Chemistry 235, Winter 2008 Name: General rules:
... • Hydrogen in an organic compound is assigned an oxidation number of +1. • Oxygen in an organic compound generally is assigned an oxidation number of –2. • The sum of all of the oxidation numbers of atoms in a neutral compound should be zero. The sum of all of the oxidation numbers of atoms in an io ...
... • Hydrogen in an organic compound is assigned an oxidation number of +1. • Oxygen in an organic compound generally is assigned an oxidation number of –2. • The sum of all of the oxidation numbers of atoms in a neutral compound should be zero. The sum of all of the oxidation numbers of atoms in an io ...
File
... • Ethene is made from ethanol from removing water using AlO as catalyst • Elimination reaction is one in which a small molecule is removed from a larger molecule to leave a double bond in the larger molecules • Dehydration reaction • Only need to know dehydration of alcohol ...
... • Ethene is made from ethanol from removing water using AlO as catalyst • Elimination reaction is one in which a small molecule is removed from a larger molecule to leave a double bond in the larger molecules • Dehydration reaction • Only need to know dehydration of alcohol ...
Workshop 9
... change as more data become available. Mechanisms map the path by which the reactants change into products and the movement of electrons that accompanies this change. They also show how reactants come together, the intermediates formed, and structures of transition states. Bond cleavage and formation ...
... change as more data become available. Mechanisms map the path by which the reactants change into products and the movement of electrons that accompanies this change. They also show how reactants come together, the intermediates formed, and structures of transition states. Bond cleavage and formation ...
F:\CH 361 2014\Prelab 6 2011 CH361 modified for 2014.wpd
... organic chemistry textbook and the rest of experiment II in your lab manual. This prelab has two pages. ...
... organic chemistry textbook and the rest of experiment II in your lab manual. This prelab has two pages. ...
Microsoft Word
... Our synthesis began with the chiral aldehyde (2), obtained easily from D-mannitol, which was subjected to a Wittig reaction to give compound 3, in 90% yield. Hydrogenation of compound 3 over 10% Pd/C in methanol gave acetonide 4 followed on treating with conc. HCl in methanol afforded diol 5 in 97% ...
... Our synthesis began with the chiral aldehyde (2), obtained easily from D-mannitol, which was subjected to a Wittig reaction to give compound 3, in 90% yield. Hydrogenation of compound 3 over 10% Pd/C in methanol gave acetonide 4 followed on treating with conc. HCl in methanol afforded diol 5 in 97% ...
3.9alcohol rxns
... Oxidation of Alcohols The reactions of alcohols have a central role in organic chemistry because alcohols can be converted to many of the other functional groups. Reduction is • a gain of electrons • a less positive oxidation number • a gain of hydrogen atoms • the loss of oxygen atoms • the loss o ...
... Oxidation of Alcohols The reactions of alcohols have a central role in organic chemistry because alcohols can be converted to many of the other functional groups. Reduction is • a gain of electrons • a less positive oxidation number • a gain of hydrogen atoms • the loss of oxygen atoms • the loss o ...
Experiment 7: Acidity of Alcohols Williamson Ether Synthesis of
... The first step in this synthesis is preparation of sodium methoxide by reaction of anhydrous methanol with sodium metal. Anhydrous methanol (10 mL) is added to a clean, dry 25 mL round bottom flask containing a stir bar. The flask is fitted with an air condenser and a water cooled condenser as shown ...
... The first step in this synthesis is preparation of sodium methoxide by reaction of anhydrous methanol with sodium metal. Anhydrous methanol (10 mL) is added to a clean, dry 25 mL round bottom flask containing a stir bar. The flask is fitted with an air condenser and a water cooled condenser as shown ...
Word document format
... synthesis problems. If you don't have these "basics" down, your success will be minimal. 2. If you don't see how to begin the synthesis, work backwards from the final product. Use the logic from step 1, above. Ask yourself, how would I prepare the final product in one step? (How do you prepare a fun ...
... synthesis problems. If you don't have these "basics" down, your success will be minimal. 2. If you don't see how to begin the synthesis, work backwards from the final product. Use the logic from step 1, above. Ask yourself, how would I prepare the final product in one step? (How do you prepare a fun ...
Begin Chemical Equations Practice
... • In a chemical reaction, the materials to the left of the arrow are the reactants. Reactants are consumed in the reaction. • The arrow is like an equal sign in math. It can be read “react to produce” or “produces” or “forms”. • The materials to the right of the arrow are the products. They form as ...
... • In a chemical reaction, the materials to the left of the arrow are the reactants. Reactants are consumed in the reaction. • The arrow is like an equal sign in math. It can be read “react to produce” or “produces” or “forms”. • The materials to the right of the arrow are the products. They form as ...
Reactions to know from Chapters 17, 18, 19
... Starting with either an aldehyde or a ketone, you can see that the hemiacetals formed are characterized by having a carbon bonded to an OH- group and an OR- group. Here, the oxygen of the alcohol attacks and bonds with the carbonyl carbon of the aldehyde or ketone If the alcohol group and the ...
... Starting with either an aldehyde or a ketone, you can see that the hemiacetals formed are characterized by having a carbon bonded to an OH- group and an OR- group. Here, the oxygen of the alcohol attacks and bonds with the carbonyl carbon of the aldehyde or ketone If the alcohol group and the ...
CHE 312 Answers in BOLD RED EXAM 1 KEY (Ch. 16
... Williamson Ether Synthesis is best performed by reacting A. an alcohol with dilute warm acid B. the alkoxide of a primary alcohol with any alkyl halide C. any alkoxide with a primary alkyl halide D. two alcohols with dilute warm acid ...
... Williamson Ether Synthesis is best performed by reacting A. an alcohol with dilute warm acid B. the alkoxide of a primary alcohol with any alkyl halide C. any alkoxide with a primary alkyl halide D. two alcohols with dilute warm acid ...
experiment 10 - Faculty Web Pages
... where A, B, C, and D all exist as ions in solution. Will a reaction happen, and if so, what will be the products? Each of the positive ions could combine with the negative ion of the other compound, i.e. A+ and D¯ and C+ and B¯. The formation of a precipitate, the evolution of a gas, and a temperatu ...
... where A, B, C, and D all exist as ions in solution. Will a reaction happen, and if so, what will be the products? Each of the positive ions could combine with the negative ion of the other compound, i.e. A+ and D¯ and C+ and B¯. The formation of a precipitate, the evolution of a gas, and a temperatu ...
Slide 1 - Mrs. Reed Science Classes
... percentage yield of magnesium chloride if 100. g of magnesium react with excess hydrochloric acid to yield 330. g of magnesium chloride. a. 71.8% c. 81.6% b. 74.3% d. 84.2% ...
... percentage yield of magnesium chloride if 100. g of magnesium react with excess hydrochloric acid to yield 330. g of magnesium chloride. a. 71.8% c. 81.6% b. 74.3% d. 84.2% ...
PDF(343KB)
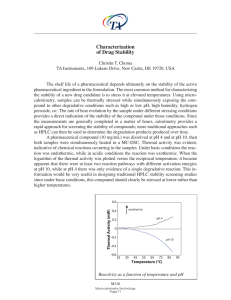
... rapid approach for screening the stability of compounds; more traditional approaches such as HPLC can then be used to determine the degradation products produced over time. A pharmaceutical compound (10 mg/mL) was dissolved at pH 4 and at pH 10, then both samples were simultaneously heated in a MC-D ...
... rapid approach for screening the stability of compounds; more traditional approaches such as HPLC can then be used to determine the degradation products produced over time. A pharmaceutical compound (10 mg/mL) was dissolved at pH 4 and at pH 10, then both samples were simultaneously heated in a MC-D ...
SYNOPSIS
... ethylamine in tetrahydrofuran and water at reflux temperature to give aziridine 20 in good yield. The NH group of aziridine 20 was protected using methyl chloroformate, TEA in dry DCM at 0oC to afford carbamate 8 in 90% yield (scheme 5). The nucleophilic ring opening of aziridine 8 with aryl cuprate ...
... ethylamine in tetrahydrofuran and water at reflux temperature to give aziridine 20 in good yield. The NH group of aziridine 20 was protected using methyl chloroformate, TEA in dry DCM at 0oC to afford carbamate 8 in 90% yield (scheme 5). The nucleophilic ring opening of aziridine 8 with aryl cuprate ...
Heck Reactions
... total synthesis of (-)-triptolide, (-)-triptonide, (+)triptophenolide, and (+)-triptquinonide." J. Org. Chem. 2000, 65, 2208-2217. An application of Crisp’s method for the synthesis of γ-lactones from β-keto esters. ...
... total synthesis of (-)-triptolide, (-)-triptonide, (+)triptophenolide, and (+)-triptquinonide." J. Org. Chem. 2000, 65, 2208-2217. An application of Crisp’s method for the synthesis of γ-lactones from β-keto esters. ...
Episode 21
... A compound formed only of hydrogen and carbon atoms. 7. What is meant by an isomer? Two molecules that have the same formula but have different structures. 8. How are organic compounds divided into categories? By functional groups. 9. How are esters used? Flavorings and scents. 10. What were the “in ...
... A compound formed only of hydrogen and carbon atoms. 7. What is meant by an isomer? Two molecules that have the same formula but have different structures. 8. How are organic compounds divided into categories? By functional groups. 9. How are esters used? Flavorings and scents. 10. What were the “in ...
Johnson Group Research
... inert under a vast majority of standard reaction conditions, certain transition metal complexes promote the activation of these bonds. Research in the Johnson group will follow several avenues of study, including: 1) Understanding Current Methodology The most current activation methodology utilizes ...
... inert under a vast majority of standard reaction conditions, certain transition metal complexes promote the activation of these bonds. Research in the Johnson group will follow several avenues of study, including: 1) Understanding Current Methodology The most current activation methodology utilizes ...
Exam 2 Fall 2005 Chemsitry 1211
... problem correctly will gain you some points but you will not receive full credit for any question you do not answer completely. Included in this exam is a periodic table, which should be stapled to the back of the exam. CHEATING IS NOT ALLOWED. ANYONE CAUGHT CHEATING WILL RECEIVE A ZERO ON THIS EXAM ...
... problem correctly will gain you some points but you will not receive full credit for any question you do not answer completely. Included in this exam is a periodic table, which should be stapled to the back of the exam. CHEATING IS NOT ALLOWED. ANYONE CAUGHT CHEATING WILL RECEIVE A ZERO ON THIS EXAM ...
Practice with Chemical Equilibrium (Chapter 14) (Due 2/17)
... (d) Addition of a catalyst (e) Pressure decrease (f) Removal of water with a desiccant ...
... (d) Addition of a catalyst (e) Pressure decrease (f) Removal of water with a desiccant ...
Organic Notes #5 - RX`ns - Winston Knoll Collegiate
... Condensation (Dehydration)-two smaller molecules are linked to create a larger molecule. This is made possible by the removal of a hydrogen form one molecule and a hydroxide from the other which creates water. Esters are made this way by reacting a carboxylic acid and an alcohol. O RCOH carboxylic ...
... Condensation (Dehydration)-two smaller molecules are linked to create a larger molecule. This is made possible by the removal of a hydrogen form one molecule and a hydroxide from the other which creates water. Esters are made this way by reacting a carboxylic acid and an alcohol. O RCOH carboxylic ...
Intro to Organic Reactions
... AlCl3 is a catalyst. Catalysts typically used in aromatic substitutions. ...
... AlCl3 is a catalyst. Catalysts typically used in aromatic substitutions. ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.