Study Guide and Solutions Manual
... t is our hope that in writing this Study Guide and Solutions Manual we will make the study of organic chemistry more meaningful and worthwhile. To be effective, a study guide should be more than just an answer book. What we present here was designed with that larger goal in mind. The Study Guide and ...
... t is our hope that in writing this Study Guide and Solutions Manual we will make the study of organic chemistry more meaningful and worthwhile. To be effective, a study guide should be more than just an answer book. What we present here was designed with that larger goal in mind. The Study Guide and ...
ORGANIC CHEMISTRY 4th ed Solution Manual
... t is our hope that in writing this Study Guide and Solutions Manual we will make the study of organic chemistry more meaningful and worthwhile. To be effective, a study guide should be more than just an answer book. What we present here was designed with that larger goal in mind. The Study Guide and ...
... t is our hope that in writing this Study Guide and Solutions Manual we will make the study of organic chemistry more meaningful and worthwhile. To be effective, a study guide should be more than just an answer book. What we present here was designed with that larger goal in mind. The Study Guide and ...
organic chemistry - carey - problems solutions
... Since the problem states that the atoms in C3H3N are connected in the order CCCN and all hydrogens are bonded to carbon, the order of attachments can only be as shown (below left) so as to have four bonds to each carbon. Three carbons contribute 12 valence electrons, three hydrogens contribute 3, an ...
... Since the problem states that the atoms in C3H3N are connected in the order CCCN and all hydrogens are bonded to carbon, the order of attachments can only be as shown (below left) so as to have four bonds to each carbon. Three carbons contribute 12 valence electrons, three hydrogens contribute 3, an ...
irm_ch17
... secondary amine used to prepare it would have two of these alkyl groups and the alkyl halide would contain the third. The three possible combinations are: ethylmethylamine and propyl chloride, ethylpropylamine and methyl chloride, and methylpropylamine and ethyl chloride. 17.50 butylethylamine, prop ...
... secondary amine used to prepare it would have two of these alkyl groups and the alkyl halide would contain the third. The three possible combinations are: ethylmethylamine and propyl chloride, ethylpropylamine and methyl chloride, and methylpropylamine and ethyl chloride. 17.50 butylethylamine, prop ...
On the composition of ammonia–sulfuric
... that the stabilizing effect of NH3 dominates for typical atmospheric conditions due to relatively low gas-phase amine concentrations (Nadykto et al., 2011). Indeed, a dominant role for NH3 is consistent with the observation that clusters during new-particle formation in the boreal forest contain mor ...
... that the stabilizing effect of NH3 dominates for typical atmospheric conditions due to relatively low gas-phase amine concentrations (Nadykto et al., 2011). Indeed, a dominant role for NH3 is consistent with the observation that clusters during new-particle formation in the boreal forest contain mor ...
Chapter15 odd probs
... a) Carbon’s electronegativity is midway between the most metallic and nonmetallic elements of period 2. To attain a filled outer shell, carbon forms covalent bonds to other atoms in molecules (e.g., methane, CH4), network covalent solids (e.g., diamond) and polyatomic ions (e.g., carbonate, CO32–). ...
... a) Carbon’s electronegativity is midway between the most metallic and nonmetallic elements of period 2. To attain a filled outer shell, carbon forms covalent bonds to other atoms in molecules (e.g., methane, CH4), network covalent solids (e.g., diamond) and polyatomic ions (e.g., carbonate, CO32–). ...
Organic Chemistry - Zanichelli online per la scuola
... Each chapter of the text is briefly summarized. Whenever pertinent, the chapter summary is followed by a list of all the new reactions and mechanisms encountered in that chapter. These lists should be especially helpful to you as you review for examinations. When you study a new subject, it is alway ...
... Each chapter of the text is briefly summarized. Whenever pertinent, the chapter summary is followed by a list of all the new reactions and mechanisms encountered in that chapter. These lists should be especially helpful to you as you review for examinations. When you study a new subject, it is alway ...
irm_ch15
... 15.59 When an alcohol molecule (R—O—H) adds across a carbon-oxygen double bond, the fragments of the alcohol are R–O– and H–. 15.60 H– 15.61 In a hemiacetal, a carbon atom is bonded to both a hydroxyl group (—OH) and an alkoxy group (—OR). a. No, this compound is an ether; there is no hydroxyl group ...
... 15.59 When an alcohol molecule (R—O—H) adds across a carbon-oxygen double bond, the fragments of the alcohol are R–O– and H–. 15.60 H– 15.61 In a hemiacetal, a carbon atom is bonded to both a hydroxyl group (—OH) and an alkoxy group (—OR). a. No, this compound is an ether; there is no hydroxyl group ...
Active Learning in Chemical Education
... CH 3 –CH 2 –O–CH 3 Functional group is "methoxy" In the IUPAC system, the larger of the two alkyl groups attached to the oxygen is considered to be the parent compound. For the ether mentioned in the last paragraph above, the parent compound would be ethane. The smaller alkyl group and the oxygen at ...
... CH 3 –CH 2 –O–CH 3 Functional group is "methoxy" In the IUPAC system, the larger of the two alkyl groups attached to the oxygen is considered to be the parent compound. For the ether mentioned in the last paragraph above, the parent compound would be ethane. The smaller alkyl group and the oxygen at ...
Alcohols, Phenols, Thiols, & Ethers
... 1-propanol would have the higher boiling point because an alcohol can form hydrogen bonds, but the ether cannot. ...
... 1-propanol would have the higher boiling point because an alcohol can form hydrogen bonds, but the ether cannot. ...
What is Organic Chemistry?
... that is, carbon has 2 electrons in the lowest energy level, the 1s orbital 2 electrons in the next energy level, the 2s orbital 2 electrons in the third energy level, the 2p orbital 1s, 2s, 2p orbitals 1s is spherical with the same phase throughout ...
... that is, carbon has 2 electrons in the lowest energy level, the 1s orbital 2 electrons in the next energy level, the 2s orbital 2 electrons in the third energy level, the 2p orbital 1s, 2s, 2p orbitals 1s is spherical with the same phase throughout ...
Chapter 5 | Molecular Orbitals
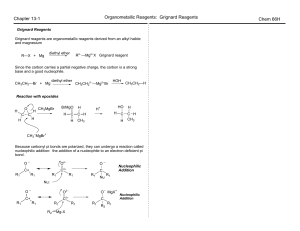
... Molecular orbital theory uses group theory to describe the bonding in molecules ; it complements and extends the introductory bonding models in Chapter 3 . In molecular orbital theory the symmetry properties and relative energies of atomic orbitals determine how these orb ...
... Molecular orbital theory uses group theory to describe the bonding in molecules ; it complements and extends the introductory bonding models in Chapter 3 . In molecular orbital theory the symmetry properties and relative energies of atomic orbitals determine how these orb ...
CH 2
... 1-propanol would have the higher boiling point because an alcohol can form hydrogen bonds, but the ether cannot. ...
... 1-propanol would have the higher boiling point because an alcohol can form hydrogen bonds, but the ether cannot. ...
PowerPoint - Naming Hydrocarbons
... double or triple bond is present, choose the longest chain that includes this bond. If there is a cyclic structure present, the longest chain starts and stops within the cyclic structure. 3. Assign numbers to each C of the parent chain. For alkenes and alkynes the first carbon of the multiple bond s ...
... double or triple bond is present, choose the longest chain that includes this bond. If there is a cyclic structure present, the longest chain starts and stops within the cyclic structure. 3. Assign numbers to each C of the parent chain. For alkenes and alkynes the first carbon of the multiple bond s ...
Alcohols, Ethers, and Epoxides
... glycol—are given in Figure 9.3. Ethanol (CH3CH2OH), formed by the fermentation of the carbohydrates in grains, grapes, and potatoes, is the alcohol present in alcoholic beverages. It is perhaps the first organic compound synthesized by humans, because alcohol production has been known for at least 4 ...
... glycol—are given in Figure 9.3. Ethanol (CH3CH2OH), formed by the fermentation of the carbohydrates in grains, grapes, and potatoes, is the alcohol present in alcoholic beverages. It is perhaps the first organic compound synthesized by humans, because alcohol production has been known for at least 4 ...
Infrared - ResearchGate
... the product of a reaction as a known compound. (require access to a file of standard spectra) • At another extreme , different bands observed can be used to deduce the symmetry of the molecule and force constants corresponding to vibrations. • At intermediate levels, deductions may be drawn about th ...
... the product of a reaction as a known compound. (require access to a file of standard spectra) • At another extreme , different bands observed can be used to deduce the symmetry of the molecule and force constants corresponding to vibrations. • At intermediate levels, deductions may be drawn about th ...
Alkenes 4 - ChemWeb (UCC)
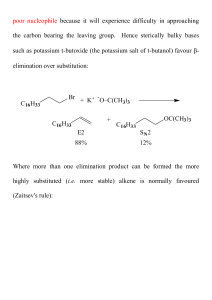
... Unlike the case of the E2 mechanism - there is no steric requirement for the conformation of the substrate in an E1 reaction. ...
... Unlike the case of the E2 mechanism - there is no steric requirement for the conformation of the substrate in an E1 reaction. ...
Alcohol, Ethers, and Thiols
... An alcohol contains a hydroxyl group (—OH) attached to a carbon chain. A phenol contains a hydroxyl group (—OH) attached to a ...
... An alcohol contains a hydroxyl group (—OH) attached to a carbon chain. A phenol contains a hydroxyl group (—OH) attached to a ...
Compounds with Oxygen Atoms
... An alcohol contains a hydroxyl group (—OH) attached to a carbon chain. A phenol contains a hydroxyl group (—OH) attached to a ...
... An alcohol contains a hydroxyl group (—OH) attached to a carbon chain. A phenol contains a hydroxyl group (—OH) attached to a ...
Course Notes
... Magnetic equivalence is usually the same as chemical equivalence. Equivalence can be established by symmetry operations such as rotation, mirror planes and centers of symmetry Chemically equivalent protons have the same chemical shifts. To determine if protons are chemically equivalent, replace one ...
... Magnetic equivalence is usually the same as chemical equivalence. Equivalence can be established by symmetry operations such as rotation, mirror planes and centers of symmetry Chemically equivalent protons have the same chemical shifts. To determine if protons are chemically equivalent, replace one ...
Chapter 13 Alcohols, Phenols, and Thiols
... • Such as ethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • Developed by 1960s were nonflammable. ...
... • Such as ethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • Developed by 1960s were nonflammable. ...
Document
... 4. More soluble in water than other hydrocarbons of similar molecular weight (H-bond with water). ...
... 4. More soluble in water than other hydrocarbons of similar molecular weight (H-bond with water). ...
Chapter 12 Organic Compounds with Oxygen and Sulfur
... Stereoisomers have identical molecular formulas, but they are not structural isomers. In stereoisomers, the atoms are bonded in the same sequence but differ in the way they are arranged in space. When stereoisomers have mirror images that are different, they are said to have “handedness.” ...
... Stereoisomers have identical molecular formulas, but they are not structural isomers. In stereoisomers, the atoms are bonded in the same sequence but differ in the way they are arranged in space. When stereoisomers have mirror images that are different, they are said to have “handedness.” ...
Lorell Thesis Final Version in PDF S
... Figure 22. 13C and 1H NMR of syn-(S,S)-90d. ........................................................ 68 Figure 23. 31P NMR of CDA derivative of 90d. ........................................................ 69 Figure 24. 13C and 1H NMR of syn-(R,R)-90e. ............................................... ...
... Figure 22. 13C and 1H NMR of syn-(S,S)-90d. ........................................................ 68 Figure 23. 31P NMR of CDA derivative of 90d. ........................................................ 69 Figure 24. 13C and 1H NMR of syn-(R,R)-90e. ............................................... ...
an introduction to organic reactions
... Most of the statements you've read so far on this page have probably started out by saying that Organic Chemistry really isn't that bad and can, in fact, be pretty interesting. I think it's important to understand from the start that this is completely true…I can almost assure you that you will enjo ...
... Most of the statements you've read so far on this page have probably started out by saying that Organic Chemistry really isn't that bad and can, in fact, be pretty interesting. I think it's important to understand from the start that this is completely true…I can almost assure you that you will enjo ...
Homoaromaticity

Homoaromaticity in organic chemistry refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thought to be a requirement for aromaticity, considerable thermodynamic stability and many of the spectroscopic, magnetic, and chemical properties associated with aromatic compounds are still observed for such compounds. This formal discontinuity is apparently bridged by p-orbital overlap, maintaining a contiguous cycle of π electrons that is responsible for this preserved chemical stability.The concept of homoaromaticity was pioneered by Saul Winstein in 1959, prompted by his studies of the “tris-homocyclopropenyl” cation. Since the publication of Winstein's paper, much research has been devoted to understanding and classifying these molecules, which represent an additional “class” of aromatic molecules included under the continuously broadening definition of aromaticity. To date, homoaromatic compounds are known to exist as cationic and anionic species, and some studies support the existence of neutral homoaromatic molecules, though these are less common. The 'homotropylium' cation (C8H9+) is perhaps the best studied example of a homoaromatic compound.