Investigating Esters
... not be possible to determine the boiling point by the distillation method and a semi micro method should be employed. The suggested method in the starter experiment is a generic one and may need to be slightly adjusted for each ester. Students should be encouraged to find details from the literature ...
... not be possible to determine the boiling point by the distillation method and a semi micro method should be employed. The suggested method in the starter experiment is a generic one and may need to be slightly adjusted for each ester. Students should be encouraged to find details from the literature ...
T. V. RajanBabu Chemistry, 730 Autumn 1997
... Chelated and unchelated transition state models to make all four aldol stereoisomers using diffrent metals Asymmetric catalysis in aldol synthesis : Mukaiyama, Carreira, Evans Ito’s gold-catalyzed aldol reactions (a Knoevenagel-type reaction, see later) of -isocyanoacetates Aldol reactions of ester ...
... Chelated and unchelated transition state models to make all four aldol stereoisomers using diffrent metals Asymmetric catalysis in aldol synthesis : Mukaiyama, Carreira, Evans Ito’s gold-catalyzed aldol reactions (a Knoevenagel-type reaction, see later) of -isocyanoacetates Aldol reactions of ester ...
biochemistry - apbiostafford
... Draw a lipid molecule (triglyceride). Point out the carboxyl, or acid, group and the hydrocarbon chains. Explain the difference between a saturated fatty acid and an unsaturated fatty acid. For butter and corn oil, indicate whether each is considered a saturated or an unsaturated fat. Using a diagra ...
... Draw a lipid molecule (triglyceride). Point out the carboxyl, or acid, group and the hydrocarbon chains. Explain the difference between a saturated fatty acid and an unsaturated fatty acid. For butter and corn oil, indicate whether each is considered a saturated or an unsaturated fat. Using a diagra ...
Synthesis of Isobutyl Propionate via Esterification
... Assemble a microscale chromatography column (see Lab Guide for review), being sure it is clamped in a vertical position. Close the valve, and fill the column with dichloromethane to the bottom of the funnel. Prepare a slurry of 1 g of silica gel in 4 mL of dichloromethane in a small beaker. Stir the ...
... Assemble a microscale chromatography column (see Lab Guide for review), being sure it is clamped in a vertical position. Close the valve, and fill the column with dichloromethane to the bottom of the funnel. Prepare a slurry of 1 g of silica gel in 4 mL of dichloromethane in a small beaker. Stir the ...
Chapter 4. Functional Group Transformations: Oxidation and
... 4.3 Chemoselective agents for oxidizing alcohols MnO2 is a highly chemoselective oxidant-allylic, benzylic, and propargyl alcohols are oxidized faster than saturated alcohols. Solvent: H2O, acetone, or CHCl3. Low reactivity: use large amount of oxidant ...
... 4.3 Chemoselective agents for oxidizing alcohols MnO2 is a highly chemoselective oxidant-allylic, benzylic, and propargyl alcohols are oxidized faster than saturated alcohols. Solvent: H2O, acetone, or CHCl3. Low reactivity: use large amount of oxidant ...
Drug Design
... Diaminechloroplatnium (II) is an inorganic complex that as been used to treat certain types of ovarian and testicular cancers. Diaminechloroplatnium (II) exists in both cis and trans isomers. ...
... Diaminechloroplatnium (II) is an inorganic complex that as been used to treat certain types of ovarian and testicular cancers. Diaminechloroplatnium (II) exists in both cis and trans isomers. ...
Chemistry - Choithram School
... m-isomers. Allyl chloride is hydrolysed more readily than n-propyl chloride. Why? ...
... m-isomers. Allyl chloride is hydrolysed more readily than n-propyl chloride. Why? ...
JSUNIL TUTORIAL, SAMASTIPUR,BIHAR Carbon and its compounds Answer the following questions:
... Class – X Subject - Science ...
... Class – X Subject - Science ...
Extra Unit 3 Problems for the Web Site (Honors
... 9. When 4.90 g of KClO3 was heated, it showed a mass loss of 0.384 g. Find the percent of the original KClO3 that had decomposed. 10. When 10.0 g of silicon dust, Si, is exploded with 100.0 g of oxygen, O2, forming silicon dioxide, SiO2, how many grams of O2 remain uncombined? The reaction equation ...
... 9. When 4.90 g of KClO3 was heated, it showed a mass loss of 0.384 g. Find the percent of the original KClO3 that had decomposed. 10. When 10.0 g of silicon dust, Si, is exploded with 100.0 g of oxygen, O2, forming silicon dioxide, SiO2, how many grams of O2 remain uncombined? The reaction equation ...
Slide 1
... Diaminechloroplatnium (II) is an inorganic complex that as been used to treat certain types of ovarian and testicular cancers. Diaminechloroplatnium (II) exists in both cis and trans isomers. ...
... Diaminechloroplatnium (II) is an inorganic complex that as been used to treat certain types of ovarian and testicular cancers. Diaminechloroplatnium (II) exists in both cis and trans isomers. ...
Alcohols
... Give a reagent that can be used in a test to distinguish between a ketone and an aldehyde. State what you would observe in the test. Reagent ............................................................................................................. Observation with ketone ......................... ...
... Give a reagent that can be used in a test to distinguish between a ketone and an aldehyde. State what you would observe in the test. Reagent ............................................................................................................. Observation with ketone ......................... ...
twelve important naval substances – bonding
... as fuels or solvents. Most organic molecules consist of a structural backbone of C-C single bonds and one or more functional groups. Functional groups are portions of an organic molecule where carbon has bonds to atoms other than carbon or hydrogen. ...
... as fuels or solvents. Most organic molecules consist of a structural backbone of C-C single bonds and one or more functional groups. Functional groups are portions of an organic molecule where carbon has bonds to atoms other than carbon or hydrogen. ...
gr11chemreview
... The following questions highlight the main knowledge and skills from grade 11 chemistry. A good understanding of the concepts covered in grade 11 chemistry is essential for success in grade 12 chemistry and you may need to do some independent review of some material if you do not have a clear unders ...
... The following questions highlight the main knowledge and skills from grade 11 chemistry. A good understanding of the concepts covered in grade 11 chemistry is essential for success in grade 12 chemistry and you may need to do some independent review of some material if you do not have a clear unders ...
Name__________________________Review Organic Reactions
... 14. Which organic compounds are often used to create fragrances for the perfume industry? A) ethers C) alkanes ...
... 14. Which organic compounds are often used to create fragrances for the perfume industry? A) ethers C) alkanes ...
Esterification
... 1/ Weigh 1.0g of Phenol into a small conical flask. HAZARD 2/Add 18cm3 of 1M NaOH(aq) and bung the flask. 3/ HAZARD In a fume cupboard add 2cm3 of Benzoyl chloride in small quantities at a time. 4/ Fit a bung and shake vigorously with occasional cooling under the tap or in ice water. Releasing the g ...
... 1/ Weigh 1.0g of Phenol into a small conical flask. HAZARD 2/Add 18cm3 of 1M NaOH(aq) and bung the flask. 3/ HAZARD In a fume cupboard add 2cm3 of Benzoyl chloride in small quantities at a time. 4/ Fit a bung and shake vigorously with occasional cooling under the tap or in ice water. Releasing the g ...
Document
... Some active metals such as sodium and calcium will react with water to give a metallic hydroxide and hydrogen gas. Ex: Ca + 2H2O Ca(OH)2 + H2 ...
... Some active metals such as sodium and calcium will react with water to give a metallic hydroxide and hydrogen gas. Ex: Ca + 2H2O Ca(OH)2 + H2 ...
Answer Key to Assignment #7
... 10. Suggest syntheses of the following alcohols, all of which can be derived from methylcyclopentene. The number of arrows indicate the number of reactions that are needed. ...
... 10. Suggest syntheses of the following alcohols, all of which can be derived from methylcyclopentene. The number of arrows indicate the number of reactions that are needed. ...
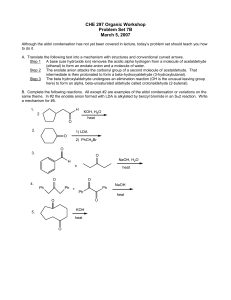
CHE 297 Organic Workshop
... A. Translate the following text into a mechanism with structures and conventional curved arrows. Step 1 A base (use hydroxide ion) removes the acidic alpha hydrogen from a molecule of acetaldehyde (ethanal) to form an enolate anion and a molecule of water. Step 2 The enolate anion attacks the carbon ...
... A. Translate the following text into a mechanism with structures and conventional curved arrows. Step 1 A base (use hydroxide ion) removes the acidic alpha hydrogen from a molecule of acetaldehyde (ethanal) to form an enolate anion and a molecule of water. Step 2 The enolate anion attacks the carbon ...
Chemical Reactions
... • We use chemical equations to summarize the process of the reactions • Chemical equations should be balanced in order to show that mass is conserved during a reaction • The principle that during chemical reactions, the mass of the products is always equal to the mass of the reactants, is known as t ...
... • We use chemical equations to summarize the process of the reactions • Chemical equations should be balanced in order to show that mass is conserved during a reaction • The principle that during chemical reactions, the mass of the products is always equal to the mass of the reactants, is known as t ...
name
... 1. A bond formed when atoms share one or more pairs of electrons 2. A range of values used to express the acidity or alkalinity of a substance 3. Any compound that releases hydrogen ions (hydronium ions) in solution 4. The attractive force between oppositely charged ions, which is created when elect ...
... 1. A bond formed when atoms share one or more pairs of electrons 2. A range of values used to express the acidity or alkalinity of a substance 3. Any compound that releases hydrogen ions (hydronium ions) in solution 4. The attractive force between oppositely charged ions, which is created when elect ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.