All you need to know about Additional Science
... relative atomic mass is therefore calculated using the equation: • (% of isotope 1 × mass of isotope 1) + (% of isotope 2 × mass of isotope 2) ÷ 100 So in the case of chlorine: ...
... relative atomic mass is therefore calculated using the equation: • (% of isotope 1 × mass of isotope 1) + (% of isotope 2 × mass of isotope 2) ÷ 100 So in the case of chlorine: ...
Atomic Structure and the Periodic Table Atomic Structure and the
... German (L) Mendeleev (1834(1834-1907) Russian (R) chemists arrange elements by mass from light to heavy Properties density, solubility, melting & boiling points, ionization energies, hardness recur at definite intervals Periods ...
... German (L) Mendeleev (1834(1834-1907) Russian (R) chemists arrange elements by mass from light to heavy Properties density, solubility, melting & boiling points, ionization energies, hardness recur at definite intervals Periods ...
Charged Particles in Magnetic Fields
... q v B = m v2 /r and r = mv /q B, (equation #1). Solving for charge to mass ratio, q / m = v / r B (equation #2). Figure #1 The Path of an Electron in a Uniform Magnetic Field ...
... q v B = m v2 /r and r = mv /q B, (equation #1). Solving for charge to mass ratio, q / m = v / r B (equation #2). Figure #1 The Path of an Electron in a Uniform Magnetic Field ...
Chapter 7. The Quantum-Mechanical Model of the Atom 100
... Know that electrons and photons behave in similar ways: both can act as particles and as waves. Know that photons and electrons, even when viewed as streams of particles, still display diffraction a ...
... Know that electrons and photons behave in similar ways: both can act as particles and as waves. Know that photons and electrons, even when viewed as streams of particles, still display diffraction a ...
Quantifying Chemical Compounds Script
... Two laws were formed based on observations of scientists conducting research in the late eighteenth and early nineteenth century. These laws predate atomic theory, and were instrumental to scientists in determining that matter is composed of atoms. A compound is two or more bonded atoms of different ...
... Two laws were formed based on observations of scientists conducting research in the late eighteenth and early nineteenth century. These laws predate atomic theory, and were instrumental to scientists in determining that matter is composed of atoms. A compound is two or more bonded atoms of different ...
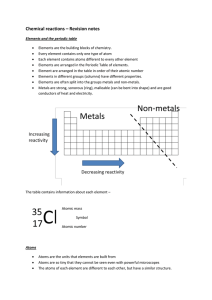
Chemical reactions revision
... Atoms are the units that elements are built from Atoms are so tiny that they cannot be seen even with powerful microscopes The atoms of each element are different to each other, but have a similar structure. ...
... Atoms are the units that elements are built from Atoms are so tiny that they cannot be seen even with powerful microscopes The atoms of each element are different to each other, but have a similar structure. ...
Matter - Moodle
... • The chemical composition ______________________ A chemical property describes how a substance ________________ into a new substance Either by: • __________________ with other elements • _________________ __________________ into new substances ...
... • The chemical composition ______________________ A chemical property describes how a substance ________________ into a new substance Either by: • __________________ with other elements • _________________ __________________ into new substances ...
Topic 2 - Jensen Chemistry
... Modern theory states: Atoms cannot be subdivided, created, or destroyed in ordinary chemical reactions. However, these changes CAN occur in nuclear reactions ...
... Modern theory states: Atoms cannot be subdivided, created, or destroyed in ordinary chemical reactions. However, these changes CAN occur in nuclear reactions ...
Midterm Review Date
... chemical change. Substance Z can be broken down by a chemical change. What can be concluded about these substances? A) Both substances are elements. B) Both substances are compounds. C) Substance A is an element and substance Z is a compound. D) Substance A is a compound and substance Z is an elemen ...
... chemical change. Substance Z can be broken down by a chemical change. What can be concluded about these substances? A) Both substances are elements. B) Both substances are compounds. C) Substance A is an element and substance Z is a compound. D) Substance A is a compound and substance Z is an elemen ...
Chapter2. Elements of quantum mechanics
... a. A projectile electron of kinetic energy 12.2 eV collides with a hydrogen atom in a gas discharge tube. Find the n-th energy level to which the electron in the hydrogen atom gets excited. b. Calculate the possible wavelengths of radiation (in nm) that will be emitted from the excited H atom in par ...
... a. A projectile electron of kinetic energy 12.2 eV collides with a hydrogen atom in a gas discharge tube. Find the n-th energy level to which the electron in the hydrogen atom gets excited. b. Calculate the possible wavelengths of radiation (in nm) that will be emitted from the excited H atom in par ...
Physical Science
... Unit 2: Chapters 3 & 4, Properties and Change/Structure of the Atom Properties of Matter – states, physical/chemical properties Changes in Matter – Physical/Chemical Change Law of Conservation of Matter Mixtures – hetero/homogenous. Separation of mixtures Elements and Comounds Law of Def ...
... Unit 2: Chapters 3 & 4, Properties and Change/Structure of the Atom Properties of Matter – states, physical/chemical properties Changes in Matter – Physical/Chemical Change Law of Conservation of Matter Mixtures – hetero/homogenous. Separation of mixtures Elements and Comounds Law of Def ...
Chapter 4 Spectroscopy
... produces continuous spectrum • Low-density hot gas produces emission spectrum • Continuous spectrum incident on cool, thin gas produces absorption spectrum ...
... produces continuous spectrum • Low-density hot gas produces emission spectrum • Continuous spectrum incident on cool, thin gas produces absorption spectrum ...
Lecture Notes, Feb 24, 2016
... In December 1925 while on vacation, Schro”odinger ( Univ of Zurich physics professor ), looked at de Broglie’s thesis. He work out a single equation, explaining the behavior of particles in terms of de Broglie waves. The lead player in the equation is a quantity called Ψ ( pronounced ”sigh” ) which ...
... In December 1925 while on vacation, Schro”odinger ( Univ of Zurich physics professor ), looked at de Broglie’s thesis. He work out a single equation, explaining the behavior of particles in terms of de Broglie waves. The lead player in the equation is a quantity called Ψ ( pronounced ”sigh” ) which ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 15. The internuclear distance of the 1H35Cl molecule is 0.128 nm. Calculate the spacing of the lines in its rotational spectrum in terms of cm-1. 16. Obtain all the possible term symbols for a 4F state. ...
... 15. The internuclear distance of the 1H35Cl molecule is 0.128 nm. Calculate the spacing of the lines in its rotational spectrum in terms of cm-1. 16. Obtain all the possible term symbols for a 4F state. ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.