kinetic and thermodynamic studies of the oxidation of perfumery
... *E-mail: [email protected] ABSTRACT Oxidation is one of the most important industrial reactions as it yields useful products. Literature survey indicates the use of a variety of organic oxidants for the oxidation of alcohols to the corresponding carbonyl compounds but inorganic oxidants have rare ...
... *E-mail: [email protected] ABSTRACT Oxidation is one of the most important industrial reactions as it yields useful products. Literature survey indicates the use of a variety of organic oxidants for the oxidation of alcohols to the corresponding carbonyl compounds but inorganic oxidants have rare ...
Alcohols Phenols and Ethers
... bond and one for the hydroxyl group. The –ol suffix is last and takes precedence in the numbering. 3. If the hydroxyl group is directly attached to an aromatic ring, the compound is named as a phenol. 4. If the hydroxyl group occurs in a carboxylic acid, aldehyde, or ketone, it is named as a substit ...
... bond and one for the hydroxyl group. The –ol suffix is last and takes precedence in the numbering. 3. If the hydroxyl group is directly attached to an aromatic ring, the compound is named as a phenol. 4. If the hydroxyl group occurs in a carboxylic acid, aldehyde, or ketone, it is named as a substit ...
C 1 hapter
... most useful reactions in organic synthesis. It provides a way of transforming alkenes into many different types of C*-X and C*-R bonds through the optically enriched organoboron adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through ...
... most useful reactions in organic synthesis. It provides a way of transforming alkenes into many different types of C*-X and C*-R bonds through the optically enriched organoboron adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through ...
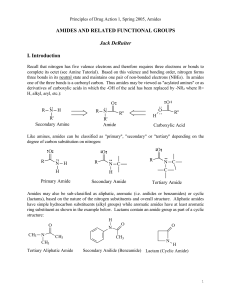
AMIDES AND RELATED FUNCTIONAL GROUPS
... A. Carbamates Carbamates may be view as "ester-amide" hybrid structures and they display chemical reactivity which is, at least, comparable to these functional groups. As amide-like structures, they do not behave as acids or bases over the normal pH range (1-14). This again, is due to delocalization ...
... A. Carbamates Carbamates may be view as "ester-amide" hybrid structures and they display chemical reactivity which is, at least, comparable to these functional groups. As amide-like structures, they do not behave as acids or bases over the normal pH range (1-14). This again, is due to delocalization ...
Hydrogenation, Transfer Hydrogenat- ion and Hydrogen Transfer Reactions
... The word “chirality” is derived from the Greek, χειρ (kheir) which means “hand”. Our hands cannot be superimposed onto each other but are mirror images of each other. Chirality can be traced back to the beginning of the 1900s, when the phrase was first introduced by Lord Kelvin,3 whose original stat ...
... The word “chirality” is derived from the Greek, χειρ (kheir) which means “hand”. Our hands cannot be superimposed onto each other but are mirror images of each other. Chirality can be traced back to the beginning of the 1900s, when the phrase was first introduced by Lord Kelvin,3 whose original stat ...
Organic Chemistry/Fourth Edition: e-Text
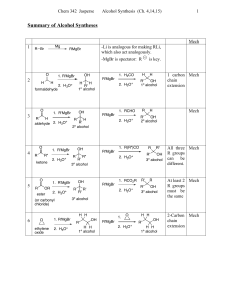
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
Chapter 3 Alcohols, Phenols, and Ethers
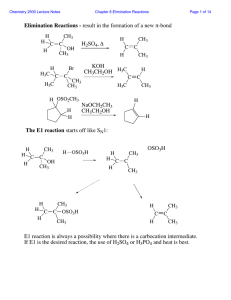
... Oxidation of Alcohols to Carbonyl Compounds • An oxidation reaction occurs when a molecule loses electrons. This is usually manifested as an increase in the number of oxygen atoms or a decrease in the number of hydrogen atoms. • Some common oxidizing agents include potassium permanganate (KMnO4), ch ...
... Oxidation of Alcohols to Carbonyl Compounds • An oxidation reaction occurs when a molecule loses electrons. This is usually manifested as an increase in the number of oxygen atoms or a decrease in the number of hydrogen atoms. • Some common oxidizing agents include potassium permanganate (KMnO4), ch ...
View/Open
... more being created daily, naming them all is a real problem. Part of the problem is due to the sheer complexity of organic structures, but part is also due to the fact that chemical names have more than one purpose. For Chemical Abstracts Service (CAS), which catalogs and indexes the worldwide chemi ...
... more being created daily, naming them all is a real problem. Part of the problem is due to the sheer complexity of organic structures, but part is also due to the fact that chemical names have more than one purpose. For Chemical Abstracts Service (CAS), which catalogs and indexes the worldwide chemi ...
Class Notes
... Super Strong Bases and Nucleophiles • The counterion metal is a spectator • Stability-reactivity principle: very unstable à very reactive • This great reactivity is very useful (as nucleophile) • This great reactivity (as base) has implication for proper technical use (see following) 7. Solvent and ...
... Super Strong Bases and Nucleophiles • The counterion metal is a spectator • Stability-reactivity principle: very unstable à very reactive • This great reactivity is very useful (as nucleophile) • This great reactivity (as base) has implication for proper technical use (see following) 7. Solvent and ...
Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... material, the intensity of signals at 1225, 1204, and 1730 cm−1, attributed to C−O (C−OH/C−O−C, hydroxyl/ epoxide) and CO (carbonyl groups), respectively, has significantly decreased. Moreover, complete disappearance of signals at 1413 and 1817 cm−1, attributed to carboxylic acid RCOO-H bending vibr ...
... material, the intensity of signals at 1225, 1204, and 1730 cm−1, attributed to C−O (C−OH/C−O−C, hydroxyl/ epoxide) and CO (carbonyl groups), respectively, has significantly decreased. Moreover, complete disappearance of signals at 1413 and 1817 cm−1, attributed to carboxylic acid RCOO-H bending vibr ...
nucleophilic addition on ketones and ketimines - ISI
... a catalyst. Intensive optimisation on the catalytic system led the authors to select i Pr-DuPHOS as a chiral ligand and a diphosphine copper(I) fluoride complex is supposed to be generated as the active catalytic species in the reaction mechanism. The fluoride ligand on copper is also essential for ca ...
... a catalyst. Intensive optimisation on the catalytic system led the authors to select i Pr-DuPHOS as a chiral ligand and a diphosphine copper(I) fluoride complex is supposed to be generated as the active catalytic species in the reaction mechanism. The fluoride ligand on copper is also essential for ca ...
Novel Transition Metal-Catalysed Syntheses of Carboxylic Acid
... known ability of rhodium(I) complexes to effect both allylic alkylation and PausonKhand reaction (PKR) to a one-pot process.[20] Alkylation of the methyl carbonate of 3buten-2-ol 13 with malonate anion 14 took place in an atmosphere of carbon monoxide and subsequent heating of the allylic substituti ...
... known ability of rhodium(I) complexes to effect both allylic alkylation and PausonKhand reaction (PKR) to a one-pot process.[20] Alkylation of the methyl carbonate of 3buten-2-ol 13 with malonate anion 14 took place in an atmosphere of carbon monoxide and subsequent heating of the allylic substituti ...
Zn(BH4)2/Al2O3: A new synthetic method for the efficient
... reduction reactions were performed efficiently in short reaction times (20–40 min) (92–95 %) (Table V). Under different conditions, attempts to reduce α-diketones into acyloins were unsatisfactory and only vicinal diols were identified as the sole products. In addition, reduction of acyloins to vici ...
... reduction reactions were performed efficiently in short reaction times (20–40 min) (92–95 %) (Table V). Under different conditions, attempts to reduce α-diketones into acyloins were unsatisfactory and only vicinal diols were identified as the sole products. In addition, reduction of acyloins to vici ...
A Simple and Advantageous Protocol for the Oxidation of Alcohols
... We regard EtOAc and DCE as the solvents of choice because they are inert and all byproducts are insoluble at room temperature, such that no purification is required beyond simple filtration. Reactions in several other solvents provided higher yields and shorter reaction times, but required chromatog ...
... We regard EtOAc and DCE as the solvents of choice because they are inert and all byproducts are insoluble at room temperature, such that no purification is required beyond simple filtration. Reactions in several other solvents provided higher yields and shorter reaction times, but required chromatog ...
2007 Nov Paper 1 - A Level Tuition
... Bupropion has a chiral carbon (marked by *) and so, is optically active. CH3 ...
... Bupropion has a chiral carbon (marked by *) and so, is optically active. CH3 ...
Extra Organic chemistry
... Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
Document
... Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
Enol esters: Versatile substrates in synthesis of fine and specialty
... Enol esters and their derivatives are promising compounds with the potential to be utilised as substrates and intermediates in organic and pharmaceutical chemistry.1,2 They can be converted to bioactive compounds such as antibiotics, steroids, and are also promising substrates for polymer synthesis ...
... Enol esters and their derivatives are promising compounds with the potential to be utilised as substrates and intermediates in organic and pharmaceutical chemistry.1,2 They can be converted to bioactive compounds such as antibiotics, steroids, and are also promising substrates for polymer synthesis ...
Efficient and Convenient Procedure for Protection of Hydroxyl
... when the reaction mixtures were stirred at room temperature for one day, and the starting materials were quantitatively recovered. The reaction conditions are mild enough not to induce any isomerization for conjugated alcohols or damage to moieties such as methoxy, which often undergoes cleavage in ...
... when the reaction mixtures were stirred at room temperature for one day, and the starting materials were quantitatively recovered. The reaction conditions are mild enough not to induce any isomerization for conjugated alcohols or damage to moieties such as methoxy, which often undergoes cleavage in ...
HIGHLY SELECTIVE RHODIUM–CATALYZED C–H BORYLATIONS IN
... knowledge in many areas of chemistry, whisky and introducing me to sweet delicious bourbon, brownest of the brown. As well, thank you Olena, Patty “2–Sheds”, Mike, Smitha and Cristina “Ooley–Mooley” for guidance, expanding my knowledge and sharing delicious beverages. Also thanks to Alphonse for ope ...
... knowledge in many areas of chemistry, whisky and introducing me to sweet delicious bourbon, brownest of the brown. As well, thank you Olena, Patty “2–Sheds”, Mike, Smitha and Cristina “Ooley–Mooley” for guidance, expanding my knowledge and sharing delicious beverages. Also thanks to Alphonse for ope ...
Haloalkane

The haloalkanes (also known, as halogenoalkanes or alkyl halides) are a group of chemical compounds derived from alkanes containing one or more halogens. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially and, consequently, are known under many chemical and commercial names. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes which contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone layer depleter. For more information, see Halomethane. Haloalkane or alkyl halides are the compounds which have the general formula ″RX″ where R is an alkyl or substituted alkyl group and X is a halogen (F, Cl, Br, I).Haloalkanes have been known for centuries. Chloroethane was produced synthetically in the 15th century. The systematic synthesis of such compounds developed in the 19th century in step with the development of organic chemistry and the understanding of the structure of alkanes. Methods were developed for the selective formation of C-halogen bonds. Especially versatile methods included the addition of halogens to alkenes, hydrohalogenation of alkenes, and the conversion of alcohols to alkyl halides. These methods are so reliable and so easily implemented that haloalkanes became cheaply available for use in industrial chemistry because the halide could be further replaced by other functional groups.While most haloalkanes are human-produced, non-artificial-source haloalkanes do occur on Earth, mostly through enzyme-mediated synthesis by bacteria, fungi, and especially sea macroalgae (seaweeds). More than 1600 halogenated organics have been identified, with bromoalkanes being the most common haloalkanes. Brominated organics in biology range from biologically produced methyl bromide to non-alkane aromatics and unsaturates (indoles, terpenes, acetogenins, and phenols). Halogenated alkanes in land plants are more rare, but do occur, as for example the fluoroacetate produced as a toxin by at least 40 species of known plants. Specific dehalogenase enzymes in bacteria which remove halogens from haloalkanes, are also known.