Topic Selection Menu - Pennsylvania State University
... – Absorption of electromagnetic radiation UV-Vis range ...
... – Absorption of electromagnetic radiation UV-Vis range ...
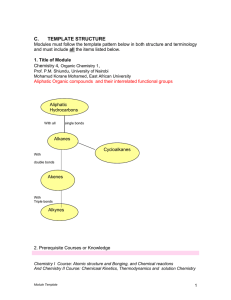
Ch-1-Alkanes and isomerism-corr
... Know general formula of simple alkanes and their names from methane to decane. Know the different methods of representing molecular formulas Know the different classes of carbon and hydrogen atoms know the hybridization and geometry of alkanes Know the rules for naming branched chain alkan ...
... Know general formula of simple alkanes and their names from methane to decane. Know the different methods of representing molecular formulas Know the different classes of carbon and hydrogen atoms know the hybridization and geometry of alkanes Know the rules for naming branched chain alkan ...
Test Review
... Make sure you review the concepts of the labs we performed: Formation of an Alkene and Evaporation and Intermolecular Attractions. ...
... Make sure you review the concepts of the labs we performed: Formation of an Alkene and Evaporation and Intermolecular Attractions. ...
Chapter 23: Substituted Hydrocarbons and Their Reactions
... commonly called vinyl is polyvinyl chloride (PVC). It can be manufactured soft or hard, as thin sheets or molded into objects. Organic halides are seldom found in nature, although human thyroid hormones are organic iodides. One example of an organic halide is shown in Figure 23-2. Alkyl halides are ...
... commonly called vinyl is polyvinyl chloride (PVC). It can be manufactured soft or hard, as thin sheets or molded into objects. Organic halides are seldom found in nature, although human thyroid hormones are organic iodides. One example of an organic halide is shown in Figure 23-2. Alkyl halides are ...
OEV I04 General Chemistry_1 - The Open University of Tanzania
... This course is designed to introduce the basic concepts of inorganic, organic and physical chemistry upon which understanding of modern chemistry depends. These concepts include atomic structure, ideal gas behavior and its deviation; covalent and ionic bonding; the concept of reaction mechanism in t ...
... This course is designed to introduce the basic concepts of inorganic, organic and physical chemistry upon which understanding of modern chemistry depends. These concepts include atomic structure, ideal gas behavior and its deviation; covalent and ionic bonding; the concept of reaction mechanism in t ...
2.6 M - Thierry Karsenti
... If you get 6 items or more correct you can consider that you are doing fine, but if you get less than 4 items correct then you have to work very hard to pass the course. ...
... If you get 6 items or more correct you can consider that you are doing fine, but if you get less than 4 items correct then you have to work very hard to pass the course. ...
The Inhibition of Carbon Steel Corrosion in Hydrochloric Acid
... MXads refer to the halide ion and ion – pair respectively in the adsorbed state. This ion – pair interaction increases the surface coverage thereby reducing metal dissolution. The order of decreasing inhibition efficiency of the investigated compounds on addition of a specific concentration of the u ...
... MXads refer to the halide ion and ion – pair respectively in the adsorbed state. This ion – pair interaction increases the surface coverage thereby reducing metal dissolution. The order of decreasing inhibition efficiency of the investigated compounds on addition of a specific concentration of the u ...
Inorganometallic Chemistry
... atoms of an organic group or molecule and the atom from the main group, transition, lanthanide or actinide metals. According to the IUPAC rules, organometallic compounds are those in which the carbon atoms are bonded to any other element with exception of H, C, N, O, F, Cl, Br, I and At. Some diffic ...
... atoms of an organic group or molecule and the atom from the main group, transition, lanthanide or actinide metals. According to the IUPAC rules, organometallic compounds are those in which the carbon atoms are bonded to any other element with exception of H, C, N, O, F, Cl, Br, I and At. Some diffic ...
Organic Chemistry - Snow College | It's SNOWing
... Thiols and Sulfides • Thiols are named by adding suffix “thiol” • Remember to keep the e • Common names are alkyl mercaptans ...
... Thiols and Sulfides • Thiols are named by adding suffix “thiol” • Remember to keep the e • Common names are alkyl mercaptans ...
EASTERN ARIZONA COLLEGE General Organic Chemistry I
... Use the polygon rule to draw the energy diagram for a cyclic system of p orbitals, and fill in the electrons to show whether a given compound or ion is aromatic or antiaromatic. ...
... Use the polygon rule to draw the energy diagram for a cyclic system of p orbitals, and fill in the electrons to show whether a given compound or ion is aromatic or antiaromatic. ...
methods and models
... opening new horizons for theoretical study of structure and properties of coordination compounds. Quantum chemical study of transition metals compounds and its reactions is supposing the using of multideterminant approaches (CAS, CASPT2) or coupled cluster method (CCSD, CCSD(T)), however a systemati ...
... opening new horizons for theoretical study of structure and properties of coordination compounds. Quantum chemical study of transition metals compounds and its reactions is supposing the using of multideterminant approaches (CAS, CASPT2) or coupled cluster method (CCSD, CCSD(T)), however a systemati ...
CHEMISTRY 331 - Western Illinois University
... Hands-on experience is a vital factor in learning chemistry. Since the laboratory experience is integral to the overall course, failure to earn a passing grade in the lab will result in automatic failure for the course. Notebooks will be checked to verify that they contain the information necessary ...
... Hands-on experience is a vital factor in learning chemistry. Since the laboratory experience is integral to the overall course, failure to earn a passing grade in the lab will result in automatic failure for the course. Notebooks will be checked to verify that they contain the information necessary ...
Lab Grade 250 pts - Western Illinois University
... Hands-on experience is a vital factor in learning chemistry. Since the laboratory experience is integral to the overall course, failure to earn a passing grade in the lab will result in automatic failure for the course. Notebooks will be checked to verify that they contain the information necessary ...
... Hands-on experience is a vital factor in learning chemistry. Since the laboratory experience is integral to the overall course, failure to earn a passing grade in the lab will result in automatic failure for the course. Notebooks will be checked to verify that they contain the information necessary ...
SOL Review Part 3 Nomenclature reactions
... When naming a transition metal that has more than one oxidation number, the numeric value of the oxidation number is indicated by a — A Roman numeral _ B Greek prefix C subscript D suffix ...
... When naming a transition metal that has more than one oxidation number, the numeric value of the oxidation number is indicated by a — A Roman numeral _ B Greek prefix C subscript D suffix ...
Basic Agricultural Chemistry - Macmillan Education South Africa
... group share similar chemical properties. The Roman numerals listed above each group are the usual number of valence electrons. For example: l Group IA is the alkali metals where the element has one electron in the orbital, for example sodium, potassium and rubidium. Alkali metals form salts and man ...
... group share similar chemical properties. The Roman numerals listed above each group are the usual number of valence electrons. For example: l Group IA is the alkali metals where the element has one electron in the orbital, for example sodium, potassium and rubidium. Alkali metals form salts and man ...
Hein and Arena
... than those of alcohols with similar molecular weights, because none of these compounds are able to form hydrogen bonds to like molecules. • Ether molecules are slightly polar as a consequence of the C-O-C linkage, but the dipole-dipole attractions that occur between ether molecules are not strong en ...
... than those of alcohols with similar molecular weights, because none of these compounds are able to form hydrogen bonds to like molecules. • Ether molecules are slightly polar as a consequence of the C-O-C linkage, but the dipole-dipole attractions that occur between ether molecules are not strong en ...
ap carbohydrates and lipids
... 2.1 Atomic Structure Is the Basis for Life’s Chemistry 2.2 Atoms Interact and Form Molecules 2.3 Carbohydrates Consist of Sugar Molecules ...
... 2.1 Atomic Structure Is the Basis for Life’s Chemistry 2.2 Atoms Interact and Form Molecules 2.3 Carbohydrates Consist of Sugar Molecules ...
PoL2e Ch02 Lecture-The Chemistry and Energy of Life
... 2.1 Atomic Structure Is the Basis for Life’s Chemistry 2.2 Atoms Interact and Form Molecules 2.3 Carbohydrates Consist of Sugar Molecules 2.4 Lipids Are Hydrophobic Molecules ...
... 2.1 Atomic Structure Is the Basis for Life’s Chemistry 2.2 Atoms Interact and Form Molecules 2.3 Carbohydrates Consist of Sugar Molecules 2.4 Lipids Are Hydrophobic Molecules ...
4 ORGANIC CHEMISTRY: STRUCTURE AND NOMENCLATURE
... Because the bond angle in a tetrahedron is 109.5°, alkane molecules that contain three or carbon atoms can no longer be thought of as “linear,” as shown in Figure O1.5. ...
... Because the bond angle in a tetrahedron is 109.5°, alkane molecules that contain three or carbon atoms can no longer be thought of as “linear,” as shown in Figure O1.5. ...
Copper Coordination Polymers with Infinite Chloride Ion Channels
... donor ligands. The Jahn-Teller effect normally leads to an elongation of the bonds towards the spectrochemically weaker ligands. At the same time there are no decisive features, such as O-H..CI hydrogen bonds, in the "non-bondcd" coordination spheres for both chlorine atoms to account for the differ ...
... donor ligands. The Jahn-Teller effect normally leads to an elongation of the bonds towards the spectrochemically weaker ligands. At the same time there are no decisive features, such as O-H..CI hydrogen bonds, in the "non-bondcd" coordination spheres for both chlorine atoms to account for the differ ...
The Formation of Solvated Electrons in the Photochemistry of the
... radiation chemicalI2 and photochemical1@data is approximately il.9. One therefore gets k~~ + e a e - j k ~+z ~ e4Q- = 0.4 to 0.6, in fair agreement with the value obtained in our treatment. These results yield a further independent support to the identification of the species formed from the excited ...
... radiation chemicalI2 and photochemical1@data is approximately il.9. One therefore gets k~~ + e a e - j k ~+z ~ e4Q- = 0.4 to 0.6, in fair agreement with the value obtained in our treatment. These results yield a further independent support to the identification of the species formed from the excited ...
Facile Oxidation of Benzyl Alcohols with Sodium Nitrate/p
... microwave irradiation for 30-300 sec using domestic microwave oven cleanly afforded the corresponding benzaldehydes. Some representative results obtained in the present study are shown in Table 1. The oxidation of various benzyl alcohols gave high yields of benzaldehydes in very short reaction times ...
... microwave irradiation for 30-300 sec using domestic microwave oven cleanly afforded the corresponding benzaldehydes. Some representative results obtained in the present study are shown in Table 1. The oxidation of various benzyl alcohols gave high yields of benzaldehydes in very short reaction times ...
Homoaromaticity

Homoaromaticity in organic chemistry refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thought to be a requirement for aromaticity, considerable thermodynamic stability and many of the spectroscopic, magnetic, and chemical properties associated with aromatic compounds are still observed for such compounds. This formal discontinuity is apparently bridged by p-orbital overlap, maintaining a contiguous cycle of π electrons that is responsible for this preserved chemical stability.The concept of homoaromaticity was pioneered by Saul Winstein in 1959, prompted by his studies of the “tris-homocyclopropenyl” cation. Since the publication of Winstein's paper, much research has been devoted to understanding and classifying these molecules, which represent an additional “class” of aromatic molecules included under the continuously broadening definition of aromaticity. To date, homoaromatic compounds are known to exist as cationic and anionic species, and some studies support the existence of neutral homoaromatic molecules, though these are less common. The 'homotropylium' cation (C8H9+) is perhaps the best studied example of a homoaromatic compound.