* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 2 - Chemistry
Inorganic chemistry wikipedia , lookup
Solvent models wikipedia , lookup
Chemical reaction wikipedia , lookup
Click chemistry wikipedia , lookup
History of electrochemistry wikipedia , lookup
Biochemistry wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Crystallization wikipedia , lookup
Electrochemistry wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Electrolysis of water wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Implicit solvation wikipedia , lookup
Equilibrium chemistry wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Stoichiometry wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Liquid–liquid extraction wikipedia , lookup
Acid dissociation constant wikipedia , lookup
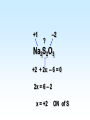
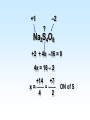
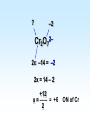
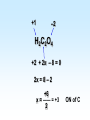
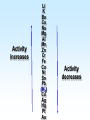
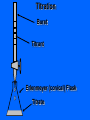
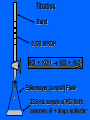
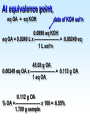
Chapter 5 Reactions in Aqueous Solutions Chapter goals • Understand the nature of ionic substances dissolved in water. • Recognize common acids and bases and understand their behavior in aqueous solution. • Recognize and write equations for the common types of reactions in aqueous solution. • Recognize common oxidizing and reducing agents and identify common oxidationreduction reactions. • Define and use the molarity in solution stoichiometry. Solution • homogeneous mixture • can be gas, liquid, or solid • solvent: component present in highest proportion exception - water • solute: component(s) in solution other than solvent We will mostly study aqueous solutions: human body is 2/3 water. Examples • mixture of 35% naphthalene and 65% benzene solvent - benzene solute – naphthalene • mixture of 10% ethanol, 40% methanol, and 50% propanol solvent - propanol solute - ethanol and methanol Examples • mixture of 40% ethanol, 40% methanol, and 20% butanol solvent - ethanol/methanol mixed solvent solute – butanol • mixture of 40% ethanol, 50% propanol, and 10% water solvent - water solute - ethanol and propanol Next… We will focus on compounds that produce ions in aqueous solutions. They are named electrolytes and may be salts, acids, or bases. Salts Salts: ionic compounds made of cations other than H+ and anions other than OH− or O2−, O22− NaCl: Na+ & Cl− K2SO4: K+ & SO42− FeBr3: Fe3+ & Br− Zn3(PO4)2: Zn2+ & PO43− Ca(HCO3)2: Ca2+ & HCO3− Electrolyte • substance that dissolves to produce an electrically conducting medium • forms ions in solution (dissociates/ionizes) • examples soluble ionic compounds H2O KBr(s) K+(aq) + Br–(aq) H2O Acids, HCl(g) H+(aq) + Cl–(aq) bases, NH3 + H2O NH4+ + OH– double arrow means equilibrium Nonelectrolytes • do not form ions in solution • do not form electrically conducting media upon dissolution • Examples: molecular compounds (alcohols, sugars & acetone) H 2O CH3OH(l) CH3OH(aq) N.D. Glucose C6H12O6(s) C6H12O6(aq) N.D. Sucrose C12H22O11(s) C12H22O11(aq) N.D. N.D. = no dissociation/ionization Types of Electrolytes • Strong: dissociate ~100% most ionic compounds (soluble salts), strong acids, and strong bases H2O KBr(s) K+(aq) + Br–(aq) HCl(g) H+(aq) + Cl–(aq) NaOH(s) Na+(aq) + OH−(aq) Weak: insoluble salts, weak acids and bases, water (H2O), and certain gases (e.g. CO2) H2O dissociate only slightly in water HF(g) H+(aq) + F–(aq) Also acetic acid, CH3COOH NH3 + H2O NH4+(aq) + OH–(aq) Solubility of Ionic compounds in Water: Solubility Rules Soluble Compounds 1. alkali metal salts (Li+, Na+, K+, Rb+…, ) except potassium perchlorate 2. ammonium (NH4+) salts 3. all nitrates(NO3−), chlorates (ClO3−), perchlorates (ClO4−), and acetates (C2H3O2−), except silver acetate and potassium perchlorate 4. all Cl−, Br−, and l− are soluble except for Ag+, Pb2+, and Hg22+ salts 5. all SO42− are soluble except for Pb2+, Sr2+, and Ba2+ salts Solubility of Ionic compounds in Water: Rules Insoluble or slightly soluble Compounds 6. metal oxides (O2−) except those of the alkali metals, Ca2+, Sr2+, and Ba2+ 7. hydroxides (OH−) except those of the alkali metals, Ba2+, Sr2+, and NH4+. Calcium hydroxide is slightly soluble 8. carbonates, phosphates, sulfides, and sulfites except those of the alkali metals and the ammonium ion (NH4+) 9. for salts of Cr2O72−, P3−, CrO42−, C2O42−, assume they are insoluble except for IA metals and NH4+ salts • • • • Precipitation Reactions: A Driving Force in Chemical Reactions formation of insoluble solid (precipitate, ppt) is a common reaction in aqueous solutions: reactants are generally water-soluble ionic compounds once substances dissolve in water they dissociate to give the appropriate cations and anions if the cation of one compound forms an insoluble compound with the anion of another, precipitation will occur Precipitation Reaction: A Double Replacement (Metathesis) Reaction • Both ionic compounds trade partner ions __________ | | AB(aq) + CD(aq) AD(s) + CB(aq) |_______| AD is an insoluble or slightly soluble salt A+, B−, C+, and D− are ions AgNO3(aq) + NaCl(aq) AgCl(s) + NaNO3(aq) weak electrolyte (unionized precipitate) Precipitation Reaction: A Double Replacement (Metathesis) Reaction A (solid) precipitate is formed. Example: complete and balance the equation (NH4)3PO4(aq) + MgSO4(aq) MgPO4(s) + NH4SO4(aq) we will write the right subscripts later Using the solubility rules, predict if at least one product is going to be insoluble in water. According to rule 8, MgPO4 (subscripts not right) is not soluble in water. Ions are Mg2+, PO43−, NH4+, and SO42−; subscripts (NH4)3PO4(aq) + MgSO4(aq) Mg3(PO4)2(s) + (NH4)2SO4(aq) balancing 2(NH4)3PO4(aq) + 3MgSO4(aq) Mg3(PO4)2(s) + 3(NH4)2SO4(aq) Example: complete and balance the equation • Na2SO4(aq) + BaBr2(aq) Na2SO4(aq) + BaBr2(aq) BaSO4(s) + NaBr Na2SO4 + BaBr2 BaSO4(s) + 2NaBr(aq) driving force = formation of insoluble barium sulfate (precipitate) • Os(NO3)5(aq) + Rb2S(aq) Os(NO3)5 + Rb2S Os2S5(s) + RbNO3(aq) 2 Os(NO3)5 + 5 Rb2S Os2S5(s) + 10 RbNO3 driving force = form. of insoluble Os5+ sulfide (precipitate) Net Ionic Equations: Spectator Ions The equation AgNO3(aq) + NaCl(aq) AgCl(s) + NaNO3(aq) is not quite correct, because three salts are dissociated in ions while AgCl is a precipitate. Ag+(aq) + NO3−(aq) + Na+(aq) + Cl−(aq) AgCl(s) + Na+(aq) + NO3−(aq) before reaction after reaction Na+ and NO3− are present on both sides of equation, i.e., before and after reaction. They are called spectator ions; they do not participate in net reaction; they can be removed from the equation, but they remain in the solution. Ag+(aq) + Cl−(aq) AgCl(s) is the net ionic equation Net Ionic Equations: Spectator Ions For two previous examples: 2(NH4)3PO4(aq) + 3MgSO4(aq) Mg3(PO4)2(s) + 3(NH4)2SO4(aq) 6NH4+(aq) + 2PO43−(aq) + 3Mg2+(aq) + 3SO42−(aq) Mg3(PO4)2(s) + 6NH4+(aq) + 3SO42−(aq) after reaction before reaction 3Mg2+(aq) + 2PO43−(aq) Mg3(PO4)2(s) is the net equation spectator ions are eliminated from the equation ======================= Na2SO4(aq) + BaBr2(aq) BaSO4(s) + 2NaBr(aq) 2Na+(aq) + SO42−(aq) + Ba2+(aq) + 2Br−(aq) BaSO4(s) + 2Na+(aq) + 2Br− Ba2+(aq) + SO42−(aq) BaSO4(s) net ionic equation Net Ionic Equations: Spectator Ions For the metathesis reaction 2 KF(aq) + Pb(NO3)2(aq) PbF2(s) + 2 KNO3(aq) formula unit equation spectator ions are eliminated 2K+ + 2F– + Pb2+ + 2 NO3– PbF2 + 2K+ + 2NO3– ionic equation PbF2 is the precipitate 2F–(aq) + Pb2+(aq) PbF2(s) net ionic equation Net Ionic Equations: Spectator Ions NH4Cl(aq) + KNO3(aq) NH4NO3(aq) + KCl(aq) NH4+ + Cl– + K+ + NO3– NH4+ + NO3– + K+ + Cl– all ions are spectators; all can be cancelled no net ionic equation no driving force for reaction N.R. (there is no reaction) The two salts are just dissolved in water. Acids and Bases Acid • Arrhenius definition substance that ionizes in water to produce H+, hydrogen ion, and hence increases the concentration of this ion HCl(aq) H+(aq) + Cl–(aq) • Brønsted-Lowry definition substance capable of donating H+ HCl + H2O H3O+ + Cl–(aq) Acids and Bases Base • Arrhenius definition substance that increases the concentration of OH– in aqueous solution KOH(aq) K+(aq) + OH–(aq) NH3 + H2O NH4+ + OH– • Brønsted/Lowry definition substance capable of accepting H+ KOH(aq) K+(aq) + OH–(aq) OH– + H+ H2O (OH– from NaOH accepts H+) NH3 + H2O NH4+ + OH– (NH3 accepts H+) Water can act as both an acid and a base: it is an amphoteric substance HClO4 + H2O H3O+ acid base (accepts H+ from HClO4) + ClO4– NH3 + H2O NH4+ + OH– base acid (donates H+ to NH3) Strong Acids dissociate ~100% • HCl, HBr, HI (no HF) hydro…ic acid • HNO3 nitric acid • HClO3 chloric acid (moderate) • HClO4 perchloric acid • H2SO4 (first proton) sulfuric acid H2SO4(aq) H+(aq) + HSO4−(aq) 2nd weak: HSO4−(aq) H+(aq) + SO42−(aq) Weak Acids • dissociate <100% • most other acids HF hydrofluoric acid HCN hydrocyanic acid HNO2 nitrous acid CH3CO2H acetic acid H2CO3 carbonic acid (both protons) H3PO4 phosphoric acid (all protons) H2SO3 sulfurous acid (both protons) oxalic acid H2C2O4(aq) H+(aq) + HC2O4−(aq) Strong Bases dissociate ~100% • alkali metal hydroxides LiOH, NaOH, KOH, RbOH name: lithium hydroxide • hydroxide of Ca Ca(OH)2 calcium hydroxide Ba Ba(OH)2 Sr Sr(OH)2 Ammonia, NH3, is a weak base Neutralization Reactions • acid + OH-ctg. base salt + water (a double replacement reaction) HF(aq) + KOH(aq) KF(aq) + H2O HF(aq) + K+(aq) + OH–(aq) K+(aq) + F–(aq) + H2O HF(aq) + OH–(aq) F–(aq) + H2O net ionic spectator ions are eliminated from equation HF is a weak acid and HCl is a strong acid • acid + non-OH-ctg base salt HCl(aq) + NH3(aq) NH4Cl(aq) H+(aq) + Cl–(aq) + NH3(aq) NH4+(aq) + Cl–(aq) H+(aq) + NH3(aq) NH4+(aq) net ionic equation Neutralization Reactions • Strong acid + strong base salt + water (a double replacement reaction) HClO3(aq) + NaOH(aq) NaClO3(aq) + H2O chloric acid H+(aq) + ClO3−(aq) + Na+(aq) + OH–(aq) Na+(aq) + ClO3–(aq) + H2O H+(aq) + OH–(aq) H2O net ionic equation spectator ions, ClO3− + Na+, are eliminated from equation Formation of a Weak Acid or Base as a Driving Force (another double replacement reaction) • HNO3(aq) + KCN(aq) HCN(aq) + KNO3(aq) H+(aq) + NO3–(aq) + K+(aq) + CN–(aq) HCN (aq) + K+(aq) + NO3–(aq) H+(aq) + CN–(aq) HCN(aq) (a weak acid) • NH4Cl + NaOH(aq) NH4OH + NaCl(aq) NH4+(aq) + Cl–(aq) + Na+(aq) + OH–(aq) NH4OH Na+(aq) + Cl–(aq) NH4+(aq) + OH–(aq) NH4OH (a weak base) NH4OH is NH3 in water, i.e., NH3 + H2O When no Weak Electrolytes are Formed • HNO3(aq) + KCl(aq) HCl(aq) + KNO3(aq) H+(aq) + NO3–(aq) + K+(aq) + Cl–(aq) H+(aq) + Cl–(aq) + K+(aq) + NO3–(aq) There is no net reaction: N.R. No driving force All ions are spectators. • BaCl2(aq) + 2NaOH(aq) Ba(OH)2(aq) + 2NaCl(aq) Ba2+(aq) + 2Cl–(aq) + 2Na+(aq) + 2OH–(aq) Ba2+(aq) + 2OH–(aq) + 2Na+(aq) + 2Cl–(aq) There is no net reaction: N.R. No driving force Gas Forming Reactions (a Driving Force) Some of the weak acids and bases that are formed at double replacement reactions decompose to form a gas and water CO2 Na2CO3(aq) + 2HCl(aq) H2CO3(aq) + 2NaCl(aq) H2CO3(aq) H2O + CO2(g) Na2CO3(aq) + 2HCl(aq) H2O + CO2(g) + 2NaCl(aq) SO2 Na2SO3(aq) + 2HCl(aq) H2SO3(aq) + 2NaCl(aq) H2SO3(aq) H2O + SO2(g) Na2SO3(aq) + 2HCl(aq) H2O + SO2(g) + 2NaCl(aq) Redox (Oxidation-Reduction) Reactions • • • • involve transfer of electron(s) oxidation: loss of electron(s) reduction: gain of electron(s) some can be identified when an uncombined element is a reactant or a product • • • • • eg. Zn(s) + Cu2+(aq) Zn2+(aq) + Cu(s) Zn Zn2+ Zn(s) Zn2+(aq) + 2 e–, oxidation Cu2+ Cu Cu2+(aq) + 2e– Cu(s), reduction Single Displacement Reactions • • • • Zn(s) + CuCl2(aq) Cu(s) + ZnCl2(aq) Zn oxidized to Zn2+ Cu2+ reduced to Cu occurs because zinc is more active than copper • • • • Cl2(g) + CuBr2(aq) Br2(l) + CuCl2(aq) Br oxidized from Br– to Br2 Cl reduced from Cl2 to Cl– Cl is more active than Br Oxidation Numbers • also an accounting tool • very useful • oxidation numbers of all atoms in substance add up to charge on substance e.g. (charge of species) zero for Al2(SO4)3 and H3PO4 +1 for NH4+ –2 for Cr2O72– Assigning Oxidation Numbers, ON • ON = 0 for all atoms in any substance in most elemental form, Na(s), Zn(s), Hg(l) H2(g), Cl2(g), I2(s), O2(g), C(s), P4(s), S8(s) • ON = charge for monatomic ions (Na+, S2−) • ON = –1 for F in all compounds • ON = –2 for O in compounds, usually – exceptions: peroxide, O22–, ON = –1 – superoxide, O2–, ON = –1/2 • ON = +1 for H in compounds, usually – exception: ON = –1 in metallic hydrides Assigning Oxidation Numbers, ON • ON = +1 for alkali metals in compounds • ON = +2 for alkaline earth metals in compounds • ON = +3 for Al in compounds • ON = −1 for Cl, Br, and I (iodine) in binary compounds except for those with oxygen (in these cases they variable positive) • F is always −1 Assign ON to Each Atom in the Following Substances ? –1 WCl6 x + 6(–1) = 0 x –6 = 0 x = +6 +1 ? –2 Na2S2O3 +2 + 2x – 6 = 0 2x = 6 – 2 x = +2 ON of S –2 +1 ? Na2S4O8 +2 + 4x –16 = 0 4x = 16 – 2 +14 +7 x = —— = —— 4 2 ON of S ? –2 Cr2O72– 2x –14 = –2 2x = 14 – 2 +12 x = —— = +6 2 ON of Cr +1 –2 H2C2O4 +2 + 2x – 8 = 0 2x = 8 – 2 +6 x = —— = +3 2 ON of C ? –1 MoBr5+ x – 5 = +1 x=5+1 x=6 ON of Mo = +6 Oxidizing and Reducing Agents In every redox reaction there is (at least) a Reducing agent (the one that is oxidized) and an oxidizing agent (the one that is reduced) ON increases The species is oxidized +7 +6 +5 +4 +3 +2 +1 0 −1 −2 −3 −4 −5 −6 −7 ON decreases The species is reduced • Activity (Electromotive) Series for metals Activity increases Li K Ba Ca Na Mg Al Mn Zn Cr Fe Co Ni Sn Pb (H2) Cu Ag Hg Pt Au Activity decreases Examples • Complete and balance each of the following chemical equations Li A free and chemically active metal displacing K Ba a less active metal from a compound Ca • Mg + FeCl3 Na Mg Mg + FeCl3 Fe + MgCl2 Al 3 2 Mn Mg Mg2+, oxidized; Mg reducing agent Zn Cr Fe3+ Fe, reduced; Fe3+ oxidizing agent Fe Co Ni 3 Sn Sn + CrF3 No Reaction Pb H2 Cu 3 3 Hg Pb(s) + Au(ClO3)3(aq) Au(s) + Pb(ClO3)2(aq) Ag Pt 3Pb(s) + 2Au(ClO3)3(aq) 2Au(s) + 3Pb(ClO3)2(aq) Au +3 3Mg(s) + 2FeCl (aq) 2Fe(s) + 3MgCl (aq) • Sn + CrF Sn is less reactive than Cr • Pb(s) + Au(ClO ) (aq) Pb is oxidized Au is reduced A free and chemically active metal displacing a less active metal from a compound • Zn + CrBr3 Zn + CrBr3 Cr + ZnBr2 3Zn(s) + 2CrBr3(aq) 2Cr(s) + 3 ZnBr2(aq) • Zn oxidized to Zn2+; Zn reducing agent • Cr3+ reduced to Cr; Cr3+ oxidizing agent • Ag(s) + Hg(NO3)2 No Reaction Ag is less reactive than Hg Li K Ba Ca Na Mg Al Mn Zn Cr Fe Co Ni Sn Pb H2 Cu Hg Ag Pt Au A free and chemically active metal displacing Li K Hydrogen from acids or water Ba Ca • Fe + HBr Na Mg Fe + HBr H2 + FeBr3 0 +1 0 +3 2Fe + 6HBr 3H2 + 2 FeBr3 Fe oxidized to Fe3+; Fe reducing agent H+ reduced to H2; H+ oxidizing agent • Cu + HBr Cu less active than H2 Cu + HBr No Reaction Al Mn Zn Cr Fe Co Ni Sn Pb H2 Cu Hg Ag Pt Au A free and chemically active metal displacing Li K Hydrogen from acids or water Ba • K(s) + H2O(l) Ca 0 +1 +1 0 2K(s) + 2H2O(l) 2 KOH(aq) + H2(g) K oxidized to K+; K reducing agent H+ reduced to H2; H+ oxidizing agent • Ag(s) + H2O(l) Ag less active than H2 Ag(s) + H2O(l) No Reaction • Ni(s) + H2SO4(aq) 0 +1 +2 0 Ni(s) + H2SO4(aq) NiSO4(aq) + H2(g) Ni oxidized to Ni2+; Ni reducing agent H+ reduced to H2; H+ oxidizing agent Na Mg Al Mn Zn Cr Fe Co Ni Sn Pb H2 Cu Hg Ag Pt Au • Which one of the following metals could be used safely for lining a tank intended for storage of sulfuric acid? H2SO4 • aluminum • iron • chromium • mercury • copper • tin Li K Ba Ca Na Mg Al Mn Zn Cr Fe Co Ni Sn Pb H2 Cu Hg Ag Pt Au Nonmetal Activity Series F Cl Br I same order as in periodic table F > Cl > Br > I Complete and Balance Cl2 + FeBr3 Cl2(g) + FeBr3(aq) Br2(l) + FeCl3(aq) • 0 −1 0 −1 3Cl2(g) + 2FeBr3(aq) 3 Br2(l) + 2 FeCl3(aq) Cl2 oxidizing agent; Br– reducing agent • I2(s) + NaF(aq) I2 is less active than F2 I2(s) + NaF(aq) No Reaction • F2(g) + NaCl(aq) 0 −1 o −1 F2(g) + 2 NaCl(aq) Cl2(g) + 2 NaF(aq) F2 oxidizing agent; Cl– reducing agent • Br2 + FeCl3 Br less active than Cl Br2 + FeCl3 No Reaction Identifying Oxidizing and Reducing Agents 0 +3 0 +2 3Zn(s) + 2CrBr3(aq) 2Cr(s) + 3 ZnBr2(aq) • Zn oxidized to Zn2+; Zn reducing agent • Cr3+ reduced to Cr; Cr3+ oxidizing agent 0 +1 +2 0 Ni(s) + H2SO4(aq) NiSO4(aq)) + H2(g) Ni oxidized to Ni2+; Ni reducing agent H+ reduced to H2; H+ oxidizing agent 0 -1 0 -1 F2(g) + 2 NaCl(aq) Cl2(g) + 2 NaF(aq) F2 reduced; Cl– oxidized F2 oxidizing agent; Cl– reducing agent Identifying Oxidizing and Reducing Agents • The device for testing breath for the presence of alcohol is based on the following reaction. Identify the oxidizing and reducing agents ON: –1 +6 3CH3CH2OH(aq) + 2Cr2O72–(aq) + 16H+ ethanol orange-red +3 3CH3CO2H(aq) + 4 Cr3+(aq) + 11H2O acetic acid green ethanol is oxidized (reducing agent) Cr2O72– (dichromate ion) is reduced (oxidizing agt.) Balancing redox equations in acidic media (presence of H+) Cr2O72–(aq) + Fe2+(aq) Cr3+(aq) + Fe3+(aq) ON: +6 Cr2O72– + 6e− 2Cr3+ Fe2+ Fe3+ + e− (this the reduction) (this is the oxidation) Cr2O72– + 6e− + 14H+ 2Cr3+ (to have +6 = +6, charge) Cr2O72– + 6e− + 14H+ 2Cr3+ + 7H2O (to balance H & O) Cr2O72– + 6e− + 14H+ 2Cr3+ + 7H2O 6(Fe2+ Fe3+ + e−) to equal # of electrons, 6 Cr2O72– + 14H+ + 6Fe2+ 2Cr3+ + 7H2O + 6Fe3+ Balancing redox equations in basic media (OH−) +4 +6 MnO4–(aq) + SO32−(aq) MnO2(s) + SO42−(aq) ON: +7 +4 MnO4– + 3e− MnO2 SO32− SO42− + 2e− (this the reduction) (this is the oxidation) MnO4– + 3e− MnO2 + 4OH− SO32− + 2OH− SO42− + 2e− (to equal charges, −4) (to equal charges, −4) 2(MnO4– + 3e− +2H2O MnO2 + 4OH−) 3(SO32− + 2OH− SO42− + 2e− + H2O) to equal H, O, and electrons Measuring Concentrations of Compounds in Solutions Concentration Terms Parts Per • Hundred (percent, %) weight/weight, %(w/w) (most common) mass solute (g) ———————— x 100 mass solution (g) volume/volume, %(v/v) V solute (mL) ———————— x 100 V solution (mL) liquid solute in liquid solvent Parts Per • Hundred (percent, %) weight/volume, %(w/v) mass solute (g) ———————— x 100 V solution (mL) Learning Check A solution is prepared by mixing 15.0 g of Na2CO3 and 235 g of H2O. The final V of solution is 242 mL. Calculate the %w/w and %w/v concentration of the solution. g solution = 15.0 g Na2CO3 + 235 g H2O = 250. g 15.0 g solute %w/w = ——————— x 100 = 6.00% Na2CO3 250. g solution 15.0 g solute %w/v = ——————— x 100 = 6.20% Na2CO3 242 mL solution Molarity, M The Molarity, M, usually known as the molar concentration of a solute in a solution, is the number of moles of solute per liter (1000 mL) of solution / or mmoles per mL of solution. To calculate it we need moles of solute and V(in liters) of solution (or mmol and mL) and to divide mol solute mmol solute M = —————— = ——————— V(L) solution V(mL) solution Calculation of Molarity, M What is the molarity of 500. mL NaOH solution if it contains 6.00 g NaOH? 500. mL 1000 = 0.500 L (volume in liters) FW (NaOH) = 40.0 g/mol (from periodic table) How many moles of NaOH? 1 mol NaOH 6.00 g x —————— = 0.150 mol NaOH 40.0 g NaOH mol solute 0.150 mol M = —————— = ————— = 0.300 M NaOH V(L) solution 0.500 L (0.300 mol in 1 L or 0.300 mmol in 1 mL) Formality, F is the same as molarity, but referred to ionic compounds in aqueous solution FW (formula weights) of solute per L of solution 1 FW # FW = g solute x ————— = # of FW = # of moles g solute # FW F = —————— V(L) solution Formality = Molarity (# of formula weights) (volume in liters) Molality, m is the amount (moles) of solute per kg of solvent (usually but not necessarily water). What is the m of a solution prepared by dissolving 25.3 g Na2CO3 in 458 g water? 458 g = 0.458 kg (after dividing by 1000) 1 mol Na2CO3 25.3 g Na2CO3 x —————— = 0.239 mol Na2CO3 106.0 g Na2CO3 mol solute 0.239 mol m = —————— = ————— = 0.522 m Na2CO3 kg H2O 0.458 kg (0.522 mol in 1 kg H2O) Mole Fraction, X is the amount (mol) of a given component of a solution per mol of solution. Here we need # moles of every component (solute(s) and solvent) and the total e.g. for a solution with n1, n2, n3, … mol ni ni mol fraction Xi = —————— = —— (no units) n1+ n2+ n3 + … nt ni: moles of component i (1, 2, 3, …) nt: total number of moles How many g of NaCl are contained in 250.0 mL of 0.2193 M NaCl solution? 250.0 mL 1000 = 0.2500 L Now, M as a conversion factor 0.2193 mol 0.2500 L x ————— = 0.0548 mol NaCl 1 L sol’n grams out of moles and formula weight (58.44) 58.44 g NaCl 0.0548 mol x —————— = 3.204 g NaCl 1 mol NaCl Making Solutions • Consider the making of 1.00 L of 1.00 M NaCl solution. • need 1.00 mol NaCl or 58.5 g NaCl • dissolve 58.5 g NaCl in 1.00 L water? NO!! • dissolve 58.5 g NaCl in less than 1.000 L water and dilute to a total volume of 1.000 L Example: Describe the preparation of 300.0 mL of 0.4281 M silver nitrate solution. 300.0 mL 1000 = 0.3000 L AgNO3 FW = 169.97 g/mol 0.4281 mol 0.3000 L x ————— = 0.1284 mol AgNO3 1 L sol’n 169.97 g 0.1284 mol x ————— = 21.82 g AgNO3 1 mol AgNO3 • Dissolve 21.82 g AgNO3 in 300.0 mL water? NO!! • Dissolve 21.82 g AgNO3 in water and dilute to 300.0 mL. How many milliliters of 2.00 M HNO3 contain 24.0 g HNO3? HNO3 FW = 63.0 g/mol How many moles? 1 mol HNO3 24.0 g HNO3 x —————— = 0.381 mol HNO3 63.0 g HNO3 Now the M with the volume on top to get mL 1 L sol’n 1000 mL 0.381 mol x —————— x ———— = 191 mL 2.00 mol HNO3 1 L sl’n How many grams of AlCl3 are needed to prepare 25 mL of a 0.150 M solution? 25 mL 1000 = 0.025 L FW (AlCl3) = 133.5 g/mol All at once: V(L) of sol’n and M to calculate mol of AlCl3 mol and FW to calculate g of AlCl3 0.025 L x 0.150 mole x 133.5 g = 0.500 g AlCl3 1L 1 mole Dilution the process of decreasing the concentration of solutes in a solution by addition of solvent or another solution that does not contain the same solutes • volume increases and concentration decreases. concentrated diluted solution volumetric flask: calibrated to contain, tc pipet: calibrated to deliver, td Example: Calculate the concentration of a solution made by diluting 25.0 mL of 0.200 M methanol, CH3OH, solution to 100.0 mL. Key for calculations: moles of solute taken from the concentrated solution are the same in the diluted solution (only solvent is added.) moles = concentration x V = M x V Cc x Vc = Cd x Vd Mc x Vc = Md x Vd one of the four is unknown c: concentrated d: diluted L or mL can be used for volume Example: Calculate the concentration of a solution made by diluting 25.0 mL of 0.200 M methanol, CH3OH, to 100.0 mL. Mc x V c = Md x V d Md is the unknown 25.0 mL x 0.200 M = 100.0 mL x Md 25.0 mL x 0.200 M Md = ————————— = 0.0500 M 100.0 mL 0.0500 mole/L How many mL of a 0.515 M NaBr solution must be diluted to produce 500.0 mL of a 0.103 M NaBr solution? Mc x V c = Md x V d Vc is the unknown Vc x 0.515 M = 500.0 mL x 0.103 M 500.0 mL x 0.103 M Vc = ————————— = 100.0 mL 0.515 M Serial Dilutions Example: Calculate the M of NaOH in a solution made by diluting 25.0 mL of 0.928 M NaOH to 200.0 mL and then diluting 50.0 mL of the second solution to 100.0 mL. Mc x Vc = Md x Vd First dilution: 25.0 mL x 0.928 M Md = ————————— = 0.116 M 200.0 mL Second dilution: 50.0 mL x 0.116 M Md = ————————— = 0.0580 M 100.0 mL Solution Stoichiometry Use of M, V, and coefficients in equations to calculate any amount of reagent or product. • Example: What volume of 0.273 M potassium chloride solution is required to react with exactly 0.836 mmol of silver nitrate? • KCl(aq) + AgNO3(aq) • KCl(aq) + AgNO3(aq) KNO3(aq) + AgCl(s) • driving force, formation of precipitate AgCl • Key: work with mmol of KCl and AgNO3 Solution Stoichiometry What volume of 0.273 M KCl solution is required to react with exactly 0.836 mmol of AgNO3 ? KCl(aq) + AgNO3(aq) KNO3(aq) + AgCl(s) First, calculate mmol of KCl: 1 mmol KCl 0.836 mmol AgNO3 x—————— = 0.836 mmol 1 mmol AgNO3 KCl Second, calculate volume of KCl solution: 1 mL 0.836 mmol KCl x ———— = 3.06 mL of solution 0.273 mmol (KCl) Titration Buret Titrant Erlenmeyer (conical) Flask Titrate Titration • buret: calibrated td, fine tip to deliver small volumes accurately, stopcock for flow control • Erlenmeyer flask: sloped walls allow swirling of solutions without spilling or splashing • Titrant: solution containing reagent that will react with sample in well known manner • Titrate: solution containing the sample • • • • • Titration equivalence point: point in a titration at which the exact stoichiometric amount of titrant has been added to react with the titrate end point: the point in a titration at which the indicator changes, titration stopped here and volume of titrant read. ideally, end point = equivalence point reality, not so, error introduced, hopefully small error Four parameters: V, M of titrant and V, M of titrate. Usually one is unknown. Titration Example: Calculate the concentration of hydrochloric acid in a solution if 35.0 mL of it required 28.9 mL of 0.178 M potassium hydroxide solution for titration. HCl: titrated, V known, M unknown (in this problem) KOH: titrant, V and M known Titration Buret 0.178 M KOH HCl + KOH KCl + H2O Erlenmeyer (conical) Flask 35.0 mL sample of HCl Sol’n, unknown M + drops indicator Titration Strategy: mmol of KOH are calculated first. Second, by using stoichiometry coefficients mol of HCl are calculated. Finally, M of HCl is calculated with mmol and V(mL) 0.178 mmol KOH 1 mmol HCl 28.9 mLx———————— x————— = 5.14 mmol 1 mL 1 mmol KOH HCl Based on the end point concept (mol ratio) 5.14 mmol MHCl = ————— = 0.147 M (mol/L) 35.0 mL (mmol/mL) Example: Calculate the concentration of arsenic acid in a solution given that a 25.0 mL sample of that solution required 42.2 mL of 0.274 M potassium hydroxide for titration. • H3AsO4 + KOH K3AsO4 + H2O • H3AsO4 + 3 KOH K3AsO4 + 3 H2O M of H3AsO4 is the unknown. 1 mol H3AsO4 reacts with 3 mol KOH. That is the mole ratio. We can use mL and mmol instead of L and mol Example: Calculate the concentration of arsenic acid in a solution given that a 25.0 mL sample of that solution required 42.2 mL of 0.274 M potassium hydroxide for titration. • H3AsO4 + 3 KOH K3AsO4 + 3 H2O 0.274 mmol KOH 1 mmol H3AsO4 42.2 mLx———————— x————— = 3.85 mmol 1 mL 3 mmol KOH H3AsO4 3.85 mmol M H3AsO4 = ————— = 0.154 M (mol/L) 25.0 mL (mmol/mL) Purity Analysis: A 1.034 g-sample of impure oxalic acid is dissolved in water and an acidbase indicator added. The sample requires 34.47 mL of 0.485 M NaOH solution to reach the equivalence point. What is the mass of H2C2O4 and what is its mass percent in the sample? • H2C2O4 + 2 NaOH Na2C2O4 + 2 H2O Strategy: mol of H2C2O4 out of mol of NaOH (by using V and M). Then, mol of H2C2O4 will be used to calculate g of H2C2O4 and, hence, % of it in the sample. Coefficients are 1 for H2C2O4 and 2 for NaOH. Oxalic acid: Oxac M W = 90.04 g/mol H2C2O4 + 2 NaOH Na2C2O4 + 2 H2O 0.485 mol 1 mol Oxac 34.47 mLx————— x—————— = 0.00836 mol 1000 mL 2 mol NaOH Oxac Based on the end point concept (mol ratio) 90.04 g 0.00836 mol x ———— = 0.753 g oxalic acid 1 mol 0.753 g oxac %Oxac = ——————— x 100 = 72.8% 1.034 g of sample Molar Mass of an Acid: A 1.056 g of a pure acid, HA, is dissolved in water and an acid-base indicator added. The solution requires 33.78 mL of 0.256 M NaOH solution to reach the equivalence point. What is the molar mass of the acid? We don’t know what A is • HA + NaOH NaA + H2O Strategy: mol of HA out of mol of NaOH (by using V and M). Then, mol of HA and g of HA will be used to calculate the molar mass. Coefficients are 1 for HA and 1 for NaOH. Molar Mass of HA 33.78 mL NaOH solution = 0.03378 L HA + NaOH NaA + H2O 0.256 mol NaOH 1 mol HA 0.03378 Lx———————— x————— = 8.65x10-3 1 L sol’n 1 mol NaOH mol HA Molar mass, 1.056 g HA M W = ——————— = 122 g/mol 8.65x10-3 mol HA 122 grams per mol Vinegar: A 25.0-mL sample of vinegar (which contains the weak acetic acid, CH3CO2H) requires 28.33 mL of a 0.953 M solution of NaOH for titration to the equivalence point. What mass of the acid, in grams, is in the vinegar sample and what is the M of acetic acid in the vinegar? • CH3CO2H + NaOH NaCH3CO2 + H2O Strategy: mol of CH3CO2H (HOAc) out of mol of NaOH (by using V and M). Then, g of HOAc will be calculated with the molar mass. The M will be calculated by using the volume of vinegar. Coefficients are 1 for HOAc and 1 for NaOH. CH3CO2H: HOAc M W = 60.05 g/mol 0.953 mol 1 mol HOAc 28.33 mLx————— x—————— = 0.0270 mol 1000 mL 1 mol NaOH HOAc 60.05 g 0.0270 mol x ——— = 1.62 g acetic acid in vinegar 1 mol For the molarity, Vvinegar = 25.0 mL = 0.0250 L (sol’n) 0.0270 mol HOAc M HOAc = ————————— = 1.08 M 0.0250 L sol’n Problem: To analyze an iron-containing compound, you convert all the iron into Fe2+ in aqueous solution and then titrate the solution with standardized KMnO4. The balanced-net ionic equation is MnO4− + 5Fe2+ + 8H+ Mn2+ + 5Fe3+ + 4H2O A 0.598-g sample of the iron compound requires 22.25 mL of 0.0123 M KMnO4 solution for titration to the equivalence point. What is the mass % of iron in the sample? Strategy: mL and M of MnO4− to mol of MnO4− and Fe2+ Coefficients are 1 and 5 for MnO4− and Fe2+ respectively Fe2+ and MnO4− (mole ratio is 5 to 1): 0.0123 mol MnO4− 5 mol Fe2+ 22.25 mLx ———————— x————— = 1.37x10−3 1000 mL 1 mol MnO4− mol Fe2+ 55.85 g Fe g of iron: 1.37 x 10−3 mol Fe2+ x ————— = 0.0765 1 mol Fe2+ g Fe Now, for the mass % of iron: 0.0765 g Fe ———————— x 100 = 12.8% Fe 0.598 g sample Normality • equivalents (eq) solute per liter solution • milliequivalents (meq) solute per mL solution eq solute meq solute N = —————— = —————— (calculated by dividing) V(L) sol’n V(mL) sol’n • equivalent = 1 equivalent weight • Equivalent Weight (EW); given in g/eq acid/base: the mass of a substance required to furnish or react with exactly 1 mol H+ redox reactions: the mass of substance able to gain or lose 1 mol e− Equivalent weight HCl • HCl H+ + Cl– • 1 mol HCl 1 mol H+, EW = MW H2SO4 • H2SO4 2H+ + SO42– • 1 mol H2SO4 2 mol H+, EW = MW/2 HnA (in general) • EW = MW/n, n = # H+ in molecule of acid KOH • KOH K+ + OH– • OH– + H+ H2O • , 1 mol KOH reacts with 1 mol H+, EW = FW • continued with polyhydroxides… Equivalent weight Fe(OH)3 • 1 mol Fe(OH)3 3OH– • 1 mol reacts with 3 mol H+, EW = FW/3 M(OH)n (in general) • EW = FW/n, n = # OH– in formula unit of base Redox: +7 MnO4– + 5e– Mn2+ FW MnO4– EW = ————— 5 0 Zn Zn2+ + 2 e– AW Zn EW = ——— 2 Example: Calculate the normality of barium hydroxide in a solution made by dissolving 0.991 g of barium hydroxide in water and diluting to 100.0 mL. Ba(OH)2 FW 171.32 g mg EW = —— = ———— = 85.66 —— = 85.66 —— 2 2 eq meq 1 eq Ba(OH)2 0.991 g Ba(OH)2 x —————— = 0.0116 eq Ba(OH)2 85.66 g Ba(OH)2 0.0116 eq Ba(OH)2 N Ba(OH)2 = ————————— = 0.116 N Ba(OH)2 0.1000 L sol’n Example: Describe the preparation of 250.0 mL of 0.100 N oxalic acid solution from the solid. Oxalic acid is H2C2O4. • 1 mol oxalic acid 2 mol H+ • EW = 1/2 MW = 1/2(90.04) = 45.03g/eq • 0.100 N indicates 0.100 eq OA/L sol’n 0.100 eq OA 0.2500 L x —————— 1 L sol’n = 0.0250 eq OA 45.03 g OA 0.0250 eq OA x —————— = 1.13 g OA 1 eq OA • Dissolve 1.13 g of oxalic acid in water and dilute to a total volume of 250.0 mL Example: What is the normality of a 0.300 M arsenic acid solution? • H3AsO4 • 1 mol H3AsO4 3 mol H+ MW EW = —— 3 g H3AsO4 g H3AsO4 g H3AsO4 eq = ————— = ———— = 3 x ————— = 3 mol EW H3AsO4 MW MW H3AsO4 H3AsO4 3 eq N = —— V(L) mol M = ——— V(L) Then, N H3AsO4 = 3 x M H3AsO4 = 3 x 0.300 = 0.900 N CONCLUSION: For an Acid HnA or a Base M(OH)n N =nxM n = # H+ in molecule of acid or # OH– in formula unit of base By definition, 1 eq of anything will react with exactly 1 eq of anything else Equivalence Point The point in a titration at which eq titrant = eq titrate meq titrant = meq titrate Example: Calculate the concentration of phosphoric acid in a solution given that a 25.0 mL sample of that solution required 42.2 mL of 0.274 N potassium hydroxide for titration. • H3PO4 + 3 KOH K3PO4 + 3 H2O • at Eq. Pt., meq H3PO4 = meq KOH meq H3PO4 = meq KOH (mL H3PO4)(N H3PO4) = ( mL KOH)(N KOH) (25.0 mL)(X ) = (42.2 mL)(0.274 N ) X = 0.463 N Now, N = 3x M, then M = N/3 X = 0.463/3 = 0.154 M Example: Calculate the % oxalic acid (H2C2O4) in a solid given that a 1.709 g sample of the solid required 24.9 mL of 0.0998 N potassium hydroxide for titration. • H2C2O4 + 2KOH K2C2O4 + 2H2O Due to the two protons of H2C2O4 (OA), • EW = MW/2 = 45.03g/eq = 45.03mg/meq g OA % OA = ————— x 100 g sample Strategy: calculate eq of NaOH that are the same for OA; then, calculate g of OA with eq and EW of OA At equivalence point, eq OA = eq KOH data of KOH sol’n 0.0998 eq KOH eq OA = 0.0249 L x ——————— = 0.00249 eq 1 L sol’n 45.03 g OA 0.00249 eq OA x ——————— = 0.112 g OA 1 eq OA 0.112 g OA % OA = ——————— x 100 = 6.55% 1.709 g sample