are weakly deactivating but are also ortho, para directors ...
... Chloro and bromo groups are weakly deactivating but are also ortho, para directors ...
CI 12.4 - Sackville School
... The carbocation has a C with a positive charge; that C had a half share of the two electrons in the bond which was broken. It now has no share of that pair and is now a C atom with one less electron so it has a single positive charge. ...
... The carbocation has a C with a positive charge; that C had a half share of the two electrons in the bond which was broken. It now has no share of that pair and is now a C atom with one less electron so it has a single positive charge. ...
Practice Problem - HCC Southeast Commons
... stabilized (lower in energy) – The positive charge is directly on the alkylsubstituted carbon (3o carbon) and is stabilized by the inductive electron-donating effect of the alkyl group ...
... stabilized (lower in energy) – The positive charge is directly on the alkylsubstituted carbon (3o carbon) and is stabilized by the inductive electron-donating effect of the alkyl group ...
Chapter 18 – Carbonyl Compounds II (Last Chapter we mostly talk
... nucleophile. This reaction was first discovered in the 1800’s by Hugo Schiff a German Chemist, and the product of this reaction is often referred to as a Schiff base). *** ppt *** Fill in the Reagent of Product in the Box ...
... nucleophile. This reaction was first discovered in the 1800’s by Hugo Schiff a German Chemist, and the product of this reaction is often referred to as a Schiff base). *** ppt *** Fill in the Reagent of Product in the Box ...
Isoborneol from Camphor: A Reduction
... 1. Explain why the gem-dimethyl groups appear as separate peaks in the proton NMR spectrum of isoborneol while they are an unresolved singlet in borneol. 2. The reduction mechanism is often shown with a hydride ion (H:-) attacking the carbonyl carbon. Why might one assign a partial negative charge t ...
... 1. Explain why the gem-dimethyl groups appear as separate peaks in the proton NMR spectrum of isoborneol while they are an unresolved singlet in borneol. 2. The reduction mechanism is often shown with a hydride ion (H:-) attacking the carbonyl carbon. Why might one assign a partial negative charge t ...
CH 19
... • Addition of amines with an atom containing a lone pair of electrons on the adjacent atom occurs very readily, giving useful, stable imines • For example, hydroxylamine forms oximes and 2,4-dinitrophenylhydrazine readily forms 2,4dinitrophenylhydrazones – These are usually solids and help in ...
... • Addition of amines with an atom containing a lone pair of electrons on the adjacent atom occurs very readily, giving useful, stable imines • For example, hydroxylamine forms oximes and 2,4-dinitrophenylhydrazine readily forms 2,4dinitrophenylhydrazones – These are usually solids and help in ...
Synthesis of n-Butyl Acetate via Esterification
... we can, by Le Chatelier's principle, increase the concentration of either the alcohol or acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester ...
... we can, by Le Chatelier's principle, increase the concentration of either the alcohol or acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester ...
Properties of haloalkanes
... Primary haloalkanes do not react with water, but react to form alcohols with aqueous sodium hydroxide. Formation of amines Another nucleophilic substitution reaction occurs between haloalkanes and alcoholic ammonia: R—X + NH3(alc) → R—NH2 + HX amine It must be alcoholic ammonia: if water is present ...
... Primary haloalkanes do not react with water, but react to form alcohols with aqueous sodium hydroxide. Formation of amines Another nucleophilic substitution reaction occurs between haloalkanes and alcoholic ammonia: R—X + NH3(alc) → R—NH2 + HX amine It must be alcoholic ammonia: if water is present ...
Exam 1 Review Sheet Chapter 15 Chemistry 110b
... Acetals and ketals and their hemi- forms. Be able to identify these functional groups. Know the mechanism of formation/hydrolysis for an acetal (e.g., acetaldehyde + 2CH3OH + H+(cat.)). Be aware of the utility of acetals and ketals [and their thio (sulfur) analogs] as protecting groups in organic sy ...
... Acetals and ketals and their hemi- forms. Be able to identify these functional groups. Know the mechanism of formation/hydrolysis for an acetal (e.g., acetaldehyde + 2CH3OH + H+(cat.)). Be aware of the utility of acetals and ketals [and their thio (sulfur) analogs] as protecting groups in organic sy ...
EXPERIMENT 4 Objectives Principles
... of electrons, or ions with a negative charge. They can form bond by donating electrons to another molecule having a position of lower electron density (electrophiles). Examples of nucleophilic species are: water, amine, ammonia, cyanide ion, alkoxide ion, and hydroxide ion. Alkyl halides can react w ...
... of electrons, or ions with a negative charge. They can form bond by donating electrons to another molecule having a position of lower electron density (electrophiles). Examples of nucleophilic species are: water, amine, ammonia, cyanide ion, alkoxide ion, and hydroxide ion. Alkyl halides can react w ...
alcohols - Knockhardy
... protonation of the alcohol using a lone pair on oxygen loss of a water molecule to generate a carbocation (carbonium ion) a bromide ion behaves as a nucleophile and attacks the carbocation ...
... protonation of the alcohol using a lone pair on oxygen loss of a water molecule to generate a carbocation (carbonium ion) a bromide ion behaves as a nucleophile and attacks the carbocation ...
Organic molecules with functional groups containing oxygen
... • The arrows are double headed in this case, indicating the movement of a pair of electrons • The arrows begin at a definite pair of electrons - a bond or a lone pair – and move towards a positive charge • If they move into the space between two atoms, a bond is formed • If they move out of the spac ...
... • The arrows are double headed in this case, indicating the movement of a pair of electrons • The arrows begin at a definite pair of electrons - a bond or a lone pair – and move towards a positive charge • If they move into the space between two atoms, a bond is formed • If they move out of the spac ...
Substitution and Elimination Reactions . 7.1. Definitions.
... attached; that is, the alkyl group in the alkyl halide is either Me, primary (1°, two H's), or secondary (2°, one H), but never tertiary (3°, no H's). The last phenomenon is due to steric hindrance of the SN2 substitution reaction. We can draw a reaction coordinate diagram for the SN2 reaction. A re ...
... attached; that is, the alkyl group in the alkyl halide is either Me, primary (1°, two H's), or secondary (2°, one H), but never tertiary (3°, no H's). The last phenomenon is due to steric hindrance of the SN2 substitution reaction. We can draw a reaction coordinate diagram for the SN2 reaction. A re ...
Ch 10 Haloalkanes n haloarenes
... Tert-butyl chloride reacts via SN¹ mechanism because the heterolytic cleavage of C — Cl bond in tert-butyl chloride gives 3 carbocation which is highly stable and favourable for SN¹ mechanism. Moreover, tert-butyl chloride (3°) bring a bulky molecule has steric hindrance which will not allow SN² mec ...
... Tert-butyl chloride reacts via SN¹ mechanism because the heterolytic cleavage of C — Cl bond in tert-butyl chloride gives 3 carbocation which is highly stable and favourable for SN¹ mechanism. Moreover, tert-butyl chloride (3°) bring a bulky molecule has steric hindrance which will not allow SN² mec ...
Alcohols and Phenols
... Unusually high boiling points due to hydrogen bonding between molecules ...
... Unusually high boiling points due to hydrogen bonding between molecules ...
exam3 answers - Moorpark College
... A. List all the intermolecular forces that need to be overcome for the compound A given below to boil? ...
... A. List all the intermolecular forces that need to be overcome for the compound A given below to boil? ...
chemistry- sch4u - final exam
... e. impossible to tell from this information ____ 59. Consider the above reaction mechanism. The reaction intermediates are formed in a. elementary step 1 b. elementary step 2 c. elementary step 3 d. elementary steps 1 and 2 e. impossible to tell from this information ____ 60. Consider the above reac ...
... e. impossible to tell from this information ____ 59. Consider the above reaction mechanism. The reaction intermediates are formed in a. elementary step 1 b. elementary step 2 c. elementary step 3 d. elementary steps 1 and 2 e. impossible to tell from this information ____ 60. Consider the above reac ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.