Iodine and Lipase Based Green Oxidation Technology
... Generally speaking, formation of waste during any process should be judged according to the E factor. 29, 30 Solvents that are less toxic, less harmful and that can be easily recovered and recycled are the desired ones. 1.1.4 Recovery and recycling Ease of recovery of reagents an ...
... Generally speaking, formation of waste during any process should be judged according to the E factor. 29, 30 Solvents that are less toxic, less harmful and that can be easily recovered and recycled are the desired ones. 1.1.4 Recovery and recycling Ease of recovery of reagents an ...
CHAPTER 22 ORGANIC AND BIOLOGICAL MOLECULES 1
... chain in the compound. This creates bond dipoles in the molecule leading to a polar molecule which exhibits additional dipole-dipole forces. Most of the functional groups have carbonoxygen polar bonds leading to a polar molecule. In halohydrocarbons, the polar bond is CX where X is a halogen. In am ...
... chain in the compound. This creates bond dipoles in the molecule leading to a polar molecule which exhibits additional dipole-dipole forces. Most of the functional groups have carbonoxygen polar bonds leading to a polar molecule. In halohydrocarbons, the polar bond is CX where X is a halogen. In am ...
Complete Solution Manual
... chain in the compound. This creates bond dipoles in the molecule leading to a polar molecule which exhibits additional dipole-dipole forces. Most of the functional groups have carbonoxygen polar bonds leading to a polar molecule. In halohydrocarbons, the polar bond is CX where X is a halogen. In am ...
... chain in the compound. This creates bond dipoles in the molecule leading to a polar molecule which exhibits additional dipole-dipole forces. Most of the functional groups have carbonoxygen polar bonds leading to a polar molecule. In halohydrocarbons, the polar bond is CX where X is a halogen. In am ...
an introduction to organic reactions
... By optional I mean that if you put the work in (some time before the night before a test) by reading the textbook before class, taking notes in Ziegler's (helpful) lectures, spending time working through the problem sets and going to your invaluable TA's at section, then you'll probably find orgo to ...
... By optional I mean that if you put the work in (some time before the night before a test) by reading the textbook before class, taking notes in Ziegler's (helpful) lectures, spending time working through the problem sets and going to your invaluable TA's at section, then you'll probably find orgo to ...
Efficient hydrogenation of organic carbonates, carbamates and
... he hydrogenation of polar bonds, in particular organic carbonyl groups, has captured much attention during the past four decades1–3, mainly due to its synthetic significance as an environmentally benign approach to fundamental synthetic building blocks such as alcohols and amines. Much progress has b ...
... he hydrogenation of polar bonds, in particular organic carbonyl groups, has captured much attention during the past four decades1–3, mainly due to its synthetic significance as an environmentally benign approach to fundamental synthetic building blocks such as alcohols and amines. Much progress has b ...
Esterification and Esters
... Use of Azeotropes to Remove Water. With the aliphatic alcohols and esters of medium volatility, a variety of azeotropes is encountered on distillation (see DISTILLATION, AZEOTROPIC AND EXTRACTIVE). Removal of these azeotropes from the esterification reaction mixture drives the equilibrium in favor o ...
... Use of Azeotropes to Remove Water. With the aliphatic alcohols and esters of medium volatility, a variety of azeotropes is encountered on distillation (see DISTILLATION, AZEOTROPIC AND EXTRACTIVE). Removal of these azeotropes from the esterification reaction mixture drives the equilibrium in favor o ...
doc
... The characteristics of organic compounds (boiling point, odour, reactivity etc.) depend on the composition and arrangement of atoms. For example the properties of alkanes depend greatly on the number of carbon atoms in the hydrocarbon chain due to the increased strength of the van der Waal attractio ...
... The characteristics of organic compounds (boiling point, odour, reactivity etc.) depend on the composition and arrangement of atoms. For example the properties of alkanes depend greatly on the number of carbon atoms in the hydrocarbon chain due to the increased strength of the van der Waal attractio ...
Chemistry.of Organic Compounds
... the students majoring in chemistry are preparing themselves for industrial positions. Hence the practical applications and economic aspects of organic chemistry frequently are discussed in some detail. For some years textbooks have been published which present aliphatic and aromatic compounds simult ...
... the students majoring in chemistry are preparing themselves for industrial positions. Hence the practical applications and economic aspects of organic chemistry frequently are discussed in some detail. For some years textbooks have been published which present aliphatic and aromatic compounds simult ...
CHAPTER TWENTY-TWO ORGANIC AND BIOLOGICAL MOLECULES
... chain in the compound. This creates bond dipoles in the molecule leading to a polar molecule which exhibits additional dipole-dipole forces. Most of the functional groups have carbonoxygen polar bonds leading to a polar molecule. In halohydrocarbons, the polar bond is C−X where X is a halogen. In am ...
... chain in the compound. This creates bond dipoles in the molecule leading to a polar molecule which exhibits additional dipole-dipole forces. Most of the functional groups have carbonoxygen polar bonds leading to a polar molecule. In halohydrocarbons, the polar bond is C−X where X is a halogen. In am ...
Elimination Reactions
... • E1 reactions occur under essentially neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 ...
... • E1 reactions occur under essentially neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 ...
chapter2
... (Alkanes are typically non-reactive. They don’t react with acids, bases, active metals, oxidizing agents, reducing agents, halogens, etc.) ...
... (Alkanes are typically non-reactive. They don’t react with acids, bases, active metals, oxidizing agents, reducing agents, halogens, etc.) ...
Chapter 7 Hydrosilylation of Carbon
... gen peroxide in the presence of a large excess of potassium fluoride and potassium bicarbonate gave (1S,2S,4R)-exo-2-norbornanol (23) with 93% ee in high yield. Lowering of the temperature to –20 ˚C raised the enantiomeric excess to 96% ee. A bicyclo[2.2.2]octene, a diester of norbornenedicarboxylic ...
... gen peroxide in the presence of a large excess of potassium fluoride and potassium bicarbonate gave (1S,2S,4R)-exo-2-norbornanol (23) with 93% ee in high yield. Lowering of the temperature to –20 ˚C raised the enantiomeric excess to 96% ee. A bicyclo[2.2.2]octene, a diester of norbornenedicarboxylic ...
chapter-15
... carbonyl group • for an aldehyde, change the suffix from -e to -al • for an unsaturated aldehyde, show the carbon-carbon double bond by changing the infix from -an- to -en-; the location of the suffix determines the numbering pattern • for a cyclic molecule in which -CHO is bonded to the ring, add t ...
... carbonyl group • for an aldehyde, change the suffix from -e to -al • for an unsaturated aldehyde, show the carbon-carbon double bond by changing the infix from -an- to -en-; the location of the suffix determines the numbering pattern • for a cyclic molecule in which -CHO is bonded to the ring, add t ...
Retrosynthetic Analysis of Nabumetone
... Grignard reactions that produce secondary alcohols, will require oxidation to the corresponding ketone to arrive at the desired product. Starting with the bond between the ring and the side chain, we are given two pairs of synthons as shown in Scheme 2. The synthetic equivalent to the aromatic anion ...
... Grignard reactions that produce secondary alcohols, will require oxidation to the corresponding ketone to arrive at the desired product. Starting with the bond between the ring and the side chain, we are given two pairs of synthons as shown in Scheme 2. The synthetic equivalent to the aromatic anion ...
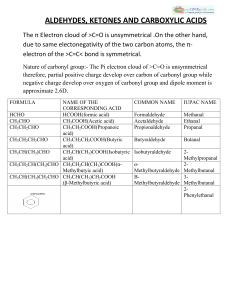
ALDEHYDES, KETONES AND CARBOXYLIC ACIDS
... :-Aldehydes, Ketones and Carboxylic acids are important classes of organic compounds containing carbonyl groups. :-They are highly polar molecules. :-They boil at higher temperatures than the corresponding hydrocarbons and weakly polar compounds such as ethers. :-Lower members are soluble in water b ...
... :-Aldehydes, Ketones and Carboxylic acids are important classes of organic compounds containing carbonyl groups. :-They are highly polar molecules. :-They boil at higher temperatures than the corresponding hydrocarbons and weakly polar compounds such as ethers. :-Lower members are soluble in water b ...
R - Evans - Harvard University
... To assay the inherent diastereoselection of the different bromination methods, we employed hydrocinnamyl carboximide l a (R = Bn) as the substrate. A variety of brominating agents and metal enolates were screened.’* The halogenation method that provided the highest diastereoselectivity was the react ...
... To assay the inherent diastereoselection of the different bromination methods, we employed hydrocinnamyl carboximide l a (R = Bn) as the substrate. A variety of brominating agents and metal enolates were screened.’* The halogenation method that provided the highest diastereoselectivity was the react ...
An efficient acetylation of dextran using in situ activated acetic
... Regarding the esterification of polysaccharides, a few methods are reported in the literature, such as using acetyl chloride with pyridine,14 acetic anhydride with pyridine and 4-(dimethylamino)pyridine,15,16 carboxylic acids in situ activated with tosyl chloride,17–19 iminium chloride20 and 1,1’-ca ...
... Regarding the esterification of polysaccharides, a few methods are reported in the literature, such as using acetyl chloride with pyridine,14 acetic anhydride with pyridine and 4-(dimethylamino)pyridine,15,16 carboxylic acids in situ activated with tosyl chloride,17–19 iminium chloride20 and 1,1’-ca ...
stoker-C15
... (1) The compound 4-oxopentanal contains both an aldehyde and an ether functional group. (2) An alkoxy group and a hydroxy group attached to the same carbon atom are present in both hemiacetals and acetals. (3) Cyclic aldehyde structures are possible but cyclic ketone structures are not possible. A) ...
... (1) The compound 4-oxopentanal contains both an aldehyde and an ether functional group. (2) An alkoxy group and a hydroxy group attached to the same carbon atom are present in both hemiacetals and acetals. (3) Cyclic aldehyde structures are possible but cyclic ketone structures are not possible. A) ...
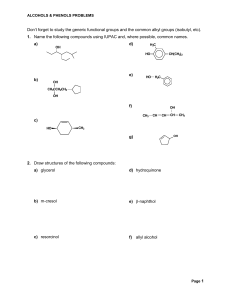
Don`t forget to study the generic functional groups and the common
... 11. Write equations to show how the following transformation can be carried out. More than one step may be necessary. There are marks assigned for each intermediate product (not charged transition states) and there are marks for the reagents used; so list them all. No marks are assigned for mechanis ...
... 11. Write equations to show how the following transformation can be carried out. More than one step may be necessary. There are marks assigned for each intermediate product (not charged transition states) and there are marks for the reagents used; so list them all. No marks are assigned for mechanis ...
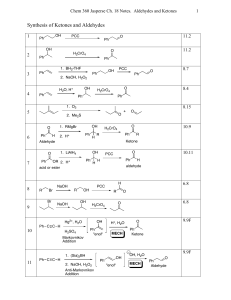
Synthesis of Ketones and Aldehydes
... Classification of Mechanisms Associated With Ketone/Aldehyde Reactions. • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict wh ...
... Classification of Mechanisms Associated With Ketone/Aldehyde Reactions. • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict wh ...
Module I Oxidation Reactions
... MnO2 is a useful selective oxidizing reagent in organic synthesis. It is commercially available, and it can also be prepared by the reaction of MnSO4∙4H2O with KMnO4 in aqueous NaOH. G. Cahiez, M. Alami, Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons, Inc., L. A. Paquette Ed., N ...
... MnO2 is a useful selective oxidizing reagent in organic synthesis. It is commercially available, and it can also be prepared by the reaction of MnSO4∙4H2O with KMnO4 in aqueous NaOH. G. Cahiez, M. Alami, Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons, Inc., L. A. Paquette Ed., N ...
Synthesis of Ketones and Aldehydes
... Classification of Mechanisms Associated With Ketone/Aldehyde Reactions. • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict wh ...
... Classification of Mechanisms Associated With Ketone/Aldehyde Reactions. • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict wh ...
chemistry - Textbooks Online
... Chemistry, a branch of science concerned with the properties, structures and composition of substances and their reactions with one another. Inorganic Chemistry studies the preparation, properties and reactions of all chemical elements and their compounds, except those of carbon. Organic Chemistry s ...
... Chemistry, a branch of science concerned with the properties, structures and composition of substances and their reactions with one another. Inorganic Chemistry studies the preparation, properties and reactions of all chemical elements and their compounds, except those of carbon. Organic Chemistry s ...
Transition Metal Reagents and Catalysts
... important types of reactions classi®ed mainly by representative substrates such as organic halides and allylic derivatives are surveyed with pertinent examples. For this purpose, I cited many references; these were selected from a much larger number which I have collected over the years. I wanted to ...
... important types of reactions classi®ed mainly by representative substrates such as organic halides and allylic derivatives are surveyed with pertinent examples. For this purpose, I cited many references; these were selected from a much larger number which I have collected over the years. I wanted to ...
Haloalkane

The haloalkanes (also known, as halogenoalkanes or alkyl halides) are a group of chemical compounds derived from alkanes containing one or more halogens. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially and, consequently, are known under many chemical and commercial names. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes which contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone layer depleter. For more information, see Halomethane. Haloalkane or alkyl halides are the compounds which have the general formula ″RX″ where R is an alkyl or substituted alkyl group and X is a halogen (F, Cl, Br, I).Haloalkanes have been known for centuries. Chloroethane was produced synthetically in the 15th century. The systematic synthesis of such compounds developed in the 19th century in step with the development of organic chemistry and the understanding of the structure of alkanes. Methods were developed for the selective formation of C-halogen bonds. Especially versatile methods included the addition of halogens to alkenes, hydrohalogenation of alkenes, and the conversion of alcohols to alkyl halides. These methods are so reliable and so easily implemented that haloalkanes became cheaply available for use in industrial chemistry because the halide could be further replaced by other functional groups.While most haloalkanes are human-produced, non-artificial-source haloalkanes do occur on Earth, mostly through enzyme-mediated synthesis by bacteria, fungi, and especially sea macroalgae (seaweeds). More than 1600 halogenated organics have been identified, with bromoalkanes being the most common haloalkanes. Brominated organics in biology range from biologically produced methyl bromide to non-alkane aromatics and unsaturates (indoles, terpenes, acetogenins, and phenols). Halogenated alkanes in land plants are more rare, but do occur, as for example the fluoroacetate produced as a toxin by at least 40 species of known plants. Specific dehalogenase enzymes in bacteria which remove halogens from haloalkanes, are also known.