Alcohols
... Alcohol and phenols have nearly the same geometry around the oxygen atom as water • The C-O-H bond angle is approximately tetrahedral (108.5º in methanol) • Thiols have a more compressed C-S-H bond angle (96.5 º in methanethiol) • The oxygen atom is sp3-hybridized Alcohols and phenols, like water, h ...
... Alcohol and phenols have nearly the same geometry around the oxygen atom as water • The C-O-H bond angle is approximately tetrahedral (108.5º in methanol) • Thiols have a more compressed C-S-H bond angle (96.5 º in methanethiol) • The oxygen atom is sp3-hybridized Alcohols and phenols, like water, h ...
Preparation and Physical Properties of Chitosan Benzoic Acid
... methods using acid anhydride [6,7], mixed anhydride [8,9], acyl chlorides [10,11], benzoyl chloride and methanesulfonic acid [12], and p-toluenesulfonyl chloride, lithium chloride, and carboxylic acid [13] have been reported. Among them, the mixed anhydride method using trifluoroacetic anhydride, ph ...
... methods using acid anhydride [6,7], mixed anhydride [8,9], acyl chlorides [10,11], benzoyl chloride and methanesulfonic acid [12], and p-toluenesulfonyl chloride, lithium chloride, and carboxylic acid [13] have been reported. Among them, the mixed anhydride method using trifluoroacetic anhydride, ph ...
Alcohols
... Alcohol and phenols have nearly the same geometry around the oxygen atom as water • The C-O-H bond angle is approximately tetrahedral (108.5º in methanol) • Thiols have a more compressed C-S-H bond angle (96.5 º in methanethiol) • The oxygen atom is sp3-hybridized Alcohols and phenols, like water, h ...
... Alcohol and phenols have nearly the same geometry around the oxygen atom as water • The C-O-H bond angle is approximately tetrahedral (108.5º in methanol) • Thiols have a more compressed C-S-H bond angle (96.5 º in methanethiol) • The oxygen atom is sp3-hybridized Alcohols and phenols, like water, h ...
Anionic rearrangement of 2-benzyloxypyridine derivatives and a synthetic approach to aldingenin B
... [1,2]-Anionic rearrangements are important tools for altering the complexity of molecules at hand. In Part I of this dissertation, an anionic rearrangement of 2-benzyloxypyridine is described. Pyridine-directed metallation of the benzylic carbon leads to 1,2-migration of pyridine via a postulated as ...
... [1,2]-Anionic rearrangements are important tools for altering the complexity of molecules at hand. In Part I of this dissertation, an anionic rearrangement of 2-benzyloxypyridine is described. Pyridine-directed metallation of the benzylic carbon leads to 1,2-migration of pyridine via a postulated as ...
Stereoselective Construction of a β
... did not proceed in Et2O14 (entry 5). The rearrangement of (Z)-8 proceeded with relatively high diastereoselectivity (84-92%de) compared with that of (E)-8 (entries 8-10). Assignment of the relative stereochemistry of both anti- and syn-9 was based on 2D-NOE experiments15 and comparison of the 1H NMR ...
... did not proceed in Et2O14 (entry 5). The rearrangement of (Z)-8 proceeded with relatively high diastereoselectivity (84-92%de) compared with that of (E)-8 (entries 8-10). Assignment of the relative stereochemistry of both anti- and syn-9 was based on 2D-NOE experiments15 and comparison of the 1H NMR ...
PowerPoint 演示文稿
... Cl H + CH3 The radical Cl + H CH3 reacts with Cl CH3 + Cl Cl Cl + CH3 the mole. of product Cl + H CH2Cl ...
... Cl H + CH3 The radical Cl + H CH3 reacts with Cl CH3 + Cl Cl Cl + CH3 the mole. of product Cl + H CH2Cl ...
PDF - Nanyang Technological University
... a cooperative catalytic system to develop the enantioselective reaction between 1 a and 2 a. The reaction catalyzed by (S)proline/CuBr2 in DMF gave the desired product in 63 % yield and in a 1:1 diastereomeric ratio (d.r.), with virtually no enantioselectivity (4 %/3 % ee) (Table 1, entry 4). The re ...
... a cooperative catalytic system to develop the enantioselective reaction between 1 a and 2 a. The reaction catalyzed by (S)proline/CuBr2 in DMF gave the desired product in 63 % yield and in a 1:1 diastereomeric ratio (d.r.), with virtually no enantioselectivity (4 %/3 % ee) (Table 1, entry 4). The re ...
Substitution Reactions of Specifically Ortho
... Typical Metalation Procedure. In a flame-dried flask equipped with a serum cap was placed 2 mmol of imine in 15 ml of THF and the mixture was cooled to -78 "C with a Coyacetone bath. To the stirred solution was added a solution of 2.1 mmol of n-BuLi-hexane (AlfaVentron) over 1min via syringe through ...
... Typical Metalation Procedure. In a flame-dried flask equipped with a serum cap was placed 2 mmol of imine in 15 ml of THF and the mixture was cooled to -78 "C with a Coyacetone bath. To the stirred solution was added a solution of 2.1 mmol of n-BuLi-hexane (AlfaVentron) over 1min via syringe through ...
Amide bond formation and peptide coupling
... 2.1.1.3. Limitations of acyl chlorides. Nevertheless, acyl chlorides have limited value in peptide coupling because of the danger of hydrolysis, racemisation, cleavage of protecting groups and other side reactions (e.g., N-carboxy anhydride formation, see Section 2.4.2.3). The tendency of acyl chlor ...
... 2.1.1.3. Limitations of acyl chlorides. Nevertheless, acyl chlorides have limited value in peptide coupling because of the danger of hydrolysis, racemisation, cleavage of protecting groups and other side reactions (e.g., N-carboxy anhydride formation, see Section 2.4.2.3). The tendency of acyl chlor ...
Chemical Reactivity and Biological Activity of Diketene
... reaction, which results in an amide bond in the NBP-diketene adduct. The lability of the amide bond as opposed to the amine bonds formed by β-propiolactone and β-butyrolactone could be one of the differential factors responsible for the lack of carcinogenicity of diketene. (v) Ab initio calculations ...
... reaction, which results in an amide bond in the NBP-diketene adduct. The lability of the amide bond as opposed to the amine bonds formed by β-propiolactone and β-butyrolactone could be one of the differential factors responsible for the lack of carcinogenicity of diketene. (v) Ab initio calculations ...
Topic 17 notes - A
... If a carboxylic acid and an alcohol are heated under reflux with concentrated H2SO4, a condensation reaction takes place and an ester is formed: Carboxylic acid + alcohol == ester + H2O R1-COOH + R2OH == R1-COOR2 + H2O This reaction is known as esterification. Esterifcation is an example of condensa ...
... If a carboxylic acid and an alcohol are heated under reflux with concentrated H2SO4, a condensation reaction takes place and an ester is formed: Carboxylic acid + alcohol == ester + H2O R1-COOH + R2OH == R1-COOR2 + H2O This reaction is known as esterification. Esterifcation is an example of condensa ...
Isomeric Product Detection in the
... The distribution of products formed from heterogeneous oxidation depends strongly on molecular structure. Ruehl et al.4 analyzed the products formed by the reaction of OH with squalane (a liquid branched C30 alkane) and octacosane (a solid normal C28 alkane) aerosols and observed that the first gener ...
... The distribution of products formed from heterogeneous oxidation depends strongly on molecular structure. Ruehl et al.4 analyzed the products formed by the reaction of OH with squalane (a liquid branched C30 alkane) and octacosane (a solid normal C28 alkane) aerosols and observed that the first gener ...
Palladium and Ruthenium Catalyzed Reactions By Bryan Jaksic
... activity of commonly used precatalysts with the newly synthesized precatalyst, Pd(η5-C5H5)(η3-1Ph-C3H4), for Sonogashira cross-coupling reactions. Sonogashira reactions are important as they provide a simple method for the formation of substituted alkynes, a commonly found functionality within impor ...
... activity of commonly used precatalysts with the newly synthesized precatalyst, Pd(η5-C5H5)(η3-1Ph-C3H4), for Sonogashira cross-coupling reactions. Sonogashira reactions are important as they provide a simple method for the formation of substituted alkynes, a commonly found functionality within impor ...
File
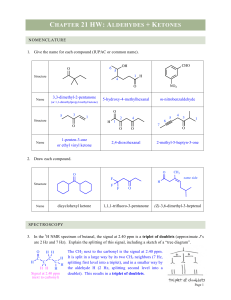
... energy. The aldehyde has one alkyl group (EDG) attached to the C=O, but the ketone has 2 EDG. Therefore, the ketone stabilizes the δ+ more and starts at a lower energy than the aldehyde. This causes the hydrate reaction to be uphill for the ketone (so higher hydrate % for aldehyde). Although it’s on ...
... energy. The aldehyde has one alkyl group (EDG) attached to the C=O, but the ketone has 2 EDG. Therefore, the ketone stabilizes the δ+ more and starts at a lower energy than the aldehyde. This causes the hydrate reaction to be uphill for the ketone (so higher hydrate % for aldehyde). Although it’s on ...
New insights into the mechanism of sorbitol transformation
... hydrocarbons over bifunctional solid catalysts, with or without hydrogen supply [8]. Hydrocarbons separate from the aqueous reaction medium and are more easily integrated into the existing fuels than ethanol [9,10]. The selective transformation of sorbitol into hexane is however a challenge since mu ...
... hydrocarbons over bifunctional solid catalysts, with or without hydrogen supply [8]. Hydrocarbons separate from the aqueous reaction medium and are more easily integrated into the existing fuels than ethanol [9,10]. The selective transformation of sorbitol into hexane is however a challenge since mu ...
Chapter 15
... We can generally not isolate the aldehyde intermediate in aqueous solution. If we look at the mechanism you will see why. The first step is the formation of the chromate ester by attack of the alcohol hydroxyl onto the chromium with subsequent loss of water. This is an acid catalyzed esterification ...
... We can generally not isolate the aldehyde intermediate in aqueous solution. If we look at the mechanism you will see why. The first step is the formation of the chromate ester by attack of the alcohol hydroxyl onto the chromium with subsequent loss of water. This is an acid catalyzed esterification ...
Organic Chemistry - UCR Chemistry
... We will learn below that, because of the different structures of their alkyl groups, nucleophilic substitution on bromomethane (CH3-Br) occurs only by SN2 mechanisms, while t-butyl bromide (2-bromo-2-methylpropane) ((CH3)3C-Br) undergoes nucleophilic substitution only by SN1 mechanisms. H2O versus - ...
... We will learn below that, because of the different structures of their alkyl groups, nucleophilic substitution on bromomethane (CH3-Br) occurs only by SN2 mechanisms, while t-butyl bromide (2-bromo-2-methylpropane) ((CH3)3C-Br) undergoes nucleophilic substitution only by SN1 mechanisms. H2O versus - ...
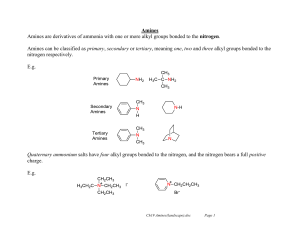
Amines Amines are derivatives of ammonia with one or more alkyl
... EAS is further inhibited by pyridine because of the tendency of the nitrogen atom to react directly with the electrophile, generating a pyridinium ion (which is still aromatic). This positively charged pyridinium ion is even more deactivated to EAS than pyridine itself. ...
... EAS is further inhibited by pyridine because of the tendency of the nitrogen atom to react directly with the electrophile, generating a pyridinium ion (which is still aromatic). This positively charged pyridinium ion is even more deactivated to EAS than pyridine itself. ...
Photoremovable Protecting Groups
... studies, sodium 2-naphthalenesulfonate, all well-known triplet quenchers; this established the short-lived triplet (3 to 14 ns) as the reactive excited state for benzoin. Further information was revealed from Stern–Volmer quenching analyses which provided the rate of release of phosphate from the be ...
... studies, sodium 2-naphthalenesulfonate, all well-known triplet quenchers; this established the short-lived triplet (3 to 14 ns) as the reactive excited state for benzoin. Further information was revealed from Stern–Volmer quenching analyses which provided the rate of release of phosphate from the be ...
Title Several Reactions of Isocyanide and Related Compounds
... LIST OF PUBLISHED PAPERS . . . . • • . . . . . . . . . . . . . . . • . . ...
... LIST OF PUBLISHED PAPERS . . . . • • . . . . . . . . . . . . . . . • . . ...
REASONING QUESTIONS IN ORGANIC CHEMISTRY
... to electron releasing effect strength of hydrogen decreases. 51. Arrange the following compounds in increasing order of their boiling points? CH3 -CH2-OH, CH3-CHO, CH3 -O-CH3, CH3 -CH2-CH3, Ans: molecular of these compounds are similar; ethanol is having inter molecular H- bond hence more boiling po ...
... to electron releasing effect strength of hydrogen decreases. 51. Arrange the following compounds in increasing order of their boiling points? CH3 -CH2-OH, CH3-CHO, CH3 -O-CH3, CH3 -CH2-CH3, Ans: molecular of these compounds are similar; ethanol is having inter molecular H- bond hence more boiling po ...
9: Formation of Alkenes and Alkynes. Elimination Reactions
... E1 and E2 with 3 Haloalkanes. The base often determines whether 2methylpropene arises via an E1 or an E2 mechanism with the 3° haloalkane t-butyl bromide (2-bromo-2-methylpropane). In contrast, we learned in Chapter 7 that this 3° haloalkane substrate undergoes substitution only by an SN1 mechanism. ...
... E1 and E2 with 3 Haloalkanes. The base often determines whether 2methylpropene arises via an E1 or an E2 mechanism with the 3° haloalkane t-butyl bromide (2-bromo-2-methylpropane). In contrast, we learned in Chapter 7 that this 3° haloalkane substrate undergoes substitution only by an SN1 mechanism. ...
REASONING QUESTIONS IN ORGANIC CHEMISTRY TEXT
... to electron releasing effect strength of hydrogen decreases. 51. Arrange the following compounds in increasing order of their boiling points? CH3 -CH2-OH, CH3-CHO, CH3 -O-CH3, CH3 -CH2-CH3, Ans: molecular of these compounds are similar; ethanol is having inter molecular H- bond hence more boiling po ...
... to electron releasing effect strength of hydrogen decreases. 51. Arrange the following compounds in increasing order of their boiling points? CH3 -CH2-OH, CH3-CHO, CH3 -O-CH3, CH3 -CH2-CH3, Ans: molecular of these compounds are similar; ethanol is having inter molecular H- bond hence more boiling po ...
Organometallic Methods for Forming and Cleaving Carbon
... First and foremost, I would like to show my gratitude to Prof. Robert Madsen for accepting me in his group and for his valuable guidance and expertise during my time as a Ph.D. student. I deeply appreciate Dr. Phil. Torkil Holm for sharing his experience and for fruitful discussions. I would very mu ...
... First and foremost, I would like to show my gratitude to Prof. Robert Madsen for accepting me in his group and for his valuable guidance and expertise during my time as a Ph.D. student. I deeply appreciate Dr. Phil. Torkil Holm for sharing his experience and for fruitful discussions. I would very mu ...
Heterogeneous Catalysts for Biodiesel Production
... Several methods have been proposed to solve these problems,11 but the most useful seem to be the following:5,10,12–15 (a) use of enzymes. A Lypase enzyme catalyzes both transesterification of triglycerides and esterification of FFA R-COOH + MeOH a R-COOMe + H2O in one step.9,11–13 (b) use of acid ca ...
... Several methods have been proposed to solve these problems,11 but the most useful seem to be the following:5,10,12–15 (a) use of enzymes. A Lypase enzyme catalyzes both transesterification of triglycerides and esterification of FFA R-COOH + MeOH a R-COOMe + H2O in one step.9,11–13 (b) use of acid ca ...
Hofmann–Löffler reaction

The Hofmann–Löffler reaction (also referred to as Hofmann–Löffler–Freytag reaction, Löffler–Freytag reaction, Löffler–Hofmann reaction, as well as Löffler's method) is an organic reaction in which a cyclic amine 2 (pyrrolidine or, in some cases, piperidine) is generated by thermal or photochemical decomposition of N-halogenated amine 1 in the presence of a strong acid (concentrated sulfuric acid or concentrated CF3CO2H). The Hofmann–Löffler–Freytag reaction proceeds via an intramolecular hydrogen atom transfer to a nitrogen-centered radical and is an example of a remote intramolecular free radical C–H functionalization.