Slides
... amines to form enamines l Cyclic amines are often used l The reaction is catalyzed by acid l Removal of water drives enamine formation to completion ...
... amines to form enamines l Cyclic amines are often used l The reaction is catalyzed by acid l Removal of water drives enamine formation to completion ...
Chemistry Final Test
... (o) Many transition metal complexes are very intensively colorful. What are the two mechanisms responsible for it? 5、(15%) [CoCl2Br2F2]4− forms an octahedral complex. (a) Draw all possible stereoisomers of this complex. (9%) (b) Identify all possible pairs of enantiomers. (3%) (c) Identify all achir ...
... (o) Many transition metal complexes are very intensively colorful. What are the two mechanisms responsible for it? 5、(15%) [CoCl2Br2F2]4− forms an octahedral complex. (a) Draw all possible stereoisomers of this complex. (9%) (b) Identify all possible pairs of enantiomers. (3%) (c) Identify all achir ...
IR Spectroscopy of Esters - Purdue College of Science
... vegetable oil to make a fatty acid methyl ester (through the Indiana Soybean Alliance’s Soybean Science Kithttp://www.soybeansciencekit.com/ which is much more fancy now than what I have or you can just purchase your own biodiesel for comparison sake) . We obtained a sample of soy biodiesel (bottom ...
... vegetable oil to make a fatty acid methyl ester (through the Indiana Soybean Alliance’s Soybean Science Kithttp://www.soybeansciencekit.com/ which is much more fancy now than what I have or you can just purchase your own biodiesel for comparison sake) . We obtained a sample of soy biodiesel (bottom ...
• Pergamon
... A variety of substituted 2-methylpyrroles underwent allylic oxidation with the perchlorinated metalloporphyrin 2 and iodosylbenzene in 'IFAlCH2CI2 (9:1). Subsequent addition of an a-free pyrrole to the same reaction mixture afforded an efficient one-pot route to dipyrromethanes. ...
... A variety of substituted 2-methylpyrroles underwent allylic oxidation with the perchlorinated metalloporphyrin 2 and iodosylbenzene in 'IFAlCH2CI2 (9:1). Subsequent addition of an a-free pyrrole to the same reaction mixture afforded an efficient one-pot route to dipyrromethanes. ...
Alcohol Worksheet Key
... position 4. The negative charge is not delocalized onto postions 3 or 5, so electron withdrawing groups at position 3 and 5 will have very little effect on the acidity of the phenol. ...
... position 4. The negative charge is not delocalized onto postions 3 or 5, so electron withdrawing groups at position 3 and 5 will have very little effect on the acidity of the phenol. ...
Unit 5 Study Guide
... c. Neither b. Pb d. Both 15. Which of the following displacement reactions will NOT occur? a) CuSO4(aq) + Zn(s) ---> Cu(s) + ZnSO4(aq) b) 2NaNO3(aq) + Zn(s) ---> 2Na(s) + Zn(NO3)2(aq) c) 2AgNO3(aq) + Zn(s) ---> 2Ag(s) + Zn(NO3)2(aq) d) 2HCl(aq) + Zn(s) ---> H2(g) + ZnCl2(aq) 16. An iron rod is place ...
... c. Neither b. Pb d. Both 15. Which of the following displacement reactions will NOT occur? a) CuSO4(aq) + Zn(s) ---> Cu(s) + ZnSO4(aq) b) 2NaNO3(aq) + Zn(s) ---> 2Na(s) + Zn(NO3)2(aq) c) 2AgNO3(aq) + Zn(s) ---> 2Ag(s) + Zn(NO3)2(aq) d) 2HCl(aq) + Zn(s) ---> H2(g) + ZnCl2(aq) 16. An iron rod is place ...
Chemistry 199 - Department of Chemistry | Oregon State University
... What is chiral? Sketch a molecule that contains three chiral carbons. A molecule is chiral if it cannot be superimposed on its mirror image. A carbon in a molecule is chiral if it has four different groups attached to it. CH2BrF is not chiral. CHBrClF is. ...
... What is chiral? Sketch a molecule that contains three chiral carbons. A molecule is chiral if it cannot be superimposed on its mirror image. A carbon in a molecule is chiral if it has four different groups attached to it. CH2BrF is not chiral. CHBrClF is. ...
REACTIONS OF ALCOHOLS
... resulting alkyl halides • Tertiary Alcohol→ turns cloudy immediately (the alkyl halide is not soluble in water and precipitates out) • Secondary Alcohol → turns cloudy after 5 minutes • Primary Alcohol → takes much longer than 5 minutes to turn cloudy ...
... resulting alkyl halides • Tertiary Alcohol→ turns cloudy immediately (the alkyl halide is not soluble in water and precipitates out) • Secondary Alcohol → turns cloudy after 5 minutes • Primary Alcohol → takes much longer than 5 minutes to turn cloudy ...
Name
... Directions: In the table below, look at each chemical formula. Then in the box next to it, explain how many atoms are in that compound. Name each atom and write how many there are next to it. See the example, which is done for you. Substance (Common Name) ...
... Directions: In the table below, look at each chemical formula. Then in the box next to it, explain how many atoms are in that compound. Name each atom and write how many there are next to it. See the example, which is done for you. Substance (Common Name) ...
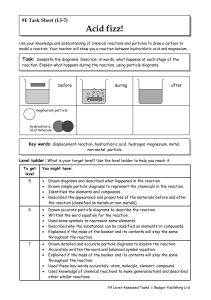
Year 9 Homework Task 9E-5 Reactions 5-7
... Drawn accurate particle diagrams to describe the reaction. Written the word equation for the reaction. Used some symbols to represent some elements. Described why the substances can be classified as elements or compounds. Explained if the mass of the beaker and its contents will stay the same throug ...
... Drawn accurate particle diagrams to describe the reaction. Written the word equation for the reaction. Used some symbols to represent some elements. Described why the substances can be classified as elements or compounds. Explained if the mass of the beaker and its contents will stay the same throug ...
Practice Questions Survey II – 1152 1. The bond angles around the
... a. the presence of one or more carbon-carbon double bonds b. the presence of one or more carbon-carbon triple bonds c. the presence of at least one carbon-carbon double bond, and at least one carbon-carbon triple bond d. the presence of a ring system 19. Given an alkane, an alkene, and an alkyne, ea ...
... a. the presence of one or more carbon-carbon double bonds b. the presence of one or more carbon-carbon triple bonds c. the presence of at least one carbon-carbon double bond, and at least one carbon-carbon triple bond d. the presence of a ring system 19. Given an alkane, an alkene, and an alkyne, ea ...
Microsoft Word - Open Access Repository of Indian Theses
... Stereoselective synthesis of (+)-(2R, 3R, 4R)-deacetylanisomycin Analogues of furanoses in which the ring oxygen is replaced by nitrogen and the anomeric hydroxyl is removed have been reported to be almost always inhibitors of the corresponding glycosidases. Glycosidases are involved in several impo ...
... Stereoselective synthesis of (+)-(2R, 3R, 4R)-deacetylanisomycin Analogues of furanoses in which the ring oxygen is replaced by nitrogen and the anomeric hydroxyl is removed have been reported to be almost always inhibitors of the corresponding glycosidases. Glycosidases are involved in several impo ...
Types of Chemical Reactions
... general form of: A + BC → AC + B 5) Double Replacement: This is when the metals and nonmetals of two different molecules switch places, forming two entirely different compounds. These reactions are in the general form: ...
... general form of: A + BC → AC + B 5) Double Replacement: This is when the metals and nonmetals of two different molecules switch places, forming two entirely different compounds. These reactions are in the general form: ...
Here is the Original File - University of New Hampshire
... 1. W.R.G. Baeyens, S.G. Schulman, Y. Zhao; Chemiluminescence-Based Detection: Principles and Analytical Applications in Flowing Streams and in Immunoassays. Journal of Pharmaceutical and Biomedical Analysis. 1998, 17(6-7), (941-953) 2. Jaworek. Christine, Lacobucci. Sarah; Wittig Reaction: The Synth ...
... 1. W.R.G. Baeyens, S.G. Schulman, Y. Zhao; Chemiluminescence-Based Detection: Principles and Analytical Applications in Flowing Streams and in Immunoassays. Journal of Pharmaceutical and Biomedical Analysis. 1998, 17(6-7), (941-953) 2. Jaworek. Christine, Lacobucci. Sarah; Wittig Reaction: The Synth ...
Exam 3 Review
... Oxidation to carbonyl compounds: PCC or CrO3 (know when to use each reagent) Conversion to R-X: dry HBr/HCl, PBr3, SOCl2 (know when to use each reagent) Elimination to alkenes - POCl3 (for all alcohols) or H2SO4 (for 3° alcohols) Protecting –OH groups: Me3Si-Cl/Et3N (TMS chloride/weak base) puts on ...
... Oxidation to carbonyl compounds: PCC or CrO3 (know when to use each reagent) Conversion to R-X: dry HBr/HCl, PBr3, SOCl2 (know when to use each reagent) Elimination to alkenes - POCl3 (for all alcohols) or H2SO4 (for 3° alcohols) Protecting –OH groups: Me3Si-Cl/Et3N (TMS chloride/weak base) puts on ...
Final Exam Review Sheet Chemistry 110a/1998
... cation, and anion using a resonance and molecular orbital argument. How does the allylic radical compare in stability to 3°, 2°, and 1°? How about the allylic cation, in this regard? The pKa of an allylic hydrogen is 41: how can you use this value to say that the allylic anion is more stable than th ...
... cation, and anion using a resonance and molecular orbital argument. How does the allylic radical compare in stability to 3°, 2°, and 1°? How about the allylic cation, in this regard? The pKa of an allylic hydrogen is 41: how can you use this value to say that the allylic anion is more stable than th ...
Answer Key - OISE-IS-Chemistry-2011-2012
... 19. Write the reaction showing how the ester below can be prepared from a carboxylic acid and an alcohol. Provide the names of all reactants and any other products, below your equation. [6 marks] ...
... 19. Write the reaction showing how the ester below can be prepared from a carboxylic acid and an alcohol. Provide the names of all reactants and any other products, below your equation. [6 marks] ...
C h e m g u i d e ... CARBOXYLIC ACIDS: PREPARATION
... c) Write a simple equation (using [O] for the oxidising agent) for the oxidation of propan-1-ol to propanoic acid. d) The electron-half-equation for the dichromate(VI) ion acting as an oxidising agent in the presence of an acid is 2Cr3+ + 7H2O Cr2O72- + 14H+ + 6e(i) Work out the electron-half-equati ...
... c) Write a simple equation (using [O] for the oxidising agent) for the oxidation of propan-1-ol to propanoic acid. d) The electron-half-equation for the dichromate(VI) ion acting as an oxidising agent in the presence of an acid is 2Cr3+ + 7H2O Cr2O72- + 14H+ + 6e(i) Work out the electron-half-equati ...
last year`s April exam
... a) OHb) OH c) H2O d) H3O+ 20) When some salt is dissolved into water, it is found that the pH of the solution is neutral. Which of the following salts might have been dissolved to create this solution? a) NaF b) NH4Cl c) NaBr d) KC2H3O2 21) Which of the following functional groups cannot form part o ...
... a) OHb) OH c) H2O d) H3O+ 20) When some salt is dissolved into water, it is found that the pH of the solution is neutral. Which of the following salts might have been dissolved to create this solution? a) NaF b) NH4Cl c) NaBr d) KC2H3O2 21) Which of the following functional groups cannot form part o ...
Glossary of Key Terms in Chapter Two
... Markovnikov’s rule (11.5) the rule that states that a hydrogen atom, adding to a carbon-carbon double bond, is more likely to add to the carbon having the larger number of hydrogens attached to it already. monomer (11.5) the individual molecules from which a polymer is formed. phenyl group (11.6) a ...
... Markovnikov’s rule (11.5) the rule that states that a hydrogen atom, adding to a carbon-carbon double bond, is more likely to add to the carbon having the larger number of hydrogens attached to it already. monomer (11.5) the individual molecules from which a polymer is formed. phenyl group (11.6) a ...
org test 1
... 2. Why is Sulphuric acid not used during reaction of alcohol with KI? 3. Why is preparation of ethers by acid catalysed dehydration of 2° and 3° alcohols not a suitable method? 4. Of benzene and phenol, which is more easily nitrated and why? 5. Ethers possess a net dipole moment even if they are sym ...
... 2. Why is Sulphuric acid not used during reaction of alcohol with KI? 3. Why is preparation of ethers by acid catalysed dehydration of 2° and 3° alcohols not a suitable method? 4. Of benzene and phenol, which is more easily nitrated and why? 5. Ethers possess a net dipole moment even if they are sym ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.