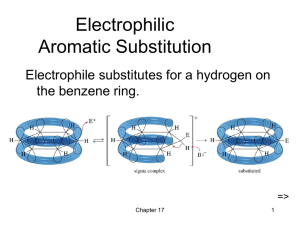
Electrophilic Aromatic Substit
... Activating, O-, PDirecting Substituents • Alkyl groups stabilize the sigma complex by induction, donating electron density through the sigma bond. • Substituents with a lone pair of electrons stabilize the sigma complex by resonance. ...
... Activating, O-, PDirecting Substituents • Alkyl groups stabilize the sigma complex by induction, donating electron density through the sigma bond. • Substituents with a lone pair of electrons stabilize the sigma complex by resonance. ...
23.2 Alcohols, Ethers, and Amines
... Halogenation Reactions The addition of bromine to carbon-carbon multiple bonds is often used as a chemical test for unsaturation in an organic molecule. • Bromine has an orange color, but most bromine-substituted organic compounds are colorless. After mixing, if the solution remains colorless, the c ...
... Halogenation Reactions The addition of bromine to carbon-carbon multiple bonds is often used as a chemical test for unsaturation in an organic molecule. • Bromine has an orange color, but most bromine-substituted organic compounds are colorless. After mixing, if the solution remains colorless, the c ...
CHAPTER 21 ELECTROCHEMISTRY: CHEMICAL CHANGE AND
... a) The metal A is being oxidized to form the metal cation. To form positive ions, an atom must always lose electrons, so this half-reaction is always an oxidation. b) The metal ion B is gaining electrons to form the metal B, so it is displaced. c) The anode is the electrode at which oxidation takes ...
... a) The metal A is being oxidized to form the metal cation. To form positive ions, an atom must always lose electrons, so this half-reaction is always an oxidation. b) The metal ion B is gaining electrons to form the metal B, so it is displaced. c) The anode is the electrode at which oxidation takes ...
Kinetic investigation of low-pH Fe(II) oxidation and development of a
... mainly from the group Acidithiobacillus Ferrooxidans. However, this method suffers from two major drawbacks: (1) the dependence of the process on the sensitive autotrophic biomass and (2) the relatively rapid precipitation of Fe(III) solids, mostly of the jarosite group. The current work investigate ...
... mainly from the group Acidithiobacillus Ferrooxidans. However, this method suffers from two major drawbacks: (1) the dependence of the process on the sensitive autotrophic biomass and (2) the relatively rapid precipitation of Fe(III) solids, mostly of the jarosite group. The current work investigate ...
CH 3 - bYTEBoss
... • Aldehydes contain a CHO attached to the end of a carbon chain The C has a double bonded O and a single bonded H attached to it. Since it is at the end of a chain there is no need to number. • The ending changes to “al” ...
... • Aldehydes contain a CHO attached to the end of a carbon chain The C has a double bonded O and a single bonded H attached to it. Since it is at the end of a chain there is no need to number. • The ending changes to “al” ...
Derivatization reactions for the determination of amines by gas
... sensitive to each amine, the sensitivity of this detection method is generally lower than that of ECD and NPD (Table 1). ...
... sensitive to each amine, the sensitivity of this detection method is generally lower than that of ECD and NPD (Table 1). ...
Stoichiometry - Social Circle City Schools
... this is the reverse of the process that is used to determine percent composition. The first step in the process is to understand that the percent composition of any element is equal to the mass of that element in exactly 100 g of compound. For example, a compound containing only phosphorus and chlor ...
... this is the reverse of the process that is used to determine percent composition. The first step in the process is to understand that the percent composition of any element is equal to the mass of that element in exactly 100 g of compound. For example, a compound containing only phosphorus and chlor ...
PART 6-ICHO-26-30
... What is the pH transition range of the indicator which could be used for the back titration. ...
... What is the pH transition range of the indicator which could be used for the back titration. ...
Chapter 14 Aldehydes, Ketones, and Chiral Molecules
... Carbonyl Group in Aldehydes and Ketones A carbonyl group • In an aldehyde is attached to at least one H atom. • In a ketone is attached to two carbon groups. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings ...
... Carbonyl Group in Aldehydes and Ketones A carbonyl group • In an aldehyde is attached to at least one H atom. • In a ketone is attached to two carbon groups. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings ...
"Cyano Compounds, Inorganic," in: Ullmann`s Encyclopedia of
... for this process [69]. The subsequent reaction steps, ammonia absorption and hydrogen cyanide isolation, are similar to those of the Andrussow process. A distinct advantage is the higher HCN content (Table 1) of the product gas, so that the number of steps and the size and cost of recovery equipment ...
... for this process [69]. The subsequent reaction steps, ammonia absorption and hydrogen cyanide isolation, are similar to those of the Andrussow process. A distinct advantage is the higher HCN content (Table 1) of the product gas, so that the number of steps and the size and cost of recovery equipment ...
OC 2/e 9 Alcohols
... ethanol are about as accessible as hydroxide ion for solvation; these alcohol are about as acidic as water. • as the bulk of the alkyl group increases, the ability of water to solvate the alkoxide decreases, the acidity of the alcohol decreases, and the basicity of the alkoxide ...
... ethanol are about as accessible as hydroxide ion for solvation; these alcohol are about as acidic as water. • as the bulk of the alkyl group increases, the ability of water to solvate the alkoxide decreases, the acidity of the alcohol decreases, and the basicity of the alkoxide ...
One-pot aqueous synthesis of cysteine-capped
... Research Online is the open access institutional repository for the University of Wollongong. For further information contact the UOW Library: [email protected] ...
... Research Online is the open access institutional repository for the University of Wollongong. For further information contact the UOW Library: [email protected] ...
5 Steps
... This book conducts you through the five steps necessary to prepare yourself for success on the exam. These steps will provide you with the skills and strategies vital to the exam, and the practice that will lead you to towards the perfect 5. First, we start by introducing the basic five-step plan us ...
... This book conducts you through the five steps necessary to prepare yourself for success on the exam. These steps will provide you with the skills and strategies vital to the exam, and the practice that will lead you to towards the perfect 5. First, we start by introducing the basic five-step plan us ...
Study Guide for Content Mastery - Student Edition
... is worrisome because without ozone all organisms on Earth are subject to harm from too much radiation. In your textbook, read about chlorofluorocarbons. ...
... is worrisome because without ozone all organisms on Earth are subject to harm from too much radiation. In your textbook, read about chlorofluorocarbons. ...
Document
... 24) Which of the following compounds is a weak acid? A) ethanol B) ethanal C) phenol D) cyclohexanol E) acetone Answer: C Objective: 13.3 Global Outcomes: GO2 ...
... 24) Which of the following compounds is a weak acid? A) ethanol B) ethanal C) phenol D) cyclohexanol E) acetone Answer: C Objective: 13.3 Global Outcomes: GO2 ...
Part 2-ICHO-26-30
... What is the pH transition range of the indicator which could be used for the back titration. ...
... What is the pH transition range of the indicator which could be used for the back titration. ...
CHAPTER 12 | The Chemistry of Solids
... The number of lithium cations in the unit cell is 8. The formula for the compound is Li2S. Think about It This salt is neutral because the charge on two lithium ions (1+) in the formula balances the charge on one sulfide anion (2–). 12.15. Collect and Organize We are to identify the elements highlig ...
... The number of lithium cations in the unit cell is 8. The formula for the compound is Li2S. Think about It This salt is neutral because the charge on two lithium ions (1+) in the formula balances the charge on one sulfide anion (2–). 12.15. Collect and Organize We are to identify the elements highlig ...
Document
... 43) An enantiomer is A) a stereoisomer that is not a mirror image of another molecule. B) a stereoisomer that is a mirror image of another molecule. C) a diastereoisomer. D) a structural isomer. E) a cis-trans isomer. Answer: B Objective: 14.5 Global Outcomes: GO2 ...
... 43) An enantiomer is A) a stereoisomer that is not a mirror image of another molecule. B) a stereoisomer that is a mirror image of another molecule. C) a diastereoisomer. D) a structural isomer. E) a cis-trans isomer. Answer: B Objective: 14.5 Global Outcomes: GO2 ...
1 Solutions 4a (Chapter 4 problems) Chem151 [Kua]
... as a base so this could be an acid-base reaction); (f) NH3 is a weak base, which reacts with strong acid: NH3(aq) + H3O+(aq) → H2O(l) + NH4+(aq) (acid-base reaction). 4.102 The statement of the problem indicates what reactions take place: H2S + O2 → SO2 + H2O. H and S are balanced; give O a coeffici ...
... as a base so this could be an acid-base reaction); (f) NH3 is a weak base, which reacts with strong acid: NH3(aq) + H3O+(aq) → H2O(l) + NH4+(aq) (acid-base reaction). 4.102 The statement of the problem indicates what reactions take place: H2S + O2 → SO2 + H2O. H and S are balanced; give O a coeffici ...
LIPIDS
... Lipids are made of 3 fatty acids and glycerol. Glycerol is an alcohol containing 3 hydroxyl groups (OH) . ...
... Lipids are made of 3 fatty acids and glycerol. Glycerol is an alcohol containing 3 hydroxyl groups (OH) . ...
Chapter 3 Stoichiometry
... single water molecule consists of two hydrogen atoms and one oxygen atom. An empirical formula is the simplest whole‐number ratio of elements in a compound. The empirical formula of water is the same as its molecular formula, H2O. Hydrogen peroxide, however, has the molecular formula H2O2 and ...
... single water molecule consists of two hydrogen atoms and one oxygen atom. An empirical formula is the simplest whole‐number ratio of elements in a compound. The empirical formula of water is the same as its molecular formula, H2O. Hydrogen peroxide, however, has the molecular formula H2O2 and ...
ORGANIC CHEMISTRY
... a. Add 1-pentene to water (Markovnikov Addition) and 2-pentanol will be prepared in a 100% yield ...
... a. Add 1-pentene to water (Markovnikov Addition) and 2-pentanol will be prepared in a 100% yield ...
5 Steps to a 5 AP Chemistry, 2008-2009 Edition
... This book conducts you through the five steps necessary to prepare yourself for success on the exam. These steps will provide you with the skills and strategies vital to the exam and the practice that will lead you to towards the perfect 5. First, we start by introducing the basic five-step plan use ...
... This book conducts you through the five steps necessary to prepare yourself for success on the exam. These steps will provide you with the skills and strategies vital to the exam and the practice that will lead you to towards the perfect 5. First, we start by introducing the basic five-step plan use ...
Organic Chemistry with a Biological Emphasis Volume I
... Introduction: Louis Pasteur and the discovery of molecular chirality Section 1: Conformations of open-chain organic molecules Section 2: Conformations of cyclic organic molecules Section 3: Chirality and stereoisomers Section 4: Labeling chiral centers Section 5: Optical activity Section 6: Compound ...
... Introduction: Louis Pasteur and the discovery of molecular chirality Section 1: Conformations of open-chain organic molecules Section 2: Conformations of cyclic organic molecules Section 3: Chirality and stereoisomers Section 4: Labeling chiral centers Section 5: Optical activity Section 6: Compound ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.


















![1 Solutions 4a (Chapter 4 problems) Chem151 [Kua]](http://s1.studyres.com/store/data/002731518_1-574ec10e88e667508364281b6325aeef-300x300.png)




