* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Compounds with Oxygen Atoms
Survey
Document related concepts
Transcript
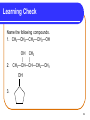
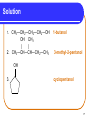
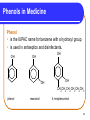
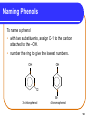
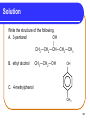
Chapter 12 Organic Compounds with Oxygen and Sulfur 12.1 Alcohols, Ethers, and Thiols Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 1 Alcohols An alcohol contains a hydroxyl group (—OH) attached to a carbon chain. A phenol contains a hydroxyl group (—OH) attached to a benzene ring. water alcohol phenol Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 2 Naming Alcohols The names of alcohols • in IUPAC replace the -e with -ol. • with common names use the name of the alkyl group followed by alcohol. Formula IUPAC CH4 methane CH3─OH methanol CH3─CH3 ethane CH3─CH2─OH ethanol Common Name methyl alcohol ethyl alcohol 3 More Names of Alcohols IUPAC names for longer chains number the chain from the end nearest the -OH group. CH3─CH2─CH2─OH 1-propanol OH │ CH3─CH─CH2─CH3 2-butanol CH3 OH │ │ CH3─CH─CH2─CH2─CH─CH3 6 5 4 3 2 1 5-methyl-2-hexanol 4 Some Typical Alcohols OH | “rubbing alcohol” CH3—CH—CH3 2-propanol (isopropyl alcohol) antifreeze HO—CH2—CH2—OH 1,2-ethanediol (ethylene glycol) OH | glycerol HO—CH2—CH—CH2—OH 1,2,3-propanetriol 5 Learning Check Name the following compounds. 1. CH3—CH2—CH2—CH2—OH 2. OH CH3 | | CH3—CH—CH—CH2—CH3 OH 3. 6 Solution 1. CH3—CH2—CH2—CH2—OH OH CH3 | | 2. CH3—CH—CH—CH2—CH3 1-butanol 3-methyl-2-pentanol OH 3. cyclopentanol 7 Phenols in Medicine Phenol • is the IUPAC name for benzene with a hydroxyl group. • is used in antiseptics and disinfectants. OH OH OH OH OH CH2CH2CH2CH2CH2CH3 phenol resorcinol 4-hexylresorcinol 8 Derivatives of Phenol Compounds of phenol are the active ingredients in the essential oils of cloves, vanilla, nutmeg, and mint. 9 Naming Phenols To name a phenol • with two substituents, assign C-1 to the carbon attached to the –OH. • number the ring to give the lowest numbers. OH 1 OH 1 3 Cl 3-chlorophenol 4 Br 4-bromophenol 10 Learning Check Write the structure of each of the following. A. 3-pentanol B. ethyl alcohol C. 4-methylphenol 11 Solution Write the structure of the following. A. 3-pentanol OH | CH3—CH2—CH—CH2—CH3 B. ethyl alcohol CH3—CH2—OH OH C. 4-methylphenol CH3 12 Classification of Alcohols Classification of alcohols is • determined by the number of alkyl groups attached to the carbon bonded to the hydroxyl. • primary (1°), secondary (2°), or tertiary(3). Primary (1º) 1 group H | CH3—C—OH | H Secondary (2º) Tertiary (3º) 2 groups 3 groups CH3 | CH3—C—OH | H CH3 | CH3—C—OH | CH3 13 Learning Check Classify each alcohol as 1) primary, 2) secondary, or 3) tertiary. OH | A. CH3—CH—CH2—CH3 B. CH3—CH2—CH2—OH OH | C. CH3—CH2—C—CH2—CH3 | CH3 14 Solution OH | A. CH3—CH—CH2—CH3 secondary B. CH3—CH2—CH2—OH primary OH | C. CH3—CH2—C—CH2—CH3 | CH3 tertiary 15 Thiols Thiols • are carbon compounds that contain a –SH group. • are named in the IUPAC system by adding thiol to the alkane name of the longest carbon chain. Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 16 Thiols Thiols • often have strong odors. • are used to detect gas leaks. • are found in onions, oysters, and garlic. 17 Ethers An ether • contains an ─O─ between two carbon groups. • has a common name that gives the alkyl names of the attached groups followed by ether. CH3─O─CH3 CH3─CH2─O─CH3 dimethyl ether ethyl methyl ether 18 Learning Check Write the structure of the following. A. 3-pentanol B. diethyl ether 19 Solution A. 3-pentanol B. diethyl ether OH | CH3─CH2─CH─CH2─CH3 CH3─CH2─O─CH2─CH3 20 Ethers as Anesthetics Anesthetics • inhibit pain signals to the brain. • like diethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • developed by the 1960’s were nonflammable. Cl F F Cl F H │ │ │ │ │ │ H─C─C─O─C─H H─C─C─O─C─H │ │ │ │ │ │ F F F H F H Ethane(enflurane) Penthrane 21 MTBE Methyl tert-butyl ether CH3 │ CH3─O─C─CH3 │ CH3 • is second in production of organic chemicals. • is an additive used to improve gasoline performance. • use is questioned since the discovery that MTBE has contaminated water supplies. 22