Unit 5: Oragnic Chemistry Notes (answers)
... - nomenclature of alkane involves the use of the suffix ~yne (like in Alk ~yne). - hydrocarbons with two triple bonds are named with the suffix ~diyne (~di yne as in two triple bonds). - hydrocarbons with three triple bonds are named with the suffix ~triyne (~tri yne as in three triple bonds). - unl ...
... - nomenclature of alkane involves the use of the suffix ~yne (like in Alk ~yne). - hydrocarbons with two triple bonds are named with the suffix ~diyne (~di yne as in two triple bonds). - hydrocarbons with three triple bonds are named with the suffix ~triyne (~tri yne as in three triple bonds). - unl ...
4. Solution Guide to Supplementary Exercises
... 50 C (1) Using new and used zinc-carbon cells at the same time in an electrical appliance is NOT dangerous, but this gives poorer results. (2) Charging a lithium ion secondary cell with a high current may overheat the pack and lead to explosion. 51 C In a magnesium-copper chemical cell, electrons fl ...
... 50 C (1) Using new and used zinc-carbon cells at the same time in an electrical appliance is NOT dangerous, but this gives poorer results. (2) Charging a lithium ion secondary cell with a high current may overheat the pack and lead to explosion. 51 C In a magnesium-copper chemical cell, electrons fl ...
Section – B - About iTutoring
... The ions of III B, IV, IV A groups are not precipitated in presence of NH4Cl and NH4OH. (81). The ions of which group are precipitated in presence of HCl and H2S in qualitative analysis? The ions of II groups are precipitated in presence of HCl and H2S. (82). What is meant by ionic product Ip? The p ...
... The ions of III B, IV, IV A groups are not precipitated in presence of NH4Cl and NH4OH. (81). The ions of which group are precipitated in presence of HCl and H2S in qualitative analysis? The ions of II groups are precipitated in presence of HCl and H2S. (82). What is meant by ionic product Ip? The p ...
Limiting Reactants and Percentage Yield
... One reactant limits the product of a reaction. Once one of the reactants is used up, no more product can be formed. The substance that is completely used up first in a reaction is called the limiting reactant. The limiting reactant is the reactant that limits the amount of the other reactant that ca ...
... One reactant limits the product of a reaction. Once one of the reactants is used up, no more product can be formed. The substance that is completely used up first in a reaction is called the limiting reactant. The limiting reactant is the reactant that limits the amount of the other reactant that ca ...
Go FIGure
... salt sealed off from the water—MgSO41s2 for hot packs and NH4NO31s2 for cold packs. When the pack is squeezed, the seal separating the solid from the water is broken and a solution forms, either increasing or decreasing the temperature. The enthalpy change for a process can provide insight into the ...
... salt sealed off from the water—MgSO41s2 for hot packs and NH4NO31s2 for cold packs. When the pack is squeezed, the seal separating the solid from the water is broken and a solution forms, either increasing or decreasing the temperature. The enthalpy change for a process can provide insight into the ...
Chapter 3 Alcohols, Phenols, and Ethers
... • The word “alcohol” comes from the Arabic term al kohl meaning “the fine powder.” Originally, this referred to an antimony sulfide (Sb2S3)compound used for eye shadow, which was ground up to form a fine powder, but then later came to refer to any finely divided powder. In the Middle Ages, this term ...
... • The word “alcohol” comes from the Arabic term al kohl meaning “the fine powder.” Originally, this referred to an antimony sulfide (Sb2S3)compound used for eye shadow, which was ground up to form a fine powder, but then later came to refer to any finely divided powder. In the Middle Ages, this term ...
aminobromination.alk..
... 2.5. Stereochemistry of BBC (3) addition to 1phenylpropenes Diastereomeric (E)- and (Z)-1-phenylpropenes7 were selected as model compounds for studying the stereochemical course of BBC 3 addition. Both crude BBC adducts to (E)- and (Z)-1-phenylpropene 11a and 11b have ...
... 2.5. Stereochemistry of BBC (3) addition to 1phenylpropenes Diastereomeric (E)- and (Z)-1-phenylpropenes7 were selected as model compounds for studying the stereochemical course of BBC 3 addition. Both crude BBC adducts to (E)- and (Z)-1-phenylpropene 11a and 11b have ...
Cliffs Notes
... Trademarks: Cliffs, CliffsNotes, CliffsAP, CliffsComplete, CliffsTestPrep, CliffsQuickReview, CliffsNote-a-Day, and all related logos and trade dress are registered trademarks or trademarks of Hungry Minds, Inc., in the United States and other countries. AP, APP, and Advanced Placement Program are r ...
... Trademarks: Cliffs, CliffsNotes, CliffsAP, CliffsComplete, CliffsTestPrep, CliffsQuickReview, CliffsNote-a-Day, and all related logos and trade dress are registered trademarks or trademarks of Hungry Minds, Inc., in the United States and other countries. AP, APP, and Advanced Placement Program are r ...
Chem Soc Rev
... well as the homolytic theory for OMC. Therefore, the mechanism for activation of methane may change from one catalytic system to another. Certain notions should be elaborated before making a conclusion. Kinetic study is a useful tool to probe the catalytic mechanism. Using a temporal product analysi ...
... well as the homolytic theory for OMC. Therefore, the mechanism for activation of methane may change from one catalytic system to another. Certain notions should be elaborated before making a conclusion. Kinetic study is a useful tool to probe the catalytic mechanism. Using a temporal product analysi ...
Chapter 14-15 - Bakersfield College
... • Enzymes in the liver oxidize ethanol. • Blood alcohol over 0.4% can be fatal. ...
... • Enzymes in the liver oxidize ethanol. • Blood alcohol over 0.4% can be fatal. ...
File
... The larger the value for Keq the closer to completion the reaction is at equilibrium. (NOTE: "Completion" means reactants have been completely converted to products.) A very small value for Keq means that there is very little product and lots of reactant at equilibrium. In other words, a very ______ ...
... The larger the value for Keq the closer to completion the reaction is at equilibrium. (NOTE: "Completion" means reactants have been completely converted to products.) A very small value for Keq means that there is very little product and lots of reactant at equilibrium. In other words, a very ______ ...
NOMENCLATURE OF ORGANIC COMPOUNDS - A
... rotation. An equimolar mixture of two optical isomers will thus have no effect on plane polarised light and is thus not optically active. Such mixtures are said to be racemic mixtures or racemates. A racemic mixture is an equimolar mixture of two optical isomers. Racemic mixtures are not optically a ...
... rotation. An equimolar mixture of two optical isomers will thus have no effect on plane polarised light and is thus not optically active. Such mixtures are said to be racemic mixtures or racemates. A racemic mixture is an equimolar mixture of two optical isomers. Racemic mixtures are not optically a ...
Chapter 4 "Reactions in Aqueous Solution"
... project's attribution page (http://2012books.lardbucket.org/attribution.html?utm_source=header). For more information on the source of this book, or why it is available for free, please see the project's home page (http://2012books.lardbucket.org/). You can browse or download additional books there. ...
... project's attribution page (http://2012books.lardbucket.org/attribution.html?utm_source=header). For more information on the source of this book, or why it is available for free, please see the project's home page (http://2012books.lardbucket.org/). You can browse or download additional books there. ...
Reactions in Aqueous Solution
... project's attribution page (http://2012books.lardbucket.org/attribution.html?utm_source=header). For more information on the source of this book, or why it is available for free, please see the project's home page (http://2012books.lardbucket.org/). You can browse or download additional books there. ...
... project's attribution page (http://2012books.lardbucket.org/attribution.html?utm_source=header). For more information on the source of this book, or why it is available for free, please see the project's home page (http://2012books.lardbucket.org/). You can browse or download additional books there. ...
Drawing Organic Structures Functional Groups Constitutional Isomers
... which are less basic than sp3-hybridized N’s • More s-character holds electrons more tightly • Electrons are less available/less basic ...
... which are less basic than sp3-hybridized N’s • More s-character holds electrons more tightly • Electrons are less available/less basic ...
Organic Chemistry - hrsbstaff.ednet.ns.ca
... nineteenth century. It is called acetylsalicylic acid (ASA). You probably know it better by its brand name, Aspirin™. Chemists refer to salicin, ASA, and more than ten million other chemicals like them as organic compounds. As described in the unit opener, an organic compound is a molecular compound ...
... nineteenth century. It is called acetylsalicylic acid (ASA). You probably know it better by its brand name, Aspirin™. Chemists refer to salicin, ASA, and more than ten million other chemicals like them as organic compounds. As described in the unit opener, an organic compound is a molecular compound ...
Chemistry.of Organic Compounds
... of reason, " it also is true that theories which correlate facts are an aid to memory. Moreover it is desirable that students majoring in chemistry and aBied sciences become acquainted with the current ideas and language of chemistry. Therefore in this text considerable emphasis is placed on the exp ...
... of reason, " it also is true that theories which correlate facts are an aid to memory. Moreover it is desirable that students majoring in chemistry and aBied sciences become acquainted with the current ideas and language of chemistry. Therefore in this text considerable emphasis is placed on the exp ...
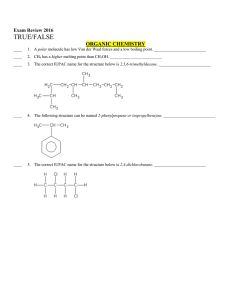
Multiple Choice Exam Review June 2016
... ____ 111. An exothermic reaction is one where a. heat is transferred from the surroundings into a system b. heat is transferred from a system into the surroundings c. kinetic energy is transformed into potential energy d. there is no transfer of heat e. none of the above ____ 112. An endothermic rea ...
... ____ 111. An exothermic reaction is one where a. heat is transferred from the surroundings into a system b. heat is transferred from a system into the surroundings c. kinetic energy is transformed into potential energy d. there is no transfer of heat e. none of the above ____ 112. An endothermic rea ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.