* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Intro to Organic Reactions
George S. Hammond wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Hydroformylation wikipedia , lookup
Polythiophene wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Ene reaction wikipedia , lookup
Petasis reaction wikipedia , lookup
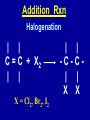
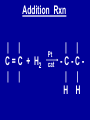
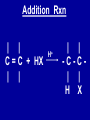
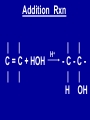
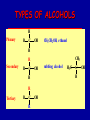
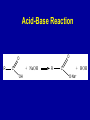
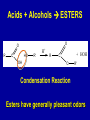
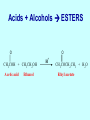
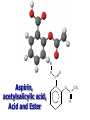
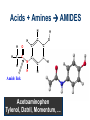
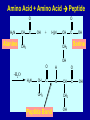
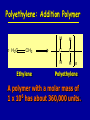
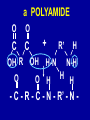
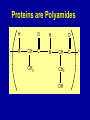
Intro to Organic Reactions Reactions of Alkanes • They burn! • Hydrocarbon and Oxygen yields Carbon Dioxide and Water Reactions of Alkanes Substitution Rxn H H + Cl2 C H H UV lite Cl H + HCl C H H Reactions of Alkenes Addition Reactions • Alkenes are unsaturated — more single bonds can form to the C atoms • Molecules such as Br2, H2, HCl, HBr, and H2O add to the double bond Addition Rxn Halogenation | | C = C + X2 | | X = Cl2, Br2, I2 | | -C-C| | X X An Addition Reaction Fat placed in Br2 vapor Unsaturated Fats An Addition Reaction The fat in bacon is partially unsaturated. The fat adds Br2 to the C=C bonds. Addition Rxn | | C = C + H2 | | Pt cat | | -C-C| | H H An Addition Reaction • Fats can be “hydrogenated” with H2. Saturated Fats Peanut butter has partially hydrogenated vegetable oil. Addition Rxn | | C = C + HX | | H+ | | -C-C| | H X Addition Rxn | | C = C + HOH | | H+ | | -C-C| | H OH Reactions of Aromatics • Substitutions — not additions — are typical. CH3 + CH3Cl AlCl3 + HCl AlCl3 is a catalyst. Catalysts typically used in aromatic substitutions. Alcohol Reactions Screen 11.6 Substitution Elimination—the reverse of addition TYPES OF ALCOHOLS H Primary R C OH CH3 CH2 OH, ethanol H CH3 R Secondary R C OH R R C R H3 C C H H Tertiary rubbing alcohol OH OH More Alcohol Reactions H H C H C OH ( ) + Oxidizin g agent H H H Eth anol H O C C H ( ) H Acetaldehyde + Oxidizin g agent Ethanol is a PRIMARY ALCOHOL. It is oxidized to an ALDEHYDE and then to an ACID. H H O C C OH ( ) H Acetic acid More Alcohol Reactions OH R—C—R' H Secondary alcohol oxidizing agent O R—C—R' Ketone (–R and –R' are organic groups. They may be the same or different.) SECONDARY ALCOHOLS are oxidized to KETONES — and reaction stops there. Reduction of the Carbonyl Group H O H2 cat C R R H C OH H aldehyde primary alcohol OH O LiAlH4 R C R' C R R' H ketone secondary alcohol Acid-Base Reaction O O R + NaOH C OH R C + HOH O-Na+ Acids + Alcohols ESTERS O O R HO C OH R' H+ R + HOH C O R' Condensation Reaction Esters have generally pleasant odors Acids + Alcohols ESTERS O O CH 3 COH + CH 3 CH 2 OH Acetic acid Ethanol H+ CH 3 COCH 2 CH 3 + H 2O Ethyl acetate H O Aspirin, acetylsalicylic acid, Acid and Ester C O O C O CH3 Acids + Amines AMIDES O O R H+ N H C OH H R' R C N O H Condensation Reaction + HOH R' Acids + Amines AMIDES N-methylacetamide Acids + Amines AMIDES H C H H H O H O C C C C C C N H H C H H Amide link Acetoaminophen Tylenol, Datril, Momentum, ... Amino Acid + Amino Acid Peptide O O H2N Alanine CH C OH + H2N CH C Serine CH2 CH3 OH OH O H C N O -H2O H2N CH CH3 Peptide Bond CH CH2 OH C OH Polymers • Giant molecules made by joining many small molecules called monomers • Average production is 150 kg per person annually in the U.S. Polymer Classifications • Thermoplastics (polyethylene) soften and flow when heated • Thermosetting plastics — soft initially but set to solid when heated. Cannot be resoftened. • Other classification: plastics, fibers, elastomers, coatings, adhesives Polymer Preparation • Addition polymers — directly adding monomer units together • Condensation polymers — combining monomer units and splitting out a small water (water) Polyethylene: Addition Polymer n H2C CH2 Ethylene H H C C H H n Polyethylene A polymer with a molar mass of 1 x 106 has about 360,000 units. Mechanism of Addition Polymerization Types of Polyethylene Linear, high density PE (HDPE) Branched, low density PE, LDPE Cross-linked PE, CLPE Types of Polyethylene Polymers based on Substituted Ethylenes, CH2=CHX CH2CH OH CH2CH n polyvinyl alcohol CH2CH OCCH3 n O polyvinyl acetate n polystyrene Table 11.12: others are PVC, acrylonitrile, polypropylene, polymethyl methacrylate H H C C H Cl H H H H H H H H H H C C C C C C C C C C H Cl H Cl H Cl H Cl H Cl H H H H H Cl H H H Cl C C C C C C C C C C H Cl Cl H H H Cl H H H Polystyrene • Polystyrene is nonpolar material and dissolves in organic solvents. • PS foam is mostly air, and when it dissolves it collapses to a much smaller volume. Slime! Slime is polyvinylalcohol cross-linked with boric acid Bubble Gum! A copolymer Styrene + butadiene Condensation Polymers O n HOC O COH + n HOCH2CH2OH terephthalic acid O C ethylene glycol O COCH2CH2O + H2 O n Polyethylene terephthalate (PET), a polyester Polyesters, PET Jackets made from recycled PET soda bottles Soda bottles, mylar film. Polyesters: Mechanism Polymer Recycling Symbols LDPE = HDPE = PP = V= Low density PE = 0.910-0.925 g/cm3 High density PE = 0.941-0.965 Polypropylene = 0.90 PVC (Vinyl chloride) = 1.30-1.58 a POLYAMIDE O O C C OH R O + OH O H-N H R’ H N-H H H - C - R - C - N - R’ - N - Polyamides: Nylon •Each monomer has 6 C atoms in its chain. •A polyamide link forms on elmination of HCl •Result = nylon 66 Proteins are Polyamides H N CH CH3 O H C N O CH CH2 OH C * Fats and Oils: Saponification Glyceryl stearate, a fat + NaOH O CH2 O CR O + 3 NaOH CH O CR O CH2 O CR R = —(CH2 )16CH3 OH 0 OH OH Glycerol + 3 R C O- Na+ sodium stearate (a soap)