Carbonyl Condensation Reactions
... carbonyl compound becomes the nucleophilic enolate and which reacts at the electrophilic carbonyl carbon. The strategy of a directed aldol reaction is as follows: [1] Prepare the enolate of one carbonyl component with LDA. [2] Add the second carbonyl compound (the electrophile) to this enolate. Beca ...
... carbonyl compound becomes the nucleophilic enolate and which reacts at the electrophilic carbonyl carbon. The strategy of a directed aldol reaction is as follows: [1] Prepare the enolate of one carbonyl component with LDA. [2] Add the second carbonyl compound (the electrophile) to this enolate. Beca ...
Recent developments in the applications of palladium complexes
... benzimidazolium salts performed better than the isolated Pd(II) complexes. Moreover, better catalytic activities were achieved with benzimidazolium salts with longer oligoether spacers. The potential reciclability of the Pd(OAc)2/5b system was explored in a model reaction. The final products were ex ...
... benzimidazolium salts performed better than the isolated Pd(II) complexes. Moreover, better catalytic activities were achieved with benzimidazolium salts with longer oligoether spacers. The potential reciclability of the Pd(OAc)2/5b system was explored in a model reaction. The final products were ex ...
Chem E2b - Organic Chemistry II What is Organic Chemistry?
... Functional group names and nomenclature Stability of carbocation and anion intermediates Resonance Curved arrow mechanism, identifying electrophiles and nucleophiles Acidity and Basicity Kinetics and thermodynamics: reaction coordinate diagrams Relative Reactivity Stereochemistry Reagents and Reacti ...
... Functional group names and nomenclature Stability of carbocation and anion intermediates Resonance Curved arrow mechanism, identifying electrophiles and nucleophiles Acidity and Basicity Kinetics and thermodynamics: reaction coordinate diagrams Relative Reactivity Stereochemistry Reagents and Reacti ...
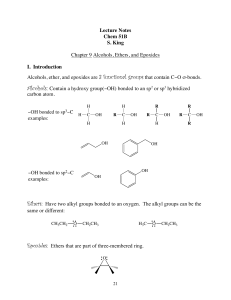
Lecture Notes Chem 51B S. King Chapter 9 Alcohols, Ethers, and
... We have seen that alcohols can be converted to alkyl halides by treating them with HCl, HBr, or HI. Better yields can be obtained , and carbocation rearrangements can be avoided if a phosphorus trihalide (PCl3, PBr3, or PI3), or thionyl chloride (SOCl2) is used instead. These act the same way: they ...
... We have seen that alcohols can be converted to alkyl halides by treating them with HCl, HBr, or HI. Better yields can be obtained , and carbocation rearrangements can be avoided if a phosphorus trihalide (PCl3, PBr3, or PI3), or thionyl chloride (SOCl2) is used instead. These act the same way: they ...
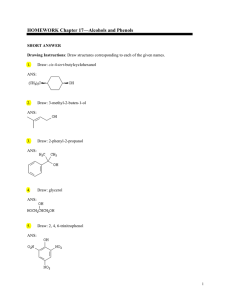
HOMEWORK Chapter 17—Alcohols and Phenols
... In E2 elimination, dehydration proceeds most readily when the two groups to be eliminated have a trans-diaxial relationship. In this compound, the only hydrogen with the proper geometric relationship to the −OH group is at C6 so the major product of this reaction is 3methylcyclohexene. ...
... In E2 elimination, dehydration proceeds most readily when the two groups to be eliminated have a trans-diaxial relationship. In this compound, the only hydrogen with the proper geometric relationship to the −OH group is at C6 so the major product of this reaction is 3methylcyclohexene. ...
Palladium and Ruthenium Catalyzed Reactions By Bryan Jaksic
... provide a simple method for the formation of substituted alkynes, a commonly found functionality within important organic molecules. These reactions are generally believed to be catalyzed by a Pd(0)L2 species which are generated in situ from a palladium precatalyst and are often co-catalyzed by CuI ...
... provide a simple method for the formation of substituted alkynes, a commonly found functionality within important organic molecules. These reactions are generally believed to be catalyzed by a Pd(0)L2 species which are generated in situ from a palladium precatalyst and are often co-catalyzed by CuI ...
Chapter - FIU Faculty Websites
... Aldehydes and Ketones—Acetals as Protecting Groups • Acetals are valuable protecting groups for aldehydes and ketones: • Suppose we wish to selectively reduce the ester group in compound A to an alcohol to give compound B, leaving the ketone untouched. • Because ketones are more readily reduced than ...
... Aldehydes and Ketones—Acetals as Protecting Groups • Acetals are valuable protecting groups for aldehydes and ketones: • Suppose we wish to selectively reduce the ester group in compound A to an alcohol to give compound B, leaving the ketone untouched. • Because ketones are more readily reduced than ...
New Applications for Sulfur-Based Leaving Groups in Synthesis
... past four years. It has been great fun to be able to work on a varied range of chemistry within his group and his stress-free approach to the subject has allowed me to form many of my own ideas and not be afraid to suggest them, no matter how absurd they seem. Towards the end of my PhD, EPR spectros ...
... past four years. It has been great fun to be able to work on a varied range of chemistry within his group and his stress-free approach to the subject has allowed me to form many of my own ideas and not be afraid to suggest them, no matter how absurd they seem. Towards the end of my PhD, EPR spectros ...
Title Several Reactions of Isocyanide and Related Compounds
... LIST OF PUBLISHED PAPERS . . . . • • . . . . . . . . . . . . . . . • . . ...
... LIST OF PUBLISHED PAPERS . . . . • • . . . . . . . . . . . . . . . • . . ...
Unit-7-Carboxylic-Acids-Phenols-Amines
... carbons that is attached to the nitrogen. • Replace the “-e” in the parent with the ending “-amine”. • Number the longest chain from the end that gets you to the the carbon to which the nitrogen is attached in the fewest number of carbons. • Use a numbered prefix to indicate which the carbon in the ...
... carbons that is attached to the nitrogen. • Replace the “-e” in the parent with the ending “-amine”. • Number the longest chain from the end that gets you to the the carbon to which the nitrogen is attached in the fewest number of carbons. • Use a numbered prefix to indicate which the carbon in the ...
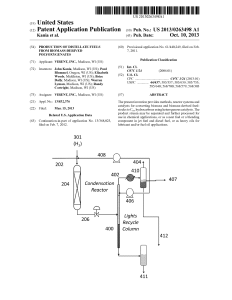
Production of Distillate Fuels from Biomass
... gas composed primarily of hydrogen and carbon monoxide, also called syngas or biosyngas. Syngas produced today is ...
... gas composed primarily of hydrogen and carbon monoxide, also called syngas or biosyngas. Syngas produced today is ...
Reactions of Alkenes
... Limited to Cl2 and Br2 F2 addition proceeds with explosive violence I2 addition is endothermic: vicinal diiodides dissociate to an alkene and I2 ...
... Limited to Cl2 and Br2 F2 addition proceeds with explosive violence I2 addition is endothermic: vicinal diiodides dissociate to an alkene and I2 ...
CH 2 CH(CH 3 ) - Parkway C-2
... Always include both the double and triple bond in the longest chain – even if it isn’t the most number of carbons! Start counting from the end closest to the double or triple bond – whichever has the lowest number If there is a tie, and only then, do double bonds take priority over triple ...
... Always include both the double and triple bond in the longest chain – even if it isn’t the most number of carbons! Start counting from the end closest to the double or triple bond – whichever has the lowest number If there is a tie, and only then, do double bonds take priority over triple ...
Polymerization Synthesis of Nylon 6,10 C11-5
... Making nylon is even easier if you use a diamine and a diacid chloride instead of a diacid. This is because acid chlorides are much more reactive than acids. The reaction is done in a two-phase system. The amine is dissolved in water, and the diacid chloride in an organic solvent. The two solutions ...
... Making nylon is even easier if you use a diamine and a diacid chloride instead of a diacid. This is because acid chlorides are much more reactive than acids. The reaction is done in a two-phase system. The amine is dissolved in water, and the diacid chloride in an organic solvent. The two solutions ...
Melt Modification of Poly(styrene-co-maleic anhydride)
... esterification to the product side by removing the in situ generated carboxylic acid from the system by a subsequent reaction. For example, 1,3-oxazolines are well known to afford quantitative conversion of carboxylic acids both in melt and in solution processes.16 Because of their high reactivity, ...
... esterification to the product side by removing the in situ generated carboxylic acid from the system by a subsequent reaction. For example, 1,3-oxazolines are well known to afford quantitative conversion of carboxylic acids both in melt and in solution processes.16 Because of their high reactivity, ...
View/Open
... As a consequence of these reactivity differences, it’s usually possible to convert a more reactive acid derivative into a less reactive one. Acid chlorides, for instance, can be directly converted into anhydrides, thioesters, esters, and amides, but amides can’t be directly converted into esters, th ...
... As a consequence of these reactivity differences, it’s usually possible to convert a more reactive acid derivative into a less reactive one. Acid chlorides, for instance, can be directly converted into anhydrides, thioesters, esters, and amides, but amides can’t be directly converted into esters, th ...
PDF carboxylic acids
... The simplest carboxylic acid is formic acid (HCOOH). Its name has been derived form the latin word formica which means ant. This acid was first obtained by distillation of ants and hence named as formic acid, which is its common name. Similarly acetic acid (CH3COOH) was so named as it gives sour tas ...
... The simplest carboxylic acid is formic acid (HCOOH). Its name has been derived form the latin word formica which means ant. This acid was first obtained by distillation of ants and hence named as formic acid, which is its common name. Similarly acetic acid (CH3COOH) was so named as it gives sour tas ...
synthetic approaches for quinoline and isoquinoline
... odour.Aged samples,if exposed to light, become yellow and later brown. Quinoline is only slightly soluble in cold water but dissolves readily in hot water and most organic solvents 1 Quinoline is mainly used as a building block to other specialty chemicals. Approximately ...
... odour.Aged samples,if exposed to light, become yellow and later brown. Quinoline is only slightly soluble in cold water but dissolves readily in hot water and most organic solvents 1 Quinoline is mainly used as a building block to other specialty chemicals. Approximately ...
15: Carbonyl Compounds: Esters, Amides, and Related Molecules
... Organic nitriles (R-C≡N) are usually considered together with R-C(=O)-Z compounds in organic chemistry textbooks. This is because nitriles (R-C≡N) are readily hydrolyzed to carboxylic acids (R-C(=O)-OH) via intermediate amides (R-C(=O)-NH2) (Figure 15.13). Figure 15.13 ...
... Organic nitriles (R-C≡N) are usually considered together with R-C(=O)-Z compounds in organic chemistry textbooks. This is because nitriles (R-C≡N) are readily hydrolyzed to carboxylic acids (R-C(=O)-OH) via intermediate amides (R-C(=O)-NH2) (Figure 15.13). Figure 15.13 ...
Aminoketone Rearrangements. 11. The Rearrangement of Phenyl a
... heated with methylamine a t 240°, 71% of the starting material was recovered with only a trace of basic material formed. The problem thus became the determination of the mechanism for the conversion of I I a to IIIa. The infrared spectra of the products obtained from the reaction of IIa with methyla ...
... heated with methylamine a t 240°, 71% of the starting material was recovered with only a trace of basic material formed. The problem thus became the determination of the mechanism for the conversion of I I a to IIIa. The infrared spectra of the products obtained from the reaction of IIa with methyla ...
Nickel Catalyzed Conversion of Cyclohexanol into Cyclohexylamine
... Amines find wide application in polymers, dyes, surfactants, pharmaceuticals, and biologically active compounds [1–10]. The amine is usually produced through direct amination of alcohol with ammonia. As shown in Scheme 1, this process includes dehydrogenation of alcohol to carbonyl compounds, aminat ...
... Amines find wide application in polymers, dyes, surfactants, pharmaceuticals, and biologically active compounds [1–10]. The amine is usually produced through direct amination of alcohol with ammonia. As shown in Scheme 1, this process includes dehydrogenation of alcohol to carbonyl compounds, aminat ...
15: Carbonyl Compounds: Esters, Amides, and Related Molecules
... oxygen. When the R' group is trans to O, the N-R' group is cis to the R group on the C=O and this is generally less sterically desirable than when R' is cis to O and trans to R. In spite of the 75 to 85 kJ/mol rotational barriers for interconversion of these geometric isomers, we cannot isolate the ...
... oxygen. When the R' group is trans to O, the N-R' group is cis to the R group on the C=O and this is generally less sterically desirable than when R' is cis to O and trans to R. In spite of the 75 to 85 kJ/mol rotational barriers for interconversion of these geometric isomers, we cannot isolate the ...
Haloalkane

The haloalkanes (also known, as halogenoalkanes or alkyl halides) are a group of chemical compounds derived from alkanes containing one or more halogens. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially and, consequently, are known under many chemical and commercial names. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes which contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone layer depleter. For more information, see Halomethane. Haloalkane or alkyl halides are the compounds which have the general formula ″RX″ where R is an alkyl or substituted alkyl group and X is a halogen (F, Cl, Br, I).Haloalkanes have been known for centuries. Chloroethane was produced synthetically in the 15th century. The systematic synthesis of such compounds developed in the 19th century in step with the development of organic chemistry and the understanding of the structure of alkanes. Methods were developed for the selective formation of C-halogen bonds. Especially versatile methods included the addition of halogens to alkenes, hydrohalogenation of alkenes, and the conversion of alcohols to alkyl halides. These methods are so reliable and so easily implemented that haloalkanes became cheaply available for use in industrial chemistry because the halide could be further replaced by other functional groups.While most haloalkanes are human-produced, non-artificial-source haloalkanes do occur on Earth, mostly through enzyme-mediated synthesis by bacteria, fungi, and especially sea macroalgae (seaweeds). More than 1600 halogenated organics have been identified, with bromoalkanes being the most common haloalkanes. Brominated organics in biology range from biologically produced methyl bromide to non-alkane aromatics and unsaturates (indoles, terpenes, acetogenins, and phenols). Halogenated alkanes in land plants are more rare, but do occur, as for example the fluoroacetate produced as a toxin by at least 40 species of known plants. Specific dehalogenase enzymes in bacteria which remove halogens from haloalkanes, are also known.