Joanna Kulesza
... Je tiens à exprimer ici la mémoire de mes anciennes amies: Anna Bresińska, Dominika Kozłowska, Anna Budzisz et Justyna Piotrowska. ...
... Je tiens à exprimer ici la mémoire de mes anciennes amies: Anna Bresińska, Dominika Kozłowska, Anna Budzisz et Justyna Piotrowska. ...
LINALOOL BIOTRANSFORMATION WITH FUNGI
... pathway we hypothesized that there are also other biological systems capable of transforming linalool into the desired lilac aroma compounds. Microorganisms, especially fungi, have been shown to be very versatile biocatalysts for the production of a wide range of flavour and fragrance compounds from ...
... pathway we hypothesized that there are also other biological systems capable of transforming linalool into the desired lilac aroma compounds. Microorganisms, especially fungi, have been shown to be very versatile biocatalysts for the production of a wide range of flavour and fragrance compounds from ...
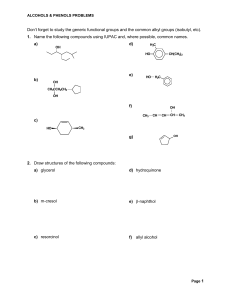
Don`t forget to study the generic functional groups and the common
... 11. Write equations to show how the following transformation can be carried out. More than one step may be necessary. There are marks assigned for each intermediate product (not charged transition states) and there are marks for the reagents used; so list them all. No marks are assigned for mechanis ...
... 11. Write equations to show how the following transformation can be carried out. More than one step may be necessary. There are marks assigned for each intermediate product (not charged transition states) and there are marks for the reagents used; so list them all. No marks are assigned for mechanis ...
(NH3)n and NH2 - Sanov Group
... Solvation of negative ions may be systematically examined via experiments on a series of clusters X− (M)n, where n neutral molecules (M) are electrostatically (as opposed to covalently) bound to a negative ion (X− ).12, 13 For such species, solvation is often thought of as having a perturbative effe ...
... Solvation of negative ions may be systematically examined via experiments on a series of clusters X− (M)n, where n neutral molecules (M) are electrostatically (as opposed to covalently) bound to a negative ion (X− ).12, 13 For such species, solvation is often thought of as having a perturbative effe ...
Introduction - St. Olaf College
... that atom positions are represented by small spheres, making it easy to identify atom locations. The tube model is similar to the wire model, except that bonds are represented by solid cylinders. The tube model is better than the wire model in conveying three-dimensional shape. The ball-and-spoke mo ...
... that atom positions are represented by small spheres, making it easy to identify atom locations. The tube model is similar to the wire model, except that bonds are represented by solid cylinders. The tube model is better than the wire model in conveying three-dimensional shape. The ball-and-spoke mo ...
IUPAC System of Nomenclature
... lower of two possible numbers. Also, the location number should be given as to where the double bond is (except ethene or propene, where the location will always be 1). The alkenes all end in -ene. For example: CH2 CHCH2 CH3 ...
... lower of two possible numbers. Also, the location number should be given as to where the double bond is (except ethene or propene, where the location will always be 1). The alkenes all end in -ene. For example: CH2 CHCH2 CH3 ...
Edita Pusvaškienė
... Since the carbon nanotube coating layer was not uniform (was thicker in the middle of the groove and thinner at the edges) the extraction of the less volatile compounds could be problematic because of their low sorption/desorption from the thicker coating layer and could cause peak tailing because ...
... Since the carbon nanotube coating layer was not uniform (was thicker in the middle of the groove and thinner at the edges) the extraction of the less volatile compounds could be problematic because of their low sorption/desorption from the thicker coating layer and could cause peak tailing because ...
Ionic Liquids in Separation of Metal Ions from Aqueous
... the recovery of Ce4+ from bastnasite leaching liquor, containing Th4+ and rare earth metals (RE), is limited by the presence of F- that negatively affects extraction efficiency. Because of this, neutral extractant (DEHEHP) has been added to [C8mim][PF6] to overcome this problem and the extraction ef ...
... the recovery of Ce4+ from bastnasite leaching liquor, containing Th4+ and rare earth metals (RE), is limited by the presence of F- that negatively affects extraction efficiency. Because of this, neutral extractant (DEHEHP) has been added to [C8mim][PF6] to overcome this problem and the extraction ef ...
As we proceed through organic chemistry we will often focus our
... chain as C1. Starting at this position one would count the longest substituent chain possible, as shown below. iii. Parentheses are used to separate the entire complex substituent name, its numbers, its branches, and its subparent name, from the principle parent name. A number and a hyphen precede t ...
... chain as C1. Starting at this position one would count the longest substituent chain possible, as shown below. iii. Parentheses are used to separate the entire complex substituent name, its numbers, its branches, and its subparent name, from the principle parent name. A number and a hyphen precede t ...
mcdonald (pam78654) – HW 1: High School Concepts – laude
... mcdonald (pam78654) – HW 1: High School Concepts – laude – (89560) This print-out should have 40 questions. Multiple-choice questions may continue on the next column or page – find all choices before answering. 001 10.0 points Calculate the number of H2 O molecules in 1.00 cm3 of water at 0◦ C (dens ...
... mcdonald (pam78654) – HW 1: High School Concepts – laude – (89560) This print-out should have 40 questions. Multiple-choice questions may continue on the next column or page – find all choices before answering. 001 10.0 points Calculate the number of H2 O molecules in 1.00 cm3 of water at 0◦ C (dens ...
GENERAL INTRODUCTION TO THE CHEMISTRY OF DYES 1
... the visible spectrum (400–700 nm), 2) have at least one chromophore (colour-bearing group), 3) have a conjugated system, i.e. a structure with alternating double and single bonds, and 4) exhibit resonance of electrons, which is a stabilizing force in organic compounds (Abrahart, 1977). When any one ...
... the visible spectrum (400–700 nm), 2) have at least one chromophore (colour-bearing group), 3) have a conjugated system, i.e. a structure with alternating double and single bonds, and 4) exhibit resonance of electrons, which is a stabilizing force in organic compounds (Abrahart, 1977). When any one ...
Organic handouts - Moore Chemistry
... 8-carbons octane Cycloalkanes These hydrocarbons exist in ring structures with general formula CnH2n. There is considerable bond strain for 3 and 4 carbon rings. 5 and 6 carbon rings have much less bond strain. They can buckle their structures to achieve more stability. You name them just as alkanes ...
... 8-carbons octane Cycloalkanes These hydrocarbons exist in ring structures with general formula CnH2n. There is considerable bond strain for 3 and 4 carbon rings. 5 and 6 carbon rings have much less bond strain. They can buckle their structures to achieve more stability. You name them just as alkanes ...
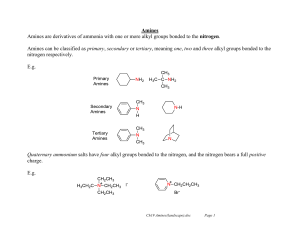
Amines Amines are derivatives of ammonia with one or more alkyl
... FC alkylations and acetylations fail, and other EAS reactions require unusually harsh reaction conditions. The deactivation arises from the electron withdrawing effect of the nitrogen atom in the ring. The lone pair of the nitrogen sticks out away from the system, and so cannot be used to stabiliz ...
... FC alkylations and acetylations fail, and other EAS reactions require unusually harsh reaction conditions. The deactivation arises from the electron withdrawing effect of the nitrogen atom in the ring. The lone pair of the nitrogen sticks out away from the system, and so cannot be used to stabiliz ...
Molecular Structure and Orbitals - Blackboard
... Molecular Orbital Theory • The valence bond model is easy to visualize, and works well for most molecules, but it does not describe magnetic and spectral properties well. A more complex model must be used to explain these phenomena. • In Molecular Orbital (MO) theory, electrons occupy molecular orbi ...
... Molecular Orbital Theory • The valence bond model is easy to visualize, and works well for most molecules, but it does not describe magnetic and spectral properties well. A more complex model must be used to explain these phenomena. • In Molecular Orbital (MO) theory, electrons occupy molecular orbi ...
Carbonyl Condensation Reactions
... As we learned in Section 23.3, the α hydrogens between two carbonyl groups are especially acidic, and so they are more readily removed than other α H atoms. As a result, the a-dicarbonyl compound always becomes the enolate component of the aldol reaction. Figure 24.2 shows the steps for the crossed ...
... As we learned in Section 23.3, the α hydrogens between two carbonyl groups are especially acidic, and so they are more readily removed than other α H atoms. As a result, the a-dicarbonyl compound always becomes the enolate component of the aldol reaction. Figure 24.2 shows the steps for the crossed ...
chapter 22 organic and biological molecules
... functional group. The monomer for a homopolymer polyester must have an alcohol functional group and a carboxylic acid functional group present in the structure. b. A polyamide forms when an amine functional group reacts with a carboxylic acid functional group. For a copolymer polyamide, one monomer ...
... functional group. The monomer for a homopolymer polyester must have an alcohol functional group and a carboxylic acid functional group present in the structure. b. A polyamide forms when an amine functional group reacts with a carboxylic acid functional group. For a copolymer polyamide, one monomer ...
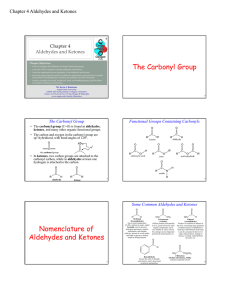
The Carbonyl Group Nomenclature of Aldehydes and Ketones
... • Ketones react with alcohols first to form hemiketals, which then react with excess alcohol to ...
... • Ketones react with alcohols first to form hemiketals, which then react with excess alcohol to ...
N-oxidation - WordPress.com
... Methemoglobinemia toxicity • Methemoglobinemia toxicity is caused by several aromatic amines, including aniline and dapsone, and is a result of the bioconversion of the aromatic amine to its N-hydroxy derivative. • The N-hydroxylamine oxidizes the Fe2+ form of hemoglobin to its Fe3+ form. This oxid ...
... Methemoglobinemia toxicity • Methemoglobinemia toxicity is caused by several aromatic amines, including aniline and dapsone, and is a result of the bioconversion of the aromatic amine to its N-hydroxy derivative. • The N-hydroxylamine oxidizes the Fe2+ form of hemoglobin to its Fe3+ form. This oxid ...
haloalkanes and arenes
... (iii) It is used as a solvent in the manufacture of pharmaceutical products. (iv) Until the mid 1960’s, carbon tetrachloride was widely used as a cleaning fluid, a degreasing agent in industries, a spot reamer in homes, and a fire extinguisher. Uses of iodoform (CHI3) Iodoform was used earlier as an ...
... (iii) It is used as a solvent in the manufacture of pharmaceutical products. (iv) Until the mid 1960’s, carbon tetrachloride was widely used as a cleaning fluid, a degreasing agent in industries, a spot reamer in homes, and a fire extinguisher. Uses of iodoform (CHI3) Iodoform was used earlier as an ...
High-resolution molecular structure of a peptide
... the cross-peak between I107 13C␦ and I107 15N, which corresponds to a ⬇4.5 Å distance, has an intensity significantly lower than the T106 13C␥–T106 15N and T106 13C␥–I107 15N crosspeaks at calculated distances of ⬇3.5 and 3 Å, respectively. As demonstrated previously (30), precise and accurate dista ...
... the cross-peak between I107 13C␦ and I107 15N, which corresponds to a ⬇4.5 Å distance, has an intensity significantly lower than the T106 13C␥–T106 15N and T106 13C␥–I107 15N crosspeaks at calculated distances of ⬇3.5 and 3 Å, respectively. As demonstrated previously (30), precise and accurate dista ...
CHAPTER 21 ORGANIC CHEMISTRY
... To explain the equivalent bonds, the π bonds can’t be situated between two carbon atoms, as is the case in simple alkenes and alkynes; that is, the π bonds can’t be localized. Instead, the ...
... To explain the equivalent bonds, the π bonds can’t be situated between two carbon atoms, as is the case in simple alkenes and alkynes; that is, the π bonds can’t be localized. Instead, the ...
Homoaromaticity

Homoaromaticity in organic chemistry refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thought to be a requirement for aromaticity, considerable thermodynamic stability and many of the spectroscopic, magnetic, and chemical properties associated with aromatic compounds are still observed for such compounds. This formal discontinuity is apparently bridged by p-orbital overlap, maintaining a contiguous cycle of π electrons that is responsible for this preserved chemical stability.The concept of homoaromaticity was pioneered by Saul Winstein in 1959, prompted by his studies of the “tris-homocyclopropenyl” cation. Since the publication of Winstein's paper, much research has been devoted to understanding and classifying these molecules, which represent an additional “class” of aromatic molecules included under the continuously broadening definition of aromaticity. To date, homoaromatic compounds are known to exist as cationic and anionic species, and some studies support the existence of neutral homoaromatic molecules, though these are less common. The 'homotropylium' cation (C8H9+) is perhaps the best studied example of a homoaromatic compound.