8th Grade Science Chapter 11 Answers
... 16. Current theory on the atom is that electrons are likely to be found in regions called electron clouds. Electron clouds are in the space surrounding the nucleus. 17. An atom consists of the electrons and nucleus, as well as the protons and neutrons which are found inside the nucleus. P 288 – Mak ...
... 16. Current theory on the atom is that electrons are likely to be found in regions called electron clouds. Electron clouds are in the space surrounding the nucleus. 17. An atom consists of the electrons and nucleus, as well as the protons and neutrons which are found inside the nucleus. P 288 – Mak ...
Name Date Class Period ______
... A. Atomic number B. Atomic mass C. Element symbol D. K shell E. Neutron F. Proton G. M shell H. Electron I. Nucleus J. Shells K. Atom L. Element M. Subatomic particles ...
... A. Atomic number B. Atomic mass C. Element symbol D. K shell E. Neutron F. Proton G. M shell H. Electron I. Nucleus J. Shells K. Atom L. Element M. Subatomic particles ...
Chapter 25 Nuclear Chemistry
... Chain reaction- after each nucleus splits, 3 neutrons are released that will collide with 3 more nuclei and so on. The splitting grows exponentially. Atomic bombs are uncontrolled nuclear chain reactions. Nuclear Power Plants - Control the chain rx with control rods (carbon and cadmium) that control ...
... Chain reaction- after each nucleus splits, 3 neutrons are released that will collide with 3 more nuclei and so on. The splitting grows exponentially. Atomic bombs are uncontrolled nuclear chain reactions. Nuclear Power Plants - Control the chain rx with control rods (carbon and cadmium) that control ...
Radioactivity Unit - hrsbstaff.ednet.ns.ca
... During the decay, they emit photons of certain smaller frequencies, which we can then see, and the atoms return to their lower-energy stable state. This makes them glow in the dark. There are 3 types of radiation that can be emitted by nuclei: alpha (), beta () and gamma (). Each of these is asso ...
... During the decay, they emit photons of certain smaller frequencies, which we can then see, and the atoms return to their lower-energy stable state. This makes them glow in the dark. There are 3 types of radiation that can be emitted by nuclei: alpha (), beta () and gamma (). Each of these is asso ...
Ch - cmpascience
... In both theories, _________________________ orbit the nucleus and each electron has an energy level associated with its location. In _________________________ model of the atom, electrons were thought to orbit the nucleus in set paths, much like planets orbiting the sun. In the ___________________ ...
... In both theories, _________________________ orbit the nucleus and each electron has an energy level associated with its location. In _________________________ model of the atom, electrons were thought to orbit the nucleus in set paths, much like planets orbiting the sun. In the ___________________ ...
Atomic Structure
... 1. Atoms are composed of a small, dense, positively charged nucleus surrounded by negatively charged electrons 2. All electrons are identical in mass and charge 3. Protons and neutrons reside in the nucleus and are almost 2,000 times more massive than electrons. Neutrons are electrically neutral and ...
... 1. Atoms are composed of a small, dense, positively charged nucleus surrounded by negatively charged electrons 2. All electrons are identical in mass and charge 3. Protons and neutrons reside in the nucleus and are almost 2,000 times more massive than electrons. Neutrons are electrically neutral and ...
NUCLEAR CHEMISTRY: INTRO
... NUCLEAR CHEMISTRY: INTRO 1. Kinetic Stability : probability that an unstable nucleus will decompose into more stable species through radioactive decay. 2. All nuclides with 84 or more protons are unstable and will decay. • Light nuclides where Z = A-Z (neutron/proton ratio is 1). • Nuclides with eve ...
... NUCLEAR CHEMISTRY: INTRO 1. Kinetic Stability : probability that an unstable nucleus will decompose into more stable species through radioactive decay. 2. All nuclides with 84 or more protons are unstable and will decay. • Light nuclides where Z = A-Z (neutron/proton ratio is 1). • Nuclides with eve ...
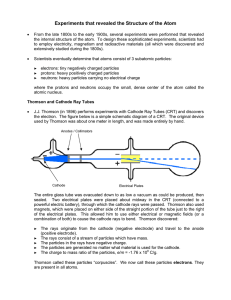
Experiments that revealed the Structure of the Atom
... Rutherford and his team (Geiger and Marsden) bombarded a number of different thin metal foils with alpha (α) particles from a radioactive source. Alpha particles are tiny, heavy particles with a positive charge. Particles passing through the foil would strike a screen coated with zinc sulfide, and a ...
... Rutherford and his team (Geiger and Marsden) bombarded a number of different thin metal foils with alpha (α) particles from a radioactive source. Alpha particles are tiny, heavy particles with a positive charge. Particles passing through the foil would strike a screen coated with zinc sulfide, and a ...
Notes on Atomic Structure atoms
... same proportions (by mass and by number) of its elements This means a given compound always has the same composition, regardless of where it came from. ...
... same proportions (by mass and by number) of its elements This means a given compound always has the same composition, regardless of where it came from. ...
Final Exam
... Physical Science Final Exam 1. Over time, there have been many scientists who have proposed different models of the atom. According to the most current atomic theory, where are the electrons located? A. in a dense, puddinglike region with the protons B. in circular orbits around the nucleus C. in th ...
... Physical Science Final Exam 1. Over time, there have been many scientists who have proposed different models of the atom. According to the most current atomic theory, where are the electrons located? A. in a dense, puddinglike region with the protons B. in circular orbits around the nucleus C. in th ...
Nuclear Fission and Fusion Notes
... To explain the difference between fusion and fission To describe half life and solve problems using half life ...
... To explain the difference between fusion and fission To describe half life and solve problems using half life ...
Electron Configuration and Chemical Periodicity
... used to describe the solutions (the orbitals are hydrogen-like) ...
... used to describe the solutions (the orbitals are hydrogen-like) ...
1.1 to 1.4
... d. noble gases (group 18) –extremely low chemical reactivity or inert (very stable) ...
... d. noble gases (group 18) –extremely low chemical reactivity or inert (very stable) ...
Energy From the Sun - Duplin County Schools
... Uppermost region of solar gases; 3 distinct layers: 1. photosphere – light sphere, made up of convective gases, about 6000oC; mostly visible light; considered surface of Sun 2. chromosphere – color sphere, can be 4000 to 50,000oC 3. corona – crown; ~2,000,000oC; thin, keeps most particles from escap ...
... Uppermost region of solar gases; 3 distinct layers: 1. photosphere – light sphere, made up of convective gases, about 6000oC; mostly visible light; considered surface of Sun 2. chromosphere – color sphere, can be 4000 to 50,000oC 3. corona – crown; ~2,000,000oC; thin, keeps most particles from escap ...
Chapter 30
... for, so Rutherford determined that there must be another component. • Through this idea the neutron was discovered, which has no charge, but makes up the rest of the mass. • The mass number (A), therefore is the sum of the protons and neutrons in an atom. • Rutherford understood that the nucleus was ...
... for, so Rutherford determined that there must be another component. • Through this idea the neutron was discovered, which has no charge, but makes up the rest of the mass. • The mass number (A), therefore is the sum of the protons and neutrons in an atom. • Rutherford understood that the nucleus was ...
Atomic Structure
... orbits called energy levels around the nucleus. • The potential energy of the electron is negative due to its attraction for the nucleus and gets more positive as the electron gets farther away from the nucleus. • These orbits are restricted (quantized) and electrons can only go from one energy leve ...
... orbits called energy levels around the nucleus. • The potential energy of the electron is negative due to its attraction for the nucleus and gets more positive as the electron gets farther away from the nucleus. • These orbits are restricted (quantized) and electrons can only go from one energy leve ...
South Pasadena • Chemistry
... Find O on the periodic table. It’s mass is ______ amu. It has ___ protons. It must have ___ neutrons. ...
... Find O on the periodic table. It’s mass is ______ amu. It has ___ protons. It must have ___ neutrons. ...
Atoms, Ions and Molecules The Building Blocks of Matter
... • From the physics and knowledge of the size of the gravitational and electrostatic forces, the charge on each droplet could be calculated • Discovered that each droplet was a whole-number multiple of 1.60 X 10-19 C, so the mass = 9.11 X 10-28 g ...
... • From the physics and knowledge of the size of the gravitational and electrostatic forces, the charge on each droplet could be calculated • Discovered that each droplet was a whole-number multiple of 1.60 X 10-19 C, so the mass = 9.11 X 10-28 g ...
Atomic nucleus
The nucleus is the small, dense region consisting of protons and neutrons at the center of an atom. The atomic nucleus was discovered in 1911 by Ernest Rutherford based on the 1909 Geiger–Marsden gold foil experiment. After the discovery of the neutron in 1932, models for a nucleus composed of protons and neutrons were quickly developed by Dmitri Ivanenko and Werner Heisenberg. Almost all of the mass of an atom is located in the nucleus, with a very small contribution from the electron cloud. Protons and neutrons are bound together to form a nucleus by the nuclear force.The diameter of the nucleus is in the range of 6985175000000000000♠1.75 fm (6985175000000000000♠1.75×10−15 m) for hydrogen (the diameter of a single proton) to about 6986150000000000000♠15 fm for the heaviest atoms, such as uranium. These dimensions are much smaller than the diameter of the atom itself (nucleus + electron cloud), by a factor of about 23,000 (uranium) to about 145,000 (hydrogen).The branch of physics concerned with the study and understanding of the atomic nucleus, including its composition and the forces which bind it together, is called nuclear physics.