Key Practice Exam 3
... attractive intermolecular forces). Carboxylic acids can form two hydrogen bonds (they are dimmers), followed by the monomeric hydrogen bonding between alcohols. Ketones can’t hydrogen bond but have electronegative oxygen and alkanes lack intermolecular forces. ...
... attractive intermolecular forces). Carboxylic acids can form two hydrogen bonds (they are dimmers), followed by the monomeric hydrogen bonding between alcohols. Ketones can’t hydrogen bond but have electronegative oxygen and alkanes lack intermolecular forces. ...
Reactions Homework Packet
... ____2. How many atoms of oxygen are there in 7 molecules of oxygen gas? II. Balancing Equations-Write balanced equations for each of the following reactions l. Mg + O2 magnesium oxide ...
... ____2. How many atoms of oxygen are there in 7 molecules of oxygen gas? II. Balancing Equations-Write balanced equations for each of the following reactions l. Mg + O2 magnesium oxide ...
Organic Reactions Worksheet
... For the questions below, give full structural diagrams and names for all reactants and products. Also indicate any catalysts/reaction conditions for the reaction. Also indicate the TYPE OF REACTION. 1. Oxidation with primary and secondary alcohols via sulfuric acid and potassium dichromate. a. What ...
... For the questions below, give full structural diagrams and names for all reactants and products. Also indicate any catalysts/reaction conditions for the reaction. Also indicate the TYPE OF REACTION. 1. Oxidation with primary and secondary alcohols via sulfuric acid and potassium dichromate. a. What ...
Final-01 - Yale Department of Chemistry
... 7. Kinetics/Thermodynamics: (20 pts.) Do one from pg. 12 and one from pg. 13. Keep your answer short and to the point! If you do more than two problems, cross out (with a large X through the solution) the one(s) that you do not want graded. a) “The more stable conformation of a compound is always th ...
... 7. Kinetics/Thermodynamics: (20 pts.) Do one from pg. 12 and one from pg. 13. Keep your answer short and to the point! If you do more than two problems, cross out (with a large X through the solution) the one(s) that you do not want graded. a) “The more stable conformation of a compound is always th ...
Conjugate (1,4
... • This synthesis is by Subhash P. Chavan, Pallavi Sharma, Rasapalli Sivappa, Mohan M. Bhadbhade, Rajesh G. Gonnade and Uttam R. Kalkote, J. Org. Chem. 2003, 68, ...
... • This synthesis is by Subhash P. Chavan, Pallavi Sharma, Rasapalli Sivappa, Mohan M. Bhadbhade, Rajesh G. Gonnade and Uttam R. Kalkote, J. Org. Chem. 2003, 68, ...
Diet and Exercise – Healthy Diet
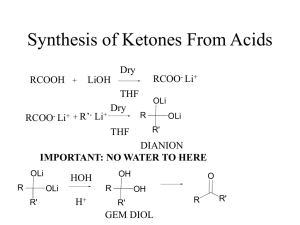
... In the reaction, the O-H bond in the alcohol is broken and water is formed. The water molecule comes from the OH of the carboxylic acid group and the H in the alcohol group ...
... In the reaction, the O-H bond in the alcohol is broken and water is formed. The water molecule comes from the OH of the carboxylic acid group and the H in the alcohol group ...
Organic Chemistry: Introduction
... tertiary carbon atoms in alcohols (-OH) and halogenoalkanes (-F, -Cl, -Br, -I) • with reference to the carbon that is directly bonded to an alcohol group or a halogen: – Primary = carbon atom is only bonded to one other carbon – Secondary = carbon atom is bonded to two other carbons – Tertiary = car ...
... tertiary carbon atoms in alcohols (-OH) and halogenoalkanes (-F, -Cl, -Br, -I) • with reference to the carbon that is directly bonded to an alcohol group or a halogen: – Primary = carbon atom is only bonded to one other carbon – Secondary = carbon atom is bonded to two other carbons – Tertiary = car ...
carboxylic acids esters amides (R
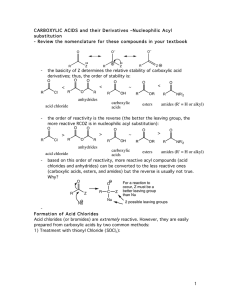
... to prepare in bulk from dirt-cheap acetic acid, and so it is often used in place of acetyl chloride (MeCOCl). Second, cyclic anhydrides are fairly easily formed by heating molecules that have two carboxylic acid functions in close proximity to high temperatures (a dehydration reaction). A couple of ...
... to prepare in bulk from dirt-cheap acetic acid, and so it is often used in place of acetyl chloride (MeCOCl). Second, cyclic anhydrides are fairly easily formed by heating molecules that have two carboxylic acid functions in close proximity to high temperatures (a dehydration reaction). A couple of ...
Organic Chemistry Control Test
... 4.1 write down a possible hypothesis for the above investigation. 4.2 why is the number of carbon atoms indicated on the x - axis? 4.3 Describe the trend that can be observed in the graph. 4.4 state one possible reason for the trend mentioned in 6.3 4.5 The boiling points of alcohols are notably hig ...
... 4.1 write down a possible hypothesis for the above investigation. 4.2 why is the number of carbon atoms indicated on the x - axis? 4.3 Describe the trend that can be observed in the graph. 4.4 state one possible reason for the trend mentioned in 6.3 4.5 The boiling points of alcohols are notably hig ...
Winter 2004 Final Exam
... a) Draw the linear form of D-glucose as a Fischer projection and circle the carbon which is inspected to determine that the configuration is ‘D’. ...
... a) Draw the linear form of D-glucose as a Fischer projection and circle the carbon which is inspected to determine that the configuration is ‘D’. ...
U. of Kentucky Chemistry 535 Synthetic Organic Chemistry Spring
... molecule. You may start with molecules containing no less than eight carbon atoms. ...
... molecule. You may start with molecules containing no less than eight carbon atoms. ...
Zumd22
... Alcohols, R–OH The –OH makes alcohol polar enough to hydrogen bond. water soluble Fermentation product but vulnerable to oxidation to aldehyde and acid. ...
... Alcohols, R–OH The –OH makes alcohol polar enough to hydrogen bond. water soluble Fermentation product but vulnerable to oxidation to aldehyde and acid. ...
Ester
... able to make soap. The reaction with water is very slow, so it is rarely used. Instead, we use a dilute acid so the reaction will happen faster. ...
... able to make soap. The reaction with water is very slow, so it is rarely used. Instead, we use a dilute acid so the reaction will happen faster. ...
Lipids practice problems
... 3) In the formation of a triglyceride, what functional groups chemically combine and what functional group is formed? 4) How does the presence of a double bond in a fatty acid (or a molecule containing a fatty acyl chain) affect its physical properties. 5) Arrange the following fatty acids in order ...
... 3) In the formation of a triglyceride, what functional groups chemically combine and what functional group is formed? 4) How does the presence of a double bond in a fatty acid (or a molecule containing a fatty acyl chain) affect its physical properties. 5) Arrange the following fatty acids in order ...
Chapter 13. Plannig and Execution of Multistep Synthesis
... The isomerization of an allyl ether to a propenyl ether can be achieved either by treatment with potassium t-butoxide in DMSO or by Wilkinson’s catalyst or (PPh3)3RhH. Heating allyl ethers with Pd/C in acidic methanol can also effect cleavage. Silyl ether plays a very important role as hydroxy-prote ...
... The isomerization of an allyl ether to a propenyl ether can be achieved either by treatment with potassium t-butoxide in DMSO or by Wilkinson’s catalyst or (PPh3)3RhH. Heating allyl ethers with Pd/C in acidic methanol can also effect cleavage. Silyl ether plays a very important role as hydroxy-prote ...
standard sample test
... 14. 2.55 g of sulfuric acid (an acid) is reacted with 1.88 g of sodium hydroxide (a base). The reaction takes place as a solution in water. After the reaction is completed the acidity or basicity of the solution is tested. Which of the following statements is true? (a) The solution was found to be a ...
... 14. 2.55 g of sulfuric acid (an acid) is reacted with 1.88 g of sodium hydroxide (a base). The reaction takes place as a solution in water. After the reaction is completed the acidity or basicity of the solution is tested. Which of the following statements is true? (a) The solution was found to be a ...
CBS Reduction
... • They used BH3 , (85% 2-MeTHF, 15% THF) and oxazaborolidine for reduction. • Under such reaction conditions, the reaction was complete in 10 minutes and alcohol was produced with 95% yield and a 91 : 9 enantiomeric ratio (highly enantioselectivety). ...
... • They used BH3 , (85% 2-MeTHF, 15% THF) and oxazaborolidine for reduction. • Under such reaction conditions, the reaction was complete in 10 minutes and alcohol was produced with 95% yield and a 91 : 9 enantiomeric ratio (highly enantioselectivety). ...
Nucleophilic Addition to Carbonyl Groups
... The reverse of acetal formation if acetal hydrolysis. This is achieved by excess water in the presence of an acid catalyst. ...
... The reverse of acetal formation if acetal hydrolysis. This is achieved by excess water in the presence of an acid catalyst. ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.