Glycosyl amines
... Fischer glycosidation • The method of preparation of glycosides from free aldoses or ketoses and aliphatic alcohols in the presence of anhydrous acids, usually hydrogen chloride. • In the course of the reaction a decrease in concentration of the starting aldose or ketose (in general, glycose) is acc ...
... Fischer glycosidation • The method of preparation of glycosides from free aldoses or ketoses and aliphatic alcohols in the presence of anhydrous acids, usually hydrogen chloride. • In the course of the reaction a decrease in concentration of the starting aldose or ketose (in general, glycose) is acc ...
4.8 Preparation of Alkyl Halides from Alcohols and Hydrogen
... electrons in this σ bond can be shared by positively charged carbon because the s orbital can overlap with the empty 2p orbital of positively charged carbon ...
... electrons in this σ bond can be shared by positively charged carbon because the s orbital can overlap with the empty 2p orbital of positively charged carbon ...
Chapter 17 Amines
... Secondary amines: both aryl amines and alkyl amines react with nitrous acid to yield N-nitrosoamines(亚硝胺) N O NHCH3 + NaNO2 + 2 HCl ...
... Secondary amines: both aryl amines and alkyl amines react with nitrous acid to yield N-nitrosoamines(亚硝胺) N O NHCH3 + NaNO2 + 2 HCl ...
Full Text - Journal of the Indian Institute of Science
... Step 1: Anhydrous methanol in dry benzene is added dropwise to a solution of chlorosulfonyl isocyanate in benzene at 25–30° C over a period of 0.5 h. The mixture is stirred for 0.5 h before olefin-free hexane is added and the flask is cooled to 0−5° C. The moisture-sensitive product is removed by fi ...
... Step 1: Anhydrous methanol in dry benzene is added dropwise to a solution of chlorosulfonyl isocyanate in benzene at 25–30° C over a period of 0.5 h. The mixture is stirred for 0.5 h before olefin-free hexane is added and the flask is cooled to 0−5° C. The moisture-sensitive product is removed by fi ...
aldehydes and ketones
... Carbonyl carbon of both aldehyde and ketones is sp2 – hybridised, One of the three sp2 hybridised orbital get involved in σ- bond formation with half –filled p-orbital of oxygen atom whereas rest of the two are consumed in σ-bond formation with hydrogen and carbon depending on the structure of ald ...
... Carbonyl carbon of both aldehyde and ketones is sp2 – hybridised, One of the three sp2 hybridised orbital get involved in σ- bond formation with half –filled p-orbital of oxygen atom whereas rest of the two are consumed in σ-bond formation with hydrogen and carbon depending on the structure of ald ...
Revised organic chemistry
... Some of the important reactions of alkanes are following. 1) Halogenations: This involves replacement of hydrogen atom by halogen atom. a) Chlorination: Alkanes react with chlorine in presence U.V light or diffused sunlight or at temperature 300-400oC to form the corresponding substituted products. ...
... Some of the important reactions of alkanes are following. 1) Halogenations: This involves replacement of hydrogen atom by halogen atom. a) Chlorination: Alkanes react with chlorine in presence U.V light or diffused sunlight or at temperature 300-400oC to form the corresponding substituted products. ...
Grignard Reagents brochure
... A.C.Frisch, N. Shaikh, A.Zapf, M.Beller, Angew.Chem. 2002, 114, 4218 K.Tamao, S.Komada, I.Nakajima, M.Kumada, A.Minato, K.Suzuki, Tetrahedron 38 ...
... A.C.Frisch, N. Shaikh, A.Zapf, M.Beller, Angew.Chem. 2002, 114, 4218 K.Tamao, S.Komada, I.Nakajima, M.Kumada, A.Minato, K.Suzuki, Tetrahedron 38 ...
Melt Modification of Poly(styrene-co-maleic anhydride)
... 3.8 and 4.2 ppm, and the olefinic hydrogens give signals at 5.0 and 5.8 ppm. Additionally, the long methylene chain has an intense signal at 1.2 ppm. C8OH gives a methylene signal at 3.6 ppm and a CH3 signal at 0.9 ppm. The copolymer SMA17 shows only two very broad signals at 1.1–2.4 (aliphatic hydr ...
... 3.8 and 4.2 ppm, and the olefinic hydrogens give signals at 5.0 and 5.8 ppm. Additionally, the long methylene chain has an intense signal at 1.2 ppm. C8OH gives a methylene signal at 3.6 ppm and a CH3 signal at 0.9 ppm. The copolymer SMA17 shows only two very broad signals at 1.1–2.4 (aliphatic hydr ...
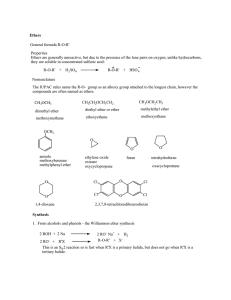
Ethers General formula R-O-R` Properties Ethers are generally
... The IUPAC rules name the R-O- group as an alkoxy group attached to the longest chain, however the compounds are often named as ethers. CH3OCH3 ...
... The IUPAC rules name the R-O- group as an alkoxy group attached to the longest chain, however the compounds are often named as ethers. CH3OCH3 ...
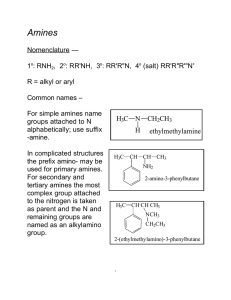
amine
... The five carbon atoms and the sp2-hybridized nitrogen atom of pyridine contribute one p electron to the aromatic sextet • The lone-pair electrons of nitrogen atom occupy an sp2 orbital in the plane of the ring • Pyridine is less basic that alkylamines because the lone-pair electrons are in an sp2 or ...
... The five carbon atoms and the sp2-hybridized nitrogen atom of pyridine contribute one p electron to the aromatic sextet • The lone-pair electrons of nitrogen atom occupy an sp2 orbital in the plane of the ring • Pyridine is less basic that alkylamines because the lone-pair electrons are in an sp2 or ...
Aromatic heterocycles 1: structures and reactions
... Benzene is aromatic because it has six electrons in a cyclic conjugated system. We know it is aromatic because it is exceptionally stable and it has a ring current and hence large chemical shifts in the proton NMR spectrum as well as a special chemistry involving substitution rather than addition wi ...
... Benzene is aromatic because it has six electrons in a cyclic conjugated system. We know it is aromatic because it is exceptionally stable and it has a ring current and hence large chemical shifts in the proton NMR spectrum as well as a special chemistry involving substitution rather than addition wi ...
4.9 Preparation of Alkyl Halides from Alcohols and Hydrogen Halides
... by positively charged carbon because the s orbital can overlap with the empty 2p orbital of positively charged carbon ...
... by positively charged carbon because the s orbital can overlap with the empty 2p orbital of positively charged carbon ...
unit 12 aldehydes, ketones and carboxylic acids
... Q.25 Melting point of an acid with even no. of carbon atoms is higher than those of its neighbour with odd no. of carbon atoms. Ans They fit into crystal lattice more readily than odd ones that is why they have higher lattice energy and higher melting point. Q.26 Why do aldehydes have lower boiling ...
... Q.25 Melting point of an acid with even no. of carbon atoms is higher than those of its neighbour with odd no. of carbon atoms. Ans They fit into crystal lattice more readily than odd ones that is why they have higher lattice energy and higher melting point. Q.26 Why do aldehydes have lower boiling ...
226 amines lec
... Vinyl and aryl halides usually do not react. A severe limitation to ammonolysis in many cases is generation of other classes of amines — ...
... Vinyl and aryl halides usually do not react. A severe limitation to ammonolysis in many cases is generation of other classes of amines — ...
Full-Text PDF
... Concerning methylbenzene derivatives (entries 11–14), when formylation took place in the absence of oxygen atoms, coordination with the metal atom was not possible and therefore the less hindered isomer was favored. In this case, it is believed that the reaction mechanism occurs through the formatio ...
... Concerning methylbenzene derivatives (entries 11–14), when formylation took place in the absence of oxygen atoms, coordination with the metal atom was not possible and therefore the less hindered isomer was favored. In this case, it is believed that the reaction mechanism occurs through the formatio ...
Amines
... However, instead of measuring an amine’s basicity using the above equilibrium, chemists usually refer to the acidity of the conjugate acid of the amine. The weaker the conjugate acid, the stronger the base strength of the amine. ...
... However, instead of measuring an amine’s basicity using the above equilibrium, chemists usually refer to the acidity of the conjugate acid of the amine. The weaker the conjugate acid, the stronger the base strength of the amine. ...
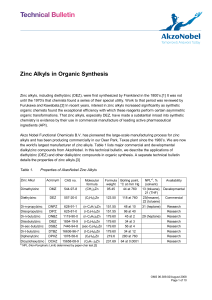
Zinc Alkyls in Organic Synthesis
... organic transformations. That zinc alkyls, especially DEZ, have made a substantial inroad into synthetic chemistry is evidence by their use in commercial manufacture of leading active pharmaceutical ingredients (API). Akzo Nobel Functional Chemicals B.V. has pioneered the large-scale manufacturing p ...
... organic transformations. That zinc alkyls, especially DEZ, have made a substantial inroad into synthetic chemistry is evidence by their use in commercial manufacture of leading active pharmaceutical ingredients (API). Akzo Nobel Functional Chemicals B.V. has pioneered the large-scale manufacturing p ...
Grignard-syn-12-ques
... Alkyl halides, vinyl halides, and aryl halides can all be used to form organolithium and organomagnesium compounds However, these organometallic compounds cannot be prepared from compounds containing acidic groups (OH, NH2, NHR, SH, C=CH, CO2H) Cannot use H2O, CH3OH, CH3CH2OH, etc. as solvents Cann ...
... Alkyl halides, vinyl halides, and aryl halides can all be used to form organolithium and organomagnesium compounds However, these organometallic compounds cannot be prepared from compounds containing acidic groups (OH, NH2, NHR, SH, C=CH, CO2H) Cannot use H2O, CH3OH, CH3CH2OH, etc. as solvents Cann ...
Organic synthesis and methodology related to the malaria drug artemisinin
... Figure 64: Initial Substrate Screening with MeCN ..................................... 121 Figure 65: Initial Substrate Screening with CH2Cl2 .................................... 122 Figure 66: Use of Electron-Rich Ethoxyacetylene ..................................... 123 Figure 67: Qualitative Scre ...
... Figure 64: Initial Substrate Screening with MeCN ..................................... 121 Figure 65: Initial Substrate Screening with CH2Cl2 .................................... 122 Figure 66: Use of Electron-Rich Ethoxyacetylene ..................................... 123 Figure 67: Qualitative Scre ...
- Opus: Online Publications Store
... However, the transformation of a C–H bond in a less useful compound to form a more useful compound having C–B, C–C, C–Si, C–N, C–O, C–F or C–S bonds is quite challenging. This is because of the inherently inert nature (bond energies of C(sp3)-H and C(sp2)–H bonds range from 90-110 kcal/mol) and low ...
... However, the transformation of a C–H bond in a less useful compound to form a more useful compound having C–B, C–C, C–Si, C–N, C–O, C–F or C–S bonds is quite challenging. This is because of the inherently inert nature (bond energies of C(sp3)-H and C(sp2)–H bonds range from 90-110 kcal/mol) and low ...
BSA - Sigma
... BSA (N,O-bis(trimethylsilyl)acetamide) is one of the most commonly used silylating reagents. Its reactivity is similar to that of BSTFA, readily silylating a wide range of acidic functional groups such as non-sterically hindered alcohols, amides, amines, amino acids, carboxylic acids, and enols. It ...
... BSA (N,O-bis(trimethylsilyl)acetamide) is one of the most commonly used silylating reagents. Its reactivity is similar to that of BSTFA, readily silylating a wide range of acidic functional groups such as non-sterically hindered alcohols, amides, amines, amino acids, carboxylic acids, and enols. It ...
Alcohols, Phenols , Phenols and Ethers Alcohols
... and applications. Alcohols and phenols are formed when a hydrogen atom in a hydrocarbon, aliphatic and aromatic respectively, is replaced by –OH group. These classes of compounds find wide applications in industry as well as in day-to-day life. For instance, have you ever noticed that ordinary spiri ...
... and applications. Alcohols and phenols are formed when a hydrogen atom in a hydrocarbon, aliphatic and aromatic respectively, is replaced by –OH group. These classes of compounds find wide applications in industry as well as in day-to-day life. For instance, have you ever noticed that ordinary spiri ...
Development of New Synthetic Routes to Organoboronates by Catalytic Allylic Substitution and
... the classical methods for the synthesis of allylboronate derivatives are often associated with problems, such as low regio- and stereoselectivity as well as poor functional group tolerance, milder methods are required for obtaining functionalized derivatives. The use of transition metal catalysis ha ...
... the classical methods for the synthesis of allylboronate derivatives are often associated with problems, such as low regio- and stereoselectivity as well as poor functional group tolerance, milder methods are required for obtaining functionalized derivatives. The use of transition metal catalysis ha ...
12_chemistry_impq_CH10_haloalkanes_and_haloarenes_02
... . Ans. The C-O bond in phenol acquires partial double bond character due to resonance and hence be cleared by X- ions to form halobenzenes. But in alcohols a pure C — O bond is maintained and can be cleared by X– ions. Q 3. Explain why o-nitrophenol is more acidic than o-methoxy phenol? Ans . Due to ...
... . Ans. The C-O bond in phenol acquires partial double bond character due to resonance and hence be cleared by X- ions to form halobenzenes. But in alcohols a pure C — O bond is maintained and can be cleared by X– ions. Q 3. Explain why o-nitrophenol is more acidic than o-methoxy phenol? Ans . Due to ...
Hofmann–Löffler reaction

The Hofmann–Löffler reaction (also referred to as Hofmann–Löffler–Freytag reaction, Löffler–Freytag reaction, Löffler–Hofmann reaction, as well as Löffler's method) is an organic reaction in which a cyclic amine 2 (pyrrolidine or, in some cases, piperidine) is generated by thermal or photochemical decomposition of N-halogenated amine 1 in the presence of a strong acid (concentrated sulfuric acid or concentrated CF3CO2H). The Hofmann–Löffler–Freytag reaction proceeds via an intramolecular hydrogen atom transfer to a nitrogen-centered radical and is an example of a remote intramolecular free radical C–H functionalization.