Organic – Nomenclature – III
... while the side chain containing OH group(a functional group having lower priority) has been excluded from the principal chain used for naming the parent. In the second example, COOEt(Et=ethyl)group has been excluded from the principal chain as the longest chain is C-6. Conclusion: Maximum number of ...
... while the side chain containing OH group(a functional group having lower priority) has been excluded from the principal chain used for naming the parent. In the second example, COOEt(Et=ethyl)group has been excluded from the principal chain as the longest chain is C-6. Conclusion: Maximum number of ...
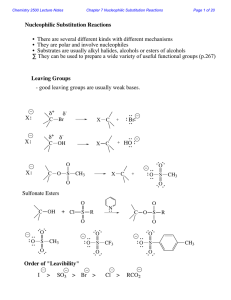
Nucleophilic Substitution Reactions
... The Hammond Postulate - widely used to rationalize differences in reaction rate "The structure of a transition state resembles that species to which it is closest in energy." Early T.S. ...
... The Hammond Postulate - widely used to rationalize differences in reaction rate "The structure of a transition state resembles that species to which it is closest in energy." Early T.S. ...
Organic Chemistry Package 2011
... This works for simple alkanes such as butane (C4H10) and pentane (C5H12). However, it becomes hopeless when larger alkanes are considered. For example there are 5 isomers of hexane (C6H14), 9 isomers of heptane (C7H16) and 75 isomers of decane (C10H22). Another problem arises as far as nomenclature ...
... This works for simple alkanes such as butane (C4H10) and pentane (C5H12). However, it becomes hopeless when larger alkanes are considered. For example there are 5 isomers of hexane (C6H14), 9 isomers of heptane (C7H16) and 75 isomers of decane (C10H22). Another problem arises as far as nomenclature ...
The Organometallic Chemistry Of Transition Metals
... longest and are often called classical or Werner complexes such as [Co(NH3 )6 ]3+ . The simplest metal–ligand bond is perhaps Ln M−NH3 , where an ammonia binds to a metal fragment. This fragment will usually also have other ligands, represented here by Ln . The bond consists of the lone pair of elec ...
... longest and are often called classical or Werner complexes such as [Co(NH3 )6 ]3+ . The simplest metal–ligand bond is perhaps Ln M−NH3 , where an ammonia binds to a metal fragment. This fragment will usually also have other ligands, represented here by Ln . The bond consists of the lone pair of elec ...
Workshop materials for Class XII
... Everything starts with a dream or concept in your mind…. The dream is given form by putting it on paper……… The construction process begins……………… The workshop has been a powerful tool and technique modeler to streamline our dream towards achieving invincible results in chemistry. The questions banks ...
... Everything starts with a dream or concept in your mind…. The dream is given form by putting it on paper……… The construction process begins……………… The workshop has been a powerful tool and technique modeler to streamline our dream towards achieving invincible results in chemistry. The questions banks ...
Chapter 16 Aldehydes and Ketones
... An aldehyde cannot have the molecular formula C5H12O. C5H12 has too many H’s. Since an aldehyde has a double bond, the number of C’s and H’s resembles an alkene, not an alkane. An aldehyde with 5 C’s would have the molecular formula C5H10O. ...
... An aldehyde cannot have the molecular formula C5H12O. C5H12 has too many H’s. Since an aldehyde has a double bond, the number of C’s and H’s resembles an alkene, not an alkane. An aldehyde with 5 C’s would have the molecular formula C5H10O. ...
Study Guide Chapter 10: An Introduction to Chemistry
... 1 × 308.0245 Mg UO 2 F2 or ? Mg UO 2 F2 = 8.0 Mg H 2 O = 68 Mg UO2F2 2 × 18.0153 Mg H 2 O b. Why do you think the reactant in excess was chosen to be in excess? Water is much less toxic and less expensive than the radioactive and rare uranium compound. Water in the form of either liquid ...
... 1 × 308.0245 Mg UO 2 F2 or ? Mg UO 2 F2 = 8.0 Mg H 2 O = 68 Mg UO2F2 2 × 18.0153 Mg H 2 O b. Why do you think the reactant in excess was chosen to be in excess? Water is much less toxic and less expensive than the radioactive and rare uranium compound. Water in the form of either liquid ...
Chapter 10 Chemical Calculations and Chemical Equations
... 1 × 308.0245 Mg UO 2 F2 or ? Mg UO 2 F2 = 8.0 Mg H 2 O = 68 Mg UO2F2 2 × 18.0153 Mg H 2 O b. Why do you think the reactant in excess was chosen to be in excess? Water is much less toxic and less expensive than the radioactive and rare uranium compound. Water in the form of either liquid ...
... 1 × 308.0245 Mg UO 2 F2 or ? Mg UO 2 F2 = 8.0 Mg H 2 O = 68 Mg UO2F2 2 × 18.0153 Mg H 2 O b. Why do you think the reactant in excess was chosen to be in excess? Water is much less toxic and less expensive than the radioactive and rare uranium compound. Water in the form of either liquid ...
Full-Text PDF
... relevant ligand features in catalysts for alkene epoxidation are corroles (3, structurally similar to porphyrins) [12] and bis-amino-phenolate ligands (4, structurally related to salen) [13], which are also represented in Figure 1. The prominence of porphyrin and salen scaffolds in the design of Mn ...
... relevant ligand features in catalysts for alkene epoxidation are corroles (3, structurally similar to porphyrins) [12] and bis-amino-phenolate ligands (4, structurally related to salen) [13], which are also represented in Figure 1. The prominence of porphyrin and salen scaffolds in the design of Mn ...
irm_ch17
... 17.44 Add HCl to form the water-soluble amine salt. 17.45 The pharmaceutical industry uses an older naming system for amine salts; the amine salts are treated as amine-acid complexes. The salt ethylmethylammonium chloride would be written ethylmethylamine hydrochloride, and its structure would be wr ...
... 17.44 Add HCl to form the water-soluble amine salt. 17.45 The pharmaceutical industry uses an older naming system for amine salts; the amine salts are treated as amine-acid complexes. The salt ethylmethylammonium chloride would be written ethylmethylamine hydrochloride, and its structure would be wr ...
irm_ch15
... them can be used as the ending of the name. The functional groups in this problem, ranked in order of decreasing priority, are: aldehyde, ketone, alcohol, and alkoxy. a. The compound has two functional groups, a ketone and an alcohol; the ketone has priority, so this compound is named as a ketone. b ...
... them can be used as the ending of the name. The functional groups in this problem, ranked in order of decreasing priority, are: aldehyde, ketone, alcohol, and alkoxy. a. The compound has two functional groups, a ketone and an alcohol; the ketone has priority, so this compound is named as a ketone. b ...
Low Temperature Precursors for SnOx Thin Films
... encouragement and useful discussions about the work presented within this thesis. I was fortunate to also have the wisdom and guidance of two other academic supervisors with Professor Kieran Molloy providing his wealth of knowledge and experience within the field of tin and materials chemistry and D ...
... encouragement and useful discussions about the work presented within this thesis. I was fortunate to also have the wisdom and guidance of two other academic supervisors with Professor Kieran Molloy providing his wealth of knowledge and experience within the field of tin and materials chemistry and D ...
Chemical Redox Agents for Organometallic
... In this context, an “innocent” reagent is one which engages exclusively in an outer-sphere ET reaction. Whereas electrodes are among the most benign reagents in synthetic applications, chemical redox agents are more likely to engage in inner-sphere electron-transfer processes and to display coordina ...
... In this context, an “innocent” reagent is one which engages exclusively in an outer-sphere ET reaction. Whereas electrodes are among the most benign reagents in synthetic applications, chemical redox agents are more likely to engage in inner-sphere electron-transfer processes and to display coordina ...
Chemical Redox Agents for Organometallic
... In this context, an “innocent” reagent is one which engages exclusively in an outer-sphere ET reaction. Whereas electrodes are among the most benign reagents in synthetic applications, chemical redox agents are more likely to engage in inner-sphere electron-transfer processes and to display coordina ...
... In this context, an “innocent” reagent is one which engages exclusively in an outer-sphere ET reaction. Whereas electrodes are among the most benign reagents in synthetic applications, chemical redox agents are more likely to engage in inner-sphere electron-transfer processes and to display coordina ...
master ap chemistry - NelnetSolutions.com
... For more information, contact Peterson’s, 2000 Lenox Drive, Lawrenceville, NJ 08648; 800-338-3282; or find us on the World Wide Web at: www.petersons.com/about. © 2007 Peterson’s, a Nelnet company Previous edition © 2005 Editor: Wallie Walker Hammond; Production Editor: Mark D. Snider; Composition M ...
... For more information, contact Peterson’s, 2000 Lenox Drive, Lawrenceville, NJ 08648; 800-338-3282; or find us on the World Wide Web at: www.petersons.com/about. © 2007 Peterson’s, a Nelnet company Previous edition © 2005 Editor: Wallie Walker Hammond; Production Editor: Mark D. Snider; Composition M ...
Unit X Organic Chem (SmartBoard)
... structures that differ from each other by one structural unit, in this case —CH2—, are called a HOMOLOGOUS SERIES. Because the chain of carbon atoms extends in a straight-line they are called “STRAIGHT-CHAIN” or “UNBRANCHED” hydrocarbons. The names of the all the above hydrocarbons end in “ANE” beca ...
... structures that differ from each other by one structural unit, in this case —CH2—, are called a HOMOLOGOUS SERIES. Because the chain of carbon atoms extends in a straight-line they are called “STRAIGHT-CHAIN” or “UNBRANCHED” hydrocarbons. The names of the all the above hydrocarbons end in “ANE” beca ...
CH 3 - bYTEBoss
... Amines are classified as primary, secondary, or tertiary. In a primary (1°) amine, one carbon group is bonded to the nitrogen atom. A secondary (2°) amine has two carbon groups. A tertiary (3°) amine has three carbon groups. H CH3 CH3 ...
... Amines are classified as primary, secondary, or tertiary. In a primary (1°) amine, one carbon group is bonded to the nitrogen atom. A secondary (2°) amine has two carbon groups. A tertiary (3°) amine has three carbon groups. H CH3 CH3 ...
Elimination Reactions
... such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 M or above. WWU -- Chemistry ...
... such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 M or above. WWU -- Chemistry ...
UNIT 1. SOME BASIC CONCEPTS OF CHEMISTRY Concept
... Q3- What is a chemical equation? What are its essential features? (L-2) Ans. the qualitative and quantitative representation of a chemical reaction in short form in terms of symbols and formulae is called chemical equation. For example, on heating calcium carbonate, it gives Caco3 →Ca0 + CO2 Essenti ...
... Q3- What is a chemical equation? What are its essential features? (L-2) Ans. the qualitative and quantitative representation of a chemical reaction in short form in terms of symbols and formulae is called chemical equation. For example, on heating calcium carbonate, it gives Caco3 →Ca0 + CO2 Essenti ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.