constructive - Purdue Physics
... can kick the electrons far enough so they do not come back to the metal. Once electrons are away one can direct them with electric fields. Certain night vision systems for example work that way. ...
... can kick the electrons far enough so they do not come back to the metal. Once electrons are away one can direct them with electric fields. Certain night vision systems for example work that way. ...
1. Define the vocabulary on page 88. Section 1
... 3. All forms of electromagnetic radiation move at a constant speed of _____________ through a vacuum. 4. _________ is the distance between corresponding points on adjacent waves. 5. What is the symbol for wavelength? 6. Frequency is defined as _______________________________________. 7. What is the ...
... 3. All forms of electromagnetic radiation move at a constant speed of _____________ through a vacuum. 4. _________ is the distance between corresponding points on adjacent waves. 5. What is the symbol for wavelength? 6. Frequency is defined as _______________________________________. 7. What is the ...
Quantum Theory of Light. Matter Waves.
... region of space, have energy and momentum, and interact with other particles (like billiard balls). ...
... region of space, have energy and momentum, and interact with other particles (like billiard balls). ...
FE Review Chemistry - UTSA College of Engineering
... • Electronegativity: is a chemical property that describes the tendency of an atom or a functional group to attract electrons (or electron density) towards itself. • Ionization energy: is the energy required to remove electrons from atoms or ions. • Atomic radius: the size of the atom ...
... • Electronegativity: is a chemical property that describes the tendency of an atom or a functional group to attract electrons (or electron density) towards itself. • Ionization energy: is the energy required to remove electrons from atoms or ions. • Atomic radius: the size of the atom ...
Tuesday Aug 19
... 1. Compare the spectra produced by sunlight (white light) and fluorescent sources. What are the similarities? What are the differences? 2. Some elements have the same color, but brighter spectral lines than others. How can the difference in the brightness be explained? ...
... 1. Compare the spectra produced by sunlight (white light) and fluorescent sources. What are the similarities? What are the differences? 2. Some elements have the same color, but brighter spectral lines than others. How can the difference in the brightness be explained? ...
UNM Physics 262, Problem Set 12, Fall 2006
... energy terms separate at this point. (b) Now use Newton's second law and the Coulomb force to rewrite the classical energy of the hydrogen atom entirely in terms of its radius. What radius corresponds to the lowest possible energy? (c) In the lowest energy quantum mechanical con guration of the hydr ...
... energy terms separate at this point. (b) Now use Newton's second law and the Coulomb force to rewrite the classical energy of the hydrogen atom entirely in terms of its radius. What radius corresponds to the lowest possible energy? (c) In the lowest energy quantum mechanical con guration of the hydr ...
Transcript - the Cassiopeia Project
... But of course, the atom is not always found in this lowest energy state. As there are other orbits allowed in the Bohr model, there are other, higher energy states in the quantum mechanical hydrogen atom. These states are defined primarily by the quantum number “n” that we talked about earlier. And ...
... But of course, the atom is not always found in this lowest energy state. As there are other orbits allowed in the Bohr model, there are other, higher energy states in the quantum mechanical hydrogen atom. These states are defined primarily by the quantum number “n” that we talked about earlier. And ...
Document
... 4. How much work must be done to pull apart the electron and the proton that make up the hydrogen atom if the atom is initially in (a) its ground state and (b) the state with n = 2? ANSWER: (a) 13.6 eV; (b) 3.40 eV 5. What is the probability that in the ground state of the hydrogen atom, the electro ...
... 4. How much work must be done to pull apart the electron and the proton that make up the hydrogen atom if the atom is initially in (a) its ground state and (b) the state with n = 2? ANSWER: (a) 13.6 eV; (b) 3.40 eV 5. What is the probability that in the ground state of the hydrogen atom, the electro ...
4 slides per page() - Wayne State University Physics and
... electron orbits. Therefore the transitions between orbits (energy levels) could be arbitrarily large or small. This leads to a continuous spectrum of emitted light. ...
... electron orbits. Therefore the transitions between orbits (energy levels) could be arbitrarily large or small. This leads to a continuous spectrum of emitted light. ...
Matter Unit Study Guide Phases of Matter
... What is the mass of the object on this triple beam balance? ...
... What is the mass of the object on this triple beam balance? ...
Introduction to Quantum Mechanics Homework #3 (Due on April 28
... 6.1) What are the electronic configurations of Ti atom and Fe3+ ion, respectively? Hint: Consider which orbital electrons are ionized first. 2) If one performs the Stern-Gelach experiment using a Fe3+ ion beam, how many iron spots do you expect to be deposited on a glass plate after passing through ...
... 6.1) What are the electronic configurations of Ti atom and Fe3+ ion, respectively? Hint: Consider which orbital electrons are ionized first. 2) If one performs the Stern-Gelach experiment using a Fe3+ ion beam, how many iron spots do you expect to be deposited on a glass plate after passing through ...
Physics 228 Today: Atomic Structure Bohr Model of H Atom
... positively charged, massive nucleus ...
... positively charged, massive nucleus ...
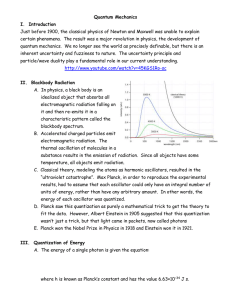
Quantum Mechanics I. Introduction Just before 1900, the classical
... C. Classical theory, modeling the atoms as harmonic oscillators, resulted in the “ultraviolet catastrophe”. Max Planck, in order to reproduce the experimental results, had to assume that each oscillator could only have an integral number of units of energy, rather than have any arbitrary amount. In ...
... C. Classical theory, modeling the atoms as harmonic oscillators, resulted in the “ultraviolet catastrophe”. Max Planck, in order to reproduce the experimental results, had to assume that each oscillator could only have an integral number of units of energy, rather than have any arbitrary amount. In ...
Chapter 8 - Fayetteville State University
... -4A. for a single element complement one another. B. can be used to identify elements in unknown samples, but only if the element is already known by classical chemical means. C. when combined together form a series of bright lines. D. for certain pairs of closely-related elements are identical. 14 ...
... -4A. for a single element complement one another. B. can be used to identify elements in unknown samples, but only if the element is already known by classical chemical means. C. when combined together form a series of bright lines. D. for certain pairs of closely-related elements are identical. 14 ...
Atoms1 - Cbsephysicstutorials
... The quantization or discretisation of a physical quantity means that it cannot very continuously to have any arbitrary value but can change only discontinuously to take certain specific values. • Bohr’s Model for the Hydrogen Atom: Basic postulates: a) Nuclear concept: An atom consists of a small ma ...
... The quantization or discretisation of a physical quantity means that it cannot very continuously to have any arbitrary value but can change only discontinuously to take certain specific values. • Bohr’s Model for the Hydrogen Atom: Basic postulates: a) Nuclear concept: An atom consists of a small ma ...
ATOMIC STRUCTURE
... En = (-RH)(1/n2) n = 1,2,3,4…. RH = Rydberg constant (2.18 x 10-18 J) n = principle quantun number ...
... En = (-RH)(1/n2) n = 1,2,3,4…. RH = Rydberg constant (2.18 x 10-18 J) n = principle quantun number ...
Zn + HCl → ZnCl 2 + H2 NaOH + H3PO4 → Na3PO4 + H2O N2 +
... This is a trial-and-error process, so work in pencil and be patient with yourself! Look at only one type of atom at a time. Start with atoms that appear only once on each side of the equation. Once those are balanced, try to balance atoms that appear in more than one species on either side o ...
... This is a trial-and-error process, so work in pencil and be patient with yourself! Look at only one type of atom at a time. Start with atoms that appear only once on each side of the equation. Once those are balanced, try to balance atoms that appear in more than one species on either side o ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.