what are acyl chlorides?
... Waals dispersion forces. However, it doesn't form hydrogen bonds. Its boiling point is therefore higher than, say, an alkane of similar size (which has no permanent dipoles), but not as high as a similarly sized alcohol (which forms hydrogen bonds in addition to everything else.) ...
... Waals dispersion forces. However, it doesn't form hydrogen bonds. Its boiling point is therefore higher than, say, an alkane of similar size (which has no permanent dipoles), but not as high as a similarly sized alcohol (which forms hydrogen bonds in addition to everything else.) ...
ALKANE ALKYL HALIDE Halogenation of Alkanes
... alkoxides are formed by reacting alcohols with NaH, Na (methyl, 1o) or K (2o) 3o alcohols cannot be used as they are too bulky to react in ...
... alkoxides are formed by reacting alcohols with NaH, Na (methyl, 1o) or K (2o) 3o alcohols cannot be used as they are too bulky to react in ...
Ch 12- 13 - Phillips Scientific Methods
... Alcohol is produced on treatment of the alkene with water in the presence of a strong acid catalyst, such as H2SO4. Markovnikov’s rule can be used to predict the product when water adds to an unsymmetrically substituted alkene. Hydrated alkenes produce alcohols. ...
... Alcohol is produced on treatment of the alkene with water in the presence of a strong acid catalyst, such as H2SO4. Markovnikov’s rule can be used to predict the product when water adds to an unsymmetrically substituted alkene. Hydrated alkenes produce alcohols. ...
Chapter 18 Ketones and Aldehydes 1) Which of the following
... 22) Which of the following represents the correct ranking in terms of increasing boiling point? A) n-butane < 1-butanol < diethyl ether < 2-butanone B) n-butane < 2-butanone < diethyl ether < 1-butanol C) 2-butanone < n-butane < diethyl ether < 1-butanol D) n-butane < diethyl ether < 1-butanol < 2-b ...
... 22) Which of the following represents the correct ranking in terms of increasing boiling point? A) n-butane < 1-butanol < diethyl ether < 2-butanone B) n-butane < 2-butanone < diethyl ether < 1-butanol C) 2-butanone < n-butane < diethyl ether < 1-butanol D) n-butane < diethyl ether < 1-butanol < 2-b ...
CHEM 2414
... a cycloalkane composed of n carbons is CnH2n. Although a cycloalkane has two fewer hydrogens than the equivalent alkane, each carbon is bonded to four other atoms so such compounds are still considered to be ...
... a cycloalkane composed of n carbons is CnH2n. Although a cycloalkane has two fewer hydrogens than the equivalent alkane, each carbon is bonded to four other atoms so such compounds are still considered to be ...
File - Mr Weng`s IB Chemistry
... • The rate determining step (slow step) in an SN1reaction depends only on the concentration of the halogenoalkane, rate = k[halogenoalkane]. For SN2, rate = k[halogenoalkane][nucleophile]. SN2 is stereospecific with an inversion of configuration at the carbon. •SN2 reactions are best conducted using ...
... • The rate determining step (slow step) in an SN1reaction depends only on the concentration of the halogenoalkane, rate = k[halogenoalkane]. For SN2, rate = k[halogenoalkane][nucleophile]. SN2 is stereospecific with an inversion of configuration at the carbon. •SN2 reactions are best conducted using ...
The Infinite Variety of Carbon Compounds
... All alcohols are toxic. Methanol for instance is oxidized to formaldehyde by liver enzymes. It can lead to blindness and death. Even ethanol is toxic. The effects of drinking ethanol are due to its toxicity. Drunk driving, alcoholism, and fetal alcohol syndrome are all effects due to the toxicity of ...
... All alcohols are toxic. Methanol for instance is oxidized to formaldehyde by liver enzymes. It can lead to blindness and death. Even ethanol is toxic. The effects of drinking ethanol are due to its toxicity. Drunk driving, alcoholism, and fetal alcohol syndrome are all effects due to the toxicity of ...
Aldehydes Ketones
... Conjugate Nucleophilic Addition to α-β-Unsaturated Aldehydes and Ketones Direct addition (aka 1,2 addition) occurs when a nucleophile attacks the carbon in the carbonyl directly. Conjugate addition (aka 1,4 addition) occurs when the nucleophile attacks the carbonyl indirectly by attacking the s ...
... Conjugate Nucleophilic Addition to α-β-Unsaturated Aldehydes and Ketones Direct addition (aka 1,2 addition) occurs when a nucleophile attacks the carbon in the carbonyl directly. Conjugate addition (aka 1,4 addition) occurs when the nucleophile attacks the carbonyl indirectly by attacking the s ...
Drawing Organic Structures Functional Groups Constitutional Isomers
... 3. Polarizability of electrons (ease of distribution of electron density) • I > Br > Cl > F ...
... 3. Polarizability of electrons (ease of distribution of electron density) • I > Br > Cl > F ...
Ch 23 Carbonyl Condensations
... - The first part is an alpha substitution, where the deprotonated C is the Nu and a + carbon (generally a carbonyl) on another molecule is the E+. - If the other molecule’s carbonyl has no LG, the reaction proceeds as Nu addition. - If the carbonyl does have an LG, the reaction proceeds as Nu sub ...
... - The first part is an alpha substitution, where the deprotonated C is the Nu and a + carbon (generally a carbonyl) on another molecule is the E+. - If the other molecule’s carbonyl has no LG, the reaction proceeds as Nu addition. - If the carbonyl does have an LG, the reaction proceeds as Nu sub ...
Electrophilic Additions: Alkenes Addition of Hydrogen Halides
... In a regioselective reaction, one constitutional isomer is the major or the only product. I: early transition state (Like reactants) ...
... In a regioselective reaction, one constitutional isomer is the major or the only product. I: early transition state (Like reactants) ...
organic synthesis
... Chemical synthesis involves the preparation of new compounds from others. Many industrial processes involve a multi stage process where functional groups are converted into other functional groups. When planning a synthetic route, chemists must consider... • the reagents required to convert one func ...
... Chemical synthesis involves the preparation of new compounds from others. Many industrial processes involve a multi stage process where functional groups are converted into other functional groups. When planning a synthetic route, chemists must consider... • the reagents required to convert one func ...
dipole/induced-dipole and dipole/induced
... Mesomeric effect: The effect is used in a qualitative way and describes the electron withdrawing or releasing properties of substituents based on relevant resonance structures and is symbolized by the letter M. The mesomeric effect is a permanent effect and operates in compounds containing at least ...
... Mesomeric effect: The effect is used in a qualitative way and describes the electron withdrawing or releasing properties of substituents based on relevant resonance structures and is symbolized by the letter M. The mesomeric effect is a permanent effect and operates in compounds containing at least ...
unit 17 organic compounds containing oxygen and nitrogen atoms
... synthetic organic chemistry as a means of carbonyl group protection. In some chemical reactions one functional group may interfere with intended reaction elsewhere in a complex molecule. We can often circumvent the problem in such cases by first protecting the interfering functional group, carrying ...
... synthetic organic chemistry as a means of carbonyl group protection. In some chemical reactions one functional group may interfere with intended reaction elsewhere in a complex molecule. We can often circumvent the problem in such cases by first protecting the interfering functional group, carrying ...
Nuggets of Knowledge for Chapter 14 – Ethers
... anesthetic, but has now been replaced by safer compounds like halothane. o Another common ether is methyl tert-butyl ether, usually called MTBE. It has been added to gasoline for many years to increase the octane rating, but recently environmental concerns have decreased its use. ...
... anesthetic, but has now been replaced by safer compounds like halothane. o Another common ether is methyl tert-butyl ether, usually called MTBE. It has been added to gasoline for many years to increase the octane rating, but recently environmental concerns have decreased its use. ...
Gas Chromatography: Analyzing Alkene Isomers David L. Flanigan
... the E1 mechanism. In order for the elimination to occur a good leaving group and a base sufficiently strong enough to remove a β-proton must be present. The hydroxyl group is not a good leaving group. To enhance its leaving potential it must be transformed into a good leaving group. This is accompli ...
... the E1 mechanism. In order for the elimination to occur a good leaving group and a base sufficiently strong enough to remove a β-proton must be present. The hydroxyl group is not a good leaving group. To enhance its leaving potential it must be transformed into a good leaving group. This is accompli ...
Elimination Reactions
... elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucleophiles. Substitution and elimination compete. In 3° and 2° alkyl halides, E2 ...
... elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucleophiles. Substitution and elimination compete. In 3° and 2° alkyl halides, E2 ...
Carboxylic Acids - BSAK Chemistry weebly
... happens in two steps. In the first, the carbonoxygen double bond reforms and a chloride ion is pushed off. ...
... happens in two steps. In the first, the carbonoxygen double bond reforms and a chloride ion is pushed off. ...
10. Alkyl Halides
... to break, a new carbon-carbon pi bond begins to form, and the leaving group begins to depart ...
... to break, a new carbon-carbon pi bond begins to form, and the leaving group begins to depart ...
Synthesis of Benzene Derivatives: Electrophilic Aromatic Substitution
... restored. This process is more favored than the nucleophilic trapping by the anion accompanying E+. ...
... restored. This process is more favored than the nucleophilic trapping by the anion accompanying E+. ...
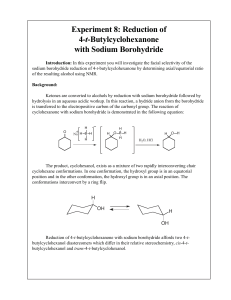
Experiment 8: Reduction of 4-t-Butylcyclohexanone with Sodium
... simply: Will the substance dissolve in a particular solvent? How much of it will dissolve? You have already seen differences in solubility used to purify compounds by recrystallization. Extraction is another common way that chemists use differences in solubility to separate or purify compounds. Our ...
... simply: Will the substance dissolve in a particular solvent? How much of it will dissolve? You have already seen differences in solubility used to purify compounds by recrystallization. Extraction is another common way that chemists use differences in solubility to separate or purify compounds. Our ...
Inorganic and organic chemistry 1
... this reaction is electrophilic addition, which has a carbocation present in the transition state, which is therefore not neutral. The hydrogen bromide splits into two ions. There is only one position for the bromine atom. Hence there are no isomers formed. ...
... this reaction is electrophilic addition, which has a carbocation present in the transition state, which is therefore not neutral. The hydrogen bromide splits into two ions. There is only one position for the bromine atom. Hence there are no isomers formed. ...
Derivatives of carboxylic acids - amides, acid anhydrides and nitriles
... In every example of this kind, the -OH group will be on the number 2 carbon atom - the one next to the CN group. The reaction isn't normally done using hydrogen cyanide itself, because this is an extremely poisonous gas. Instead, the aldehyde or ketone is mixed with a solution of sodium or potassium ...
... In every example of this kind, the -OH group will be on the number 2 carbon atom - the one next to the CN group. The reaction isn't normally done using hydrogen cyanide itself, because this is an extremely poisonous gas. Instead, the aldehyde or ketone is mixed with a solution of sodium or potassium ...
Inorganic and organic chemistry 1
... this reaction is electrophilic addition, which has a carbocation present in the transition state, which is therefore not neutral. The hydrogen bromide splits into two ions. There is only one position for the bromine atom. Hence there are no isomers formed. ...
... this reaction is electrophilic addition, which has a carbocation present in the transition state, which is therefore not neutral. The hydrogen bromide splits into two ions. There is only one position for the bromine atom. Hence there are no isomers formed. ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.