Petrochemicals - MullisChemistry
... Name the parent hydrocarbon: Use the LONGEST STRAIGHT chain of carbon atoms. Add the name of the alkyl groups attached to the chain. If more than one group is attached, use the proper numerical prefix to indicate how many groups are attached. (2=di, 3-tri,etc.) Assign numbers to the carbons in the p ...
... Name the parent hydrocarbon: Use the LONGEST STRAIGHT chain of carbon atoms. Add the name of the alkyl groups attached to the chain. If more than one group is attached, use the proper numerical prefix to indicate how many groups are attached. (2=di, 3-tri,etc.) Assign numbers to the carbons in the p ...
Chemistry II - Mr. Dougan`s Wonderful World of Chemistry
... immediately with copious amounts of water. Part One: Heat of Neutralization of Hydrochloric Acid and Sodium Hydroxide 1. Assemble your calorimeter. Use two plastic sauce containers, a lid, and a rubber band. Wrap the rubber band around one of the cups near the top. Place the cup with the rubber band ...
... immediately with copious amounts of water. Part One: Heat of Neutralization of Hydrochloric Acid and Sodium Hydroxide 1. Assemble your calorimeter. Use two plastic sauce containers, a lid, and a rubber band. Wrap the rubber band around one of the cups near the top. Place the cup with the rubber band ...
20 - Wiley
... 20 Fatty Acids and Energy • Fatty acids in triglycerides are the principle storage form of energy for most organisms • carbon chains are in a highly reduced form • the energy yield per gram of fatty acid oxidized is greater than that per gram of carbohydrate ...
... 20 Fatty Acids and Energy • Fatty acids in triglycerides are the principle storage form of energy for most organisms • carbon chains are in a highly reduced form • the energy yield per gram of fatty acid oxidized is greater than that per gram of carbohydrate ...
Rates of Reaction
... – Increasing the population of reactants increases the likelihood of a successful collision. – In some reactions, however, the rate is unaffected by the concentration of a particular reactant, as long as it is present at some concentration. ...
... – Increasing the population of reactants increases the likelihood of a successful collision. – In some reactions, however, the rate is unaffected by the concentration of a particular reactant, as long as it is present at some concentration. ...
Unit-5-Lipids
... carbohydrates, and lipids. The lipids are the subject of this unit. Of these four families, the lipids are the structurally the most diverse. This is because unlike members of the other three families, members of this families do no share a common structural feature, but rather share a common physic ...
... carbohydrates, and lipids. The lipids are the subject of this unit. Of these four families, the lipids are the structurally the most diverse. This is because unlike members of the other three families, members of this families do no share a common structural feature, but rather share a common physic ...
Unit-5-LipidsClick copy
... carbohydrates, and lipids. The lipids are the subject of this unit. Of these four families, the lipids are the structurally the most diverse. This is because unlike members of the other three families, members of this families do no share a common structural feature, but rather share a common physic ...
... carbohydrates, and lipids. The lipids are the subject of this unit. Of these four families, the lipids are the structurally the most diverse. This is because unlike members of the other three families, members of this families do no share a common structural feature, but rather share a common physic ...
Iridium(III) and Rhodium(III) compounds of dipyridyl-N
... acetonitrile showed fragmentation of molecular ion peaks at m/z = 470 and 435 due to ([(η5 -C5 Me5 )Rh {(C5 H4 N)2 C=N-Me}Cl])+ ([2]PF6 -PF6 )+ and ([(η5 C5 Me5 )Rh{(C5 H4 N)2 C=N-Me}])+ ([2]PF6 -PF6 -Cl)+ (see Supplementary Information). In addition, the spectrum also showed peak at m/z 421 due to ...
... acetonitrile showed fragmentation of molecular ion peaks at m/z = 470 and 435 due to ([(η5 -C5 Me5 )Rh {(C5 H4 N)2 C=N-Me}Cl])+ ([2]PF6 -PF6 )+ and ([(η5 C5 Me5 )Rh{(C5 H4 N)2 C=N-Me}])+ ([2]PF6 -PF6 -Cl)+ (see Supplementary Information). In addition, the spectrum also showed peak at m/z 421 due to ...
3.Redox
... B. Solubility - All ionic compounds are strong electrolytes in that they are 100% ionized, however some are not particularly soluble in water. Qualitatively the solubility of most common salts adhere to the following rules. 1. Most all Group 1 and NH4+ are soluble irrespective of the anion. 2. All ...
... B. Solubility - All ionic compounds are strong electrolytes in that they are 100% ionized, however some are not particularly soluble in water. Qualitatively the solubility of most common salts adhere to the following rules. 1. Most all Group 1 and NH4+ are soluble irrespective of the anion. 2. All ...
Chapter 2 - Chemistry
... • Ag(s) + H2O(l) Ag less active than H2 Ag(s) + H2O(l) No Reaction • Ni(s) + H2SO4(aq) ...
... • Ag(s) + H2O(l) Ag less active than H2 Ag(s) + H2O(l) No Reaction • Ni(s) + H2SO4(aq) ...
Improved Synthesis of Seven-Coordinate Molybdenum( I I) and
... for a number of years. These compounds have been prepared by a variety of methods, including reductive alkylation4v5of molybdenum(1V) cyanide complexes, chemical oxidationsJOof metal(0) carbonyl or isocyanide complexes followed by displacement of the carbonyl ligands by isocyanide, and photochemical ...
... for a number of years. These compounds have been prepared by a variety of methods, including reductive alkylation4v5of molybdenum(1V) cyanide complexes, chemical oxidationsJOof metal(0) carbonyl or isocyanide complexes followed by displacement of the carbonyl ligands by isocyanide, and photochemical ...
08272012BC Science Chem 12 Chapter 1 Answer Key
... 2. There is a common misconception that a significant increase in the volume of water will occur as water is formed in a reaction that occurs in aqueous solution. This is, of course, nonsense! As the entire reaction occurs in the solvent water, there will simply be a small amount of water formed, re ...
... 2. There is a common misconception that a significant increase in the volume of water will occur as water is formed in a reaction that occurs in aqueous solution. This is, of course, nonsense! As the entire reaction occurs in the solvent water, there will simply be a small amount of water formed, re ...
Reactions of Acyl Chlorides
... Reactions of Acyl Chlorides Acyl chlorides react with water to give carboxylic acids (carboxylate ion in base): O RCCl + H2O ...
... Reactions of Acyl Chlorides Acyl chlorides react with water to give carboxylic acids (carboxylate ion in base): O RCCl + H2O ...
Molecular geometry, polarity MULTIPLE CHOICE QUESTIONS
... 52. The compound shown below is a synthetic estrogen. It is marketed as an oral contraceptive under the name Enovid. OH ...
... 52. The compound shown below is a synthetic estrogen. It is marketed as an oral contraceptive under the name Enovid. OH ...
organic-chemistry-10th-edition-solomons-test-bank
... 52. The compound shown below is a synthetic estrogen. It is marketed as an oral contraceptive under the name Enovid. OH ...
... 52. The compound shown below is a synthetic estrogen. It is marketed as an oral contraceptive under the name Enovid. OH ...
10.1 Intro to Organic Chemistry 10.1 Organic Chemistry
... Carbon can only make four bonds since it has 4 valence electrons and most often bonds to H, O, N and S. Because the C-C single bond (348 kJ mol1) and the C-H bond (412 kJ mol-1) are strong, carbon compounds are stable. Carbon can form chains and rings. ...
... Carbon can only make four bonds since it has 4 valence electrons and most often bonds to H, O, N and S. Because the C-C single bond (348 kJ mol1) and the C-H bond (412 kJ mol-1) are strong, carbon compounds are stable. Carbon can form chains and rings. ...
Carboxylic Acids And Their Derivatives
... The Factors affecting acidity of carboxylic acids. The different ways to make carboxylic acids Salt formation reactions of carboxylic acids The nucleophilic substitution reactions at the carbonyl carbon and the specific products formed in each case. The chemistry of carboxylic acid derivatives ...
... The Factors affecting acidity of carboxylic acids. The different ways to make carboxylic acids Salt formation reactions of carboxylic acids The nucleophilic substitution reactions at the carbonyl carbon and the specific products formed in each case. The chemistry of carboxylic acid derivatives ...
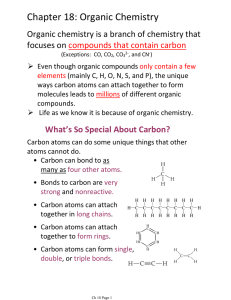
Chapter 18: Organic Chemistry
... Chapter 18: Organic Chemistry Organic chemistry is a branch of chemistry that focuses on compounds that contain carbon (Exceptions: CO, CO2, CO32-, and CN-) ...
... Chapter 18: Organic Chemistry Organic chemistry is a branch of chemistry that focuses on compounds that contain carbon (Exceptions: CO, CO2, CO32-, and CN-) ...
Carbohydrates I - ChemConnections
... Carbohydrates / Saccharides / Sugars Water, carbon dioxide, light and chlorophyll in green plants: n CO2 + n H2O Cn(H2O)n + n O2 Empirical formula = CH2O ...
... Carbohydrates / Saccharides / Sugars Water, carbon dioxide, light and chlorophyll in green plants: n CO2 + n H2O Cn(H2O)n + n O2 Empirical formula = CH2O ...
Cahn-Ingold-Prelog Notation Tutorial
... Let us return to the example of 2-methyl-1-butanol and develop the rules for assigning priority. Figure 3. An enantiomer of 2-methyl-1-butanol with priorities assigned. ...
... Let us return to the example of 2-methyl-1-butanol and develop the rules for assigning priority. Figure 3. An enantiomer of 2-methyl-1-butanol with priorities assigned. ...
Organic Chemistry II with Dr Roche
... Normal ethers do not undergo nucleophilic substitution or eliminations because the alkoxide anion is not a good leaving group. (That is why acid catalysis is required). Epoxides are different though. The release of strain when an epoxide is opened more than compensates for the poor leaving group abi ...
... Normal ethers do not undergo nucleophilic substitution or eliminations because the alkoxide anion is not a good leaving group. (That is why acid catalysis is required). Epoxides are different though. The release of strain when an epoxide is opened more than compensates for the poor leaving group abi ...
PowerPoint 演示文稿
... in his early work, to test the theory. Studies of aliphatic substitutions and eliminations, often with his long-time collaborator E. D. Hughes, led to I ncorporation into the standard language of chemistry of such words as nucleophile, electrophile, inductive and mesomeric (resonance) effects, and s ...
... in his early work, to test the theory. Studies of aliphatic substitutions and eliminations, often with his long-time collaborator E. D. Hughes, led to I ncorporation into the standard language of chemistry of such words as nucleophile, electrophile, inductive and mesomeric (resonance) effects, and s ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.