Compounds with Oxygen Atoms
... Compounds of phenol are the active ingredients in the essential oils of nutmeg, thyme, cloves, and vanilla. ...
... Compounds of phenol are the active ingredients in the essential oils of nutmeg, thyme, cloves, and vanilla. ...
Carbon–hydrogen bond activation of chloroalkanes by a rhodium
... One issue that remains to be addressed is the mechanism of b-chloride elimination to generate 3. The intermediate alkyl hydride complexes in these reactions are formally d6 octahedral Rh(III) species, which should be inert towards ligand loss. Yet b-elimination of the chloro group would be expected ...
... One issue that remains to be addressed is the mechanism of b-chloride elimination to generate 3. The intermediate alkyl hydride complexes in these reactions are formally d6 octahedral Rh(III) species, which should be inert towards ligand loss. Yet b-elimination of the chloro group would be expected ...
Slide 1
... Alcohol Properties: • Alcohol molecules are polar • Alcohol molecules can create hydrogen bonds • Alcohols usually have a neutral pH, but phenols are weak acids ...
... Alcohol Properties: • Alcohol molecules are polar • Alcohol molecules can create hydrogen bonds • Alcohols usually have a neutral pH, but phenols are weak acids ...
Developments in Synthetic Application of Selenium(IV) Oxide and
... both research and industry. Among various oxidants selenium compounds, mainly selenium(IV) oxide (commonly named selenium dioxide, (1) and organoselenium compounds, presented in Scheme 1, such as selenoxides (2), areneseleninic acids (3) and their anhydrides (4), selenides (5), diaryl diselenides (6 ...
... both research and industry. Among various oxidants selenium compounds, mainly selenium(IV) oxide (commonly named selenium dioxide, (1) and organoselenium compounds, presented in Scheme 1, such as selenoxides (2), areneseleninic acids (3) and their anhydrides (4), selenides (5), diaryl diselenides (6 ...
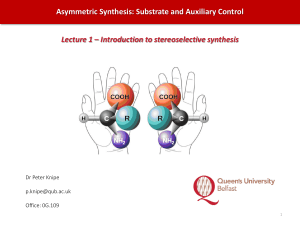
Asymmetric Synthesis: Substrate and Auxiliary Control
... ▪ Overview of stereospecificity due to Woodward-Hoffmann rules; aspects of stereoselectivity, e.g. endo rule; use of auxiliaries to control absolute stereochemistry ...
... ▪ Overview of stereospecificity due to Woodward-Hoffmann rules; aspects of stereoselectivity, e.g. endo rule; use of auxiliaries to control absolute stereochemistry ...
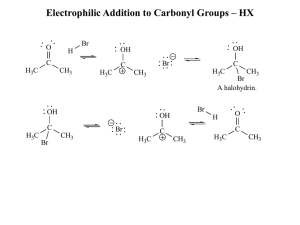
Electrophilic Addition to Carbonyl Groups – HX
... Nucleophilic Addition to Carbonyl Groups: Enolate and Aldol Reactions • This reaction is known as the aldol reaction. It takes an aldehyde and converts it into an alcohol (while extending the carbon chain). • If an aldol reaction is worked up under acidic conditions, an E2 reaction will follow, giv ...
... Nucleophilic Addition to Carbonyl Groups: Enolate and Aldol Reactions • This reaction is known as the aldol reaction. It takes an aldehyde and converts it into an alcohol (while extending the carbon chain). • If an aldol reaction is worked up under acidic conditions, an E2 reaction will follow, giv ...
No Slide Title
... Alcohols are named according to standard IUPAC rules • select the longest chain of C atoms containing the O-H group; • remove the e and add ol after the basic name • number the chain starting from the end nearer the O-H group • the number is placed after the an and before the ol ... e.g butan-2-ol • ...
... Alcohols are named according to standard IUPAC rules • select the longest chain of C atoms containing the O-H group; • remove the e and add ol after the basic name • number the chain starting from the end nearer the O-H group • the number is placed after the an and before the ol ... e.g butan-2-ol • ...
Caboxylic acid Derivatives
... Carboxylic acid derivatives are described as compounds that can be converted to carboxylic acids via simple acidic or basic hydrolysis. The most important acid derivatives are esters, amides and nitriles, although acid halides and anhydrides are also derivatives (really activated forms of a carboxyl ...
... Carboxylic acid derivatives are described as compounds that can be converted to carboxylic acids via simple acidic or basic hydrolysis. The most important acid derivatives are esters, amides and nitriles, although acid halides and anhydrides are also derivatives (really activated forms of a carboxyl ...
+ H 2 O(l) - Knockhardy
... Alcohols are named according to standard IUPAC rules • select the longest chain of C atoms containing the O-H group; • remove the e and add ol after the basic name • number the chain starting from the end nearer the O-H group • the number is placed after the an and before the ol ... e.g butan-2-ol • ...
... Alcohols are named according to standard IUPAC rules • select the longest chain of C atoms containing the O-H group; • remove the e and add ol after the basic name • number the chain starting from the end nearer the O-H group • the number is placed after the an and before the ol ... e.g butan-2-ol • ...
File
... 6. How can you tell the difference between a “cis” and “trans” isomer? In what types of hydrocarbons can they be found? 7. What is an aromatic compound, and what is the simplest aromatic structure? 8. Do aromatic hydrocarbons react more than saturated or unsaturated hydrocarbons? Explain why. 9. Wha ...
... 6. How can you tell the difference between a “cis” and “trans” isomer? In what types of hydrocarbons can they be found? 7. What is an aromatic compound, and what is the simplest aromatic structure? 8. Do aromatic hydrocarbons react more than saturated or unsaturated hydrocarbons? Explain why. 9. Wha ...
Full-Text PDF
... by the coordination between the atoms present in the aromatic moiety and those in the metal core. Keywords: formylation; TiCl4; aromatic rings; aldehydes; dichloromethyl methyl ether ...
... by the coordination between the atoms present in the aromatic moiety and those in the metal core. Keywords: formylation; TiCl4; aromatic rings; aldehydes; dichloromethyl methyl ether ...
ALcohols CPP
... Aromatic • in aromatic alcohols (or phenols) the OH is attached directly to the ring • an OH on a side chain of a ring behaves as a typical aliphatic alcohol The first two compounds are classified as aromatic alcohols (phenols) because the OH group is attached directly to the ring. ...
... Aromatic • in aromatic alcohols (or phenols) the OH is attached directly to the ring • an OH on a side chain of a ring behaves as a typical aliphatic alcohol The first two compounds are classified as aromatic alcohols (phenols) because the OH group is attached directly to the ring. ...
PDF - Nanyang Technological University
... Supporting information for this article is available on the WWW under http://dx.doi.org/10.1002/anie.201109054. Angew. Chem. Int. Ed. 2012, 51, 3649 –3652 ...
... Supporting information for this article is available on the WWW under http://dx.doi.org/10.1002/anie.201109054. Angew. Chem. Int. Ed. 2012, 51, 3649 –3652 ...
Alcohols
... common names for low-molecular-weight ethers – common names are derived by listing the alkyl groups bonded to oxygen in alphabetical order and adding the word "ether” – alternatively, name one of the groups on oxygen as an ...
... common names for low-molecular-weight ethers – common names are derived by listing the alkyl groups bonded to oxygen in alphabetical order and adding the word "ether” – alternatively, name one of the groups on oxygen as an ...
the suzuki-miyaura reaction and boron reagents – mechanism
... A) Four-coordinate “active” boron species is preformed and associates with the product of the oxidative addition: The boronate pathway. B) Ligand (alkoxy or hydroxy group) on the palladium acts as a Lewis base, generating the four-coordinate species: The oxo-palladium pathway. ...
... A) Four-coordinate “active” boron species is preformed and associates with the product of the oxidative addition: The boronate pathway. B) Ligand (alkoxy or hydroxy group) on the palladium acts as a Lewis base, generating the four-coordinate species: The oxo-palladium pathway. ...
- Sacramento - California State University
... PART ONE: INVESTIGATION OF SCHIFF BASES FOR THE VANADIUMCATALYZED ASYMMETRIC EPOXIDATION OF ALLYLIC ALCOHOLS ...
... PART ONE: INVESTIGATION OF SCHIFF BASES FOR THE VANADIUMCATALYZED ASYMMETRIC EPOXIDATION OF ALLYLIC ALCOHOLS ...
Module 2 Asymmetric Carbon-Carbon Bond Forming Reactions
... 2.1.1 Carbonyl-Ene Reaction Chiral Lewis acid catalyzed enantioselective ene reaction is one of the efficient methods for atom economical carbon-carbon bond formation. For example, TiBINOL prepared in situ catalyzes efficiently the carbonyl-ene reaction of glyoxylate with -methylstyrene in the pres ...
... 2.1.1 Carbonyl-Ene Reaction Chiral Lewis acid catalyzed enantioselective ene reaction is one of the efficient methods for atom economical carbon-carbon bond formation. For example, TiBINOL prepared in situ catalyzes efficiently the carbonyl-ene reaction of glyoxylate with -methylstyrene in the pres ...
Chemistry 2100 - Bonham Chemistry
... – Both IUPAC and common names of esters are derived from the names of the parent carboxylic acids. – Name the alkyl or aryl group bonded to oxygen first, followed by the name of the acid; replace the suffix -ic acid by -ate. – A cyclic ester is called a lactone. ...
... – Both IUPAC and common names of esters are derived from the names of the parent carboxylic acids. – Name the alkyl or aryl group bonded to oxygen first, followed by the name of the acid; replace the suffix -ic acid by -ate. – A cyclic ester is called a lactone. ...
Document
... • the role of the acid catalyst is to protonate the carbonyl oxygen and increase its electrophilic character toward attack by water to form a tetrahedral carbonyl addition intermediate • collapse of this intermediate gives the carboxylic acid and alcohol ...
... • the role of the acid catalyst is to protonate the carbonyl oxygen and increase its electrophilic character toward attack by water to form a tetrahedral carbonyl addition intermediate • collapse of this intermediate gives the carboxylic acid and alcohol ...
Isoindolone Formation via Intramolecular Diels
... was discontinued before this stage could be scaled further. Whilst it was found that the resulting isoindolone 7 could be converted easily to the corresponding acid 2 by simple hydrolysis, coupling to the amidoxime 5 could be achieved directly with better yield and processing by removing one synthet ...
... was discontinued before this stage could be scaled further. Whilst it was found that the resulting isoindolone 7 could be converted easily to the corresponding acid 2 by simple hydrolysis, coupling to the amidoxime 5 could be achieved directly with better yield and processing by removing one synthet ...
Boron and Metal Catalyzed CC and CH Bond Formation
... Research efforts focused on the use of boron and metals to form new carboncarbon and carbon-hydrogen bonds are summarized in this dissertation. Several novel reactions have been developed. These include: the deoxygenation of benzylic alcohols using chloroboranes, alkenylation of benzylic alcohols us ...
... Research efforts focused on the use of boron and metals to form new carboncarbon and carbon-hydrogen bonds are summarized in this dissertation. Several novel reactions have been developed. These include: the deoxygenation of benzylic alcohols using chloroboranes, alkenylation of benzylic alcohols us ...
Organic Chemistry II CHM 2211 Sample Exam 2 Answer Key
... When an aldehyde is considered as a group, it is called carbaldehyde group. When a ketone cannot be named as an -one, it is named as an oxo- group. ...
... When an aldehyde is considered as a group, it is called carbaldehyde group. When a ketone cannot be named as an -one, it is named as an oxo- group. ...
Alkenes and Alkynes I
... 6A. How to Favor an E2 Mechanism Use a secondary or tertiary alkyl halide if possible. (Because steric hinderance in the substrate will inhibit substitution) When a synthesis must begin with a primary alkyl halide, use a bulky base. (Because the steric bulk of the base will inhibit substitution) ...
... 6A. How to Favor an E2 Mechanism Use a secondary or tertiary alkyl halide if possible. (Because steric hinderance in the substrate will inhibit substitution) When a synthesis must begin with a primary alkyl halide, use a bulky base. (Because the steric bulk of the base will inhibit substitution) ...
Ans:- (i) Gluconic acid - Kendriya Vidyalaya No.2, Kribhco, Surat
... Ans: Negative type of deviation is present. In the negative deviation the solute-solution (A-A) interaction and solvent-solvent (B-B) interaction will be weaker than solute-solvent(A-B ) interaction. Since the new forces are stronger therefore heat is evolved and solution becomes warm. Q13. Which ty ...
... Ans: Negative type of deviation is present. In the negative deviation the solute-solution (A-A) interaction and solvent-solvent (B-B) interaction will be weaker than solute-solvent(A-B ) interaction. Since the new forces are stronger therefore heat is evolved and solution becomes warm. Q13. Which ty ...
Haloalkane

The haloalkanes (also known, as halogenoalkanes or alkyl halides) are a group of chemical compounds derived from alkanes containing one or more halogens. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially and, consequently, are known under many chemical and commercial names. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes which contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone layer depleter. For more information, see Halomethane. Haloalkane or alkyl halides are the compounds which have the general formula ″RX″ where R is an alkyl or substituted alkyl group and X is a halogen (F, Cl, Br, I).Haloalkanes have been known for centuries. Chloroethane was produced synthetically in the 15th century. The systematic synthesis of such compounds developed in the 19th century in step with the development of organic chemistry and the understanding of the structure of alkanes. Methods were developed for the selective formation of C-halogen bonds. Especially versatile methods included the addition of halogens to alkenes, hydrohalogenation of alkenes, and the conversion of alcohols to alkyl halides. These methods are so reliable and so easily implemented that haloalkanes became cheaply available for use in industrial chemistry because the halide could be further replaced by other functional groups.While most haloalkanes are human-produced, non-artificial-source haloalkanes do occur on Earth, mostly through enzyme-mediated synthesis by bacteria, fungi, and especially sea macroalgae (seaweeds). More than 1600 halogenated organics have been identified, with bromoalkanes being the most common haloalkanes. Brominated organics in biology range from biologically produced methyl bromide to non-alkane aromatics and unsaturates (indoles, terpenes, acetogenins, and phenols). Halogenated alkanes in land plants are more rare, but do occur, as for example the fluoroacetate produced as a toxin by at least 40 species of known plants. Specific dehalogenase enzymes in bacteria which remove halogens from haloalkanes, are also known.