IUPAC Provisional Recommendations
... P-83.2.1.1 A selectively labeled compound cannot be described by a unique structural formula; therefore it is represented by inserting the nuclide symbol(s) preceded by any necessary locant(s) (letters and/or numbers) but without multiplying subscripts, enclosed in square brackets, [ ], directly bef ...
... P-83.2.1.1 A selectively labeled compound cannot be described by a unique structural formula; therefore it is represented by inserting the nuclide symbol(s) preceded by any necessary locant(s) (letters and/or numbers) but without multiplying subscripts, enclosed in square brackets, [ ], directly bef ...
VUV photochemistry of small biomolecules
... ion pair formation which can occur below and beyond the first IE. For a detailed review of these processes see Berry and Leach (1981). They can be explored by numerous experimental techniques, such as photoabsorption spectroscopy, photoion mass spectrometry (PIMS), photofragment fluorescence spectrosc ...
... ion pair formation which can occur below and beyond the first IE. For a detailed review of these processes see Berry and Leach (1981). They can be explored by numerous experimental techniques, such as photoabsorption spectroscopy, photoion mass spectrometry (PIMS), photofragment fluorescence spectrosc ...
File
... same molecular formula, but different structures, is called isomerism • Theses structures are called isomers ...
... same molecular formula, but different structures, is called isomerism • Theses structures are called isomers ...
An Introduction to Functional Groups in Organic Chemistry What are
... An alkane which contains a ring made up of three or more atoms is called a cycloalkane. Likewise, a cycloalkene contains a carbon-carbon double bond somewhere in a ring of carbon atoms. Cycloalkynes are relatively rare, for reasons we will see later (think bond angles). We will not consider these as ...
... An alkane which contains a ring made up of three or more atoms is called a cycloalkane. Likewise, a cycloalkene contains a carbon-carbon double bond somewhere in a ring of carbon atoms. Cycloalkynes are relatively rare, for reasons we will see later (think bond angles). We will not consider these as ...
IR Spectroscopy
... Principles of Spectroscopy Molecules can exist at various energy levels. For example, The bonds in a given molecule may stretch, bend, or rotate. electrons may move from one orbital to another. These processes are quantized; that is, bonds may stretch, bend or rotate only with certain frequencies, ...
... Principles of Spectroscopy Molecules can exist at various energy levels. For example, The bonds in a given molecule may stretch, bend, or rotate. electrons may move from one orbital to another. These processes are quantized; that is, bonds may stretch, bend or rotate only with certain frequencies, ...
Isomers
... This occurs most often around C=C The most common cases are around asymmetric non-cyclic alkenes ...
... This occurs most often around C=C The most common cases are around asymmetric non-cyclic alkenes ...
Assignment 30 STRUCTURE OF MOLECULES AND MULTI
... In lecture you have learned about atomic orbitals (AOs)—the regions of space surrounding an atom’s nucleus that ‘house’ that atom’s electrons. A carbon atom has four available atomic orbitals--one 2s AO, and three 2p AOs (2px, 2py, 2pz)— to house its four valence electrons. Take a look at the shapes ...
... In lecture you have learned about atomic orbitals (AOs)—the regions of space surrounding an atom’s nucleus that ‘house’ that atom’s electrons. A carbon atom has four available atomic orbitals--one 2s AO, and three 2p AOs (2px, 2py, 2pz)— to house its four valence electrons. Take a look at the shapes ...
Nuclear Magnetic Resonance: The Organic
... using a FT-NMR. They can be combined to give a better signal-tonoise ratio than possible using a CW-NMR for the same duration. As you can imagine, the output of a FT-NMR is complex and the data must be processed by a computer to generate the type of spectrum shown on the first pages of these notes. ...
... using a FT-NMR. They can be combined to give a better signal-tonoise ratio than possible using a CW-NMR for the same duration. As you can imagine, the output of a FT-NMR is complex and the data must be processed by a computer to generate the type of spectrum shown on the first pages of these notes. ...
CfE Advanced Higher Chemistry Unit 2: Organic
... molecular orbitals', occupy the region between two nuclei. The attraction of positive nuclei to negative electrons occupying bonding molecular orbitals is the basis of bonding between atoms. Each molecular orbital can accommodate a maximum of two electrons. If we look at the example of H 2 (g) one o ...
... molecular orbitals', occupy the region between two nuclei. The attraction of positive nuclei to negative electrons occupying bonding molecular orbitals is the basis of bonding between atoms. Each molecular orbital can accommodate a maximum of two electrons. If we look at the example of H 2 (g) one o ...
Chemdraw B&W - Chemistry Courses
... • Amides (RCONH2) in general are not proton acceptors except in very strong acid • The C=O group is strongly electron-withdrawing, making the N a very weak base • Addition of a proton occurs on O but this destroys the double bond character of C=O as a requirement of stabilization by N ...
... • Amides (RCONH2) in general are not proton acceptors except in very strong acid • The C=O group is strongly electron-withdrawing, making the N a very weak base • Addition of a proton occurs on O but this destroys the double bond character of C=O as a requirement of stabilization by N ...
Process for the manufacture of maleic acid anhydride copolymers
... unsaturated organic compounds are ole main included in the polymer particles after the manu 60 copolymerizable ?nically unsaturated aromatic hydrocarbon compounds facture of the copolymers often prove troublesome, so such as styrene and its homologs, such as methyl styrene that turbid aqueous soluti ...
... unsaturated organic compounds are ole main included in the polymer particles after the manu 60 copolymerizable ?nically unsaturated aromatic hydrocarbon compounds facture of the copolymers often prove troublesome, so such as styrene and its homologs, such as methyl styrene that turbid aqueous soluti ...
A Lesson on the Science and Art of Perfumery
... To understand just how these notes behave and why a certain perfume smells the way it does, we turn to some very simple chemistry. Evaporation is the process by which molecules in the liquid phase become gaseous. This phenomenon can be controlled by the attractive forces between the molecules in the ...
... To understand just how these notes behave and why a certain perfume smells the way it does, we turn to some very simple chemistry. Evaporation is the process by which molecules in the liquid phase become gaseous. This phenomenon can be controlled by the attractive forces between the molecules in the ...
An algorithm to identify functional groups in organic molecules
... 3. merge all connected marked atoms to a single FG 4. extract FGs also with connected unmarked carbon atoms, these carbon atoms are not part of the FG itself, but form its environment. The algorithm described above iterates only through non-aromatic atoms. Aromatic heteroatoms are collected as sing ...
... 3. merge all connected marked atoms to a single FG 4. extract FGs also with connected unmarked carbon atoms, these carbon atoms are not part of the FG itself, but form its environment. The algorithm described above iterates only through non-aromatic atoms. Aromatic heteroatoms are collected as sing ...
DAMIETTA UNIVERSITY
... and NH3 are roughly tetrahedral (104o and 107o respectively) and CH4 is exactly tetrahedral (109.5o) !!! Problem: Orbitals available for bonding are 2s () and 2p (right angles to each other) In order to account for the observed geometry, hybridization was proposed as a convenient model. Hybrid ...
... and NH3 are roughly tetrahedral (104o and 107o respectively) and CH4 is exactly tetrahedral (109.5o) !!! Problem: Orbitals available for bonding are 2s () and 2p (right angles to each other) In order to account for the observed geometry, hybridization was proposed as a convenient model. Hybrid ...
07. Aldehydes and ketones
... The alkoxide formed in the nucleophilic addition step then abstracts a proton from the solvent (usually water or ethanol) to yield the product of aldol addition. This product is known as an aldol because it contains both an aldehyde function and a hydroxyl group (ald+ol=aldol). An important feature ...
... The alkoxide formed in the nucleophilic addition step then abstracts a proton from the solvent (usually water or ethanol) to yield the product of aldol addition. This product is known as an aldol because it contains both an aldehyde function and a hydroxyl group (ald+ol=aldol). An important feature ...
Vorlesung Supramolekulare Chemie
... aromatic rings with one binding partner more electron rich, the other more electron poor. There are two possible orientations observed in this interaction: A face to face orientation and an edge to face orientation. In crystal structures of aromatic compounds a so called herringbone motif is sometim ...
... aromatic rings with one binding partner more electron rich, the other more electron poor. There are two possible orientations observed in this interaction: A face to face orientation and an edge to face orientation. In crystal structures of aromatic compounds a so called herringbone motif is sometim ...
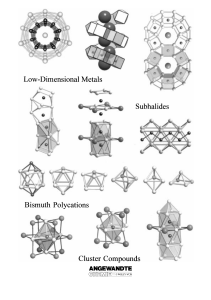
From the Metal to the Molecule
... electron-deficient s-, d-, and f-metals, but also for p-(semi-) metals albeit with far fewer examples. For example, recently significant progress has been made especially in research using the elements indium,[6] tin,[7] and bismuth.[8] This research mainly concerns the ternary and quaternary oxides ...
... electron-deficient s-, d-, and f-metals, but also for p-(semi-) metals albeit with far fewer examples. For example, recently significant progress has been made especially in research using the elements indium,[6] tin,[7] and bismuth.[8] This research mainly concerns the ternary and quaternary oxides ...
Microsoft Word - Open Access Repository of Indian Theses
... amine or the alcohol can be acylated, alkylated or contained within rings. The presence of this moiety and the relative (as well as absolute) stereochemistry are generally important for the biological activity of molecules containing a vicinal amino alcohol. The classical approach for the synthesis ...
... amine or the alcohol can be acylated, alkylated or contained within rings. The presence of this moiety and the relative (as well as absolute) stereochemistry are generally important for the biological activity of molecules containing a vicinal amino alcohol. The classical approach for the synthesis ...
EXPERIMENT 3 – Keto-Enol Equilibrium Using NMR
... in at least four different solvents (C6D6, C6D12, CD3CN, H2O/D2O, CDCl3, acetone-d6 and/or dimethyl sulfoxide-d6) at a concentration of ~1 mM. Prepare them at least 60 minutes in advance of running the proton NMR spectrum (to let the enol-keto ratio equilibrate). The solutions should be prepared dir ...
... in at least four different solvents (C6D6, C6D12, CD3CN, H2O/D2O, CDCl3, acetone-d6 and/or dimethyl sulfoxide-d6) at a concentration of ~1 mM. Prepare them at least 60 minutes in advance of running the proton NMR spectrum (to let the enol-keto ratio equilibrate). The solutions should be prepared dir ...
Exam 2
... Draw structural formulas for the following compounds. Give the molecular formula of each (CxHy.....). a) 2-methyl-3-hexene ...
... Draw structural formulas for the following compounds. Give the molecular formula of each (CxHy.....). a) 2-methyl-3-hexene ...
Document
... forms of ionizable groups depending on chemical characteristics and environmental pH (use the HH equation) 5. Apply in medical context ...
... forms of ionizable groups depending on chemical characteristics and environmental pH (use the HH equation) 5. Apply in medical context ...
Topic Selection Menu - Pennsylvania State University
... – Absorption of electromagnetic radiation UV-Vis range ...
... – Absorption of electromagnetic radiation UV-Vis range ...
Topic Selection Menu - Pennsylvania State University
... – Absorption of electromagnetic radiation UV-Vis range ...
... – Absorption of electromagnetic radiation UV-Vis range ...
Homoaromaticity

Homoaromaticity in organic chemistry refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thought to be a requirement for aromaticity, considerable thermodynamic stability and many of the spectroscopic, magnetic, and chemical properties associated with aromatic compounds are still observed for such compounds. This formal discontinuity is apparently bridged by p-orbital overlap, maintaining a contiguous cycle of π electrons that is responsible for this preserved chemical stability.The concept of homoaromaticity was pioneered by Saul Winstein in 1959, prompted by his studies of the “tris-homocyclopropenyl” cation. Since the publication of Winstein's paper, much research has been devoted to understanding and classifying these molecules, which represent an additional “class” of aromatic molecules included under the continuously broadening definition of aromaticity. To date, homoaromatic compounds are known to exist as cationic and anionic species, and some studies support the existence of neutral homoaromatic molecules, though these are less common. The 'homotropylium' cation (C8H9+) is perhaps the best studied example of a homoaromatic compound.