Chapter 9: The Need for Energy
... The process by which mitochondria break down glucose to make ATP Two types o Aerobic respiration: requires oxygen and carried out by plants, animals, and some bacteria o Anaerobic respiration: requires no oxygen and carried out by yeast, some bacteria, and sometimes animals Chemical equation for ...
... The process by which mitochondria break down glucose to make ATP Two types o Aerobic respiration: requires oxygen and carried out by plants, animals, and some bacteria o Anaerobic respiration: requires no oxygen and carried out by yeast, some bacteria, and sometimes animals Chemical equation for ...
Answers to study guide
... their are 2 turns per glucose molecule) and 4x2 = 8 and 1x2 FADH2 is also where CO2 is released and then exhaled out. Takes place in the matrix of the mitochondrion. The third stage the Electron transport chain takes the energy carriers NADH and FADH2 and to carry electrons down the electron transpo ...
... their are 2 turns per glucose molecule) and 4x2 = 8 and 1x2 FADH2 is also where CO2 is released and then exhaled out. Takes place in the matrix of the mitochondrion. The third stage the Electron transport chain takes the energy carriers NADH and FADH2 and to carry electrons down the electron transpo ...
IB Biology 11 HL
... Which process is used to produce beer and wine? Carbon dioxide (CO2) is released during which of the following stages of cellular respiration? One cause of muscle soreness is… The ATP made during fermentation is generated by which of the following? In the absence of oxygen, yeast cells can ...
... Which process is used to produce beer and wine? Carbon dioxide (CO2) is released during which of the following stages of cellular respiration? One cause of muscle soreness is… The ATP made during fermentation is generated by which of the following? In the absence of oxygen, yeast cells can ...
Cell Energy
... is a proton gradient formed between the outer and inner membrane because of a pH and electric charge difference. This difference allows the H+ to flow through channels called ATP synthase using diffusion. As H+ flows through the channels ATP is formed from ADP and a phosphate. ...
... is a proton gradient formed between the outer and inner membrane because of a pH and electric charge difference. This difference allows the H+ to flow through channels called ATP synthase using diffusion. As H+ flows through the channels ATP is formed from ADP and a phosphate. ...
Photosynthesis/Cell Resp Notes
... The process by which mitochondria break down glucose to make ATP Two types o Aerobic respiration: requires oxygen and carried out by plants, animals, and some bacteria o Anaerobic respiration: requires no oxygen and carried out by yeast, some bacteria, and sometimes animals Chemical equation for ...
... The process by which mitochondria break down glucose to make ATP Two types o Aerobic respiration: requires oxygen and carried out by plants, animals, and some bacteria o Anaerobic respiration: requires no oxygen and carried out by yeast, some bacteria, and sometimes animals Chemical equation for ...
Note 4.2 - Aerobic Respiration
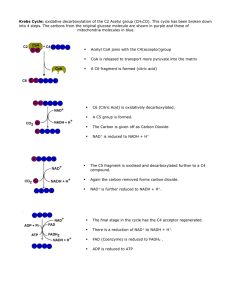
... 3. Citrate undergoes a dehydrogenization reaction where two electrons and two hydrogen protons are e released to form NADH and H+. Also a CO2 molecule is released, creating a molecule of α-ketoglutarate (5 carbons). This reaction is facilitated by the enzyme isocitrate dehydrogenase. 4. The α-ketogl ...
... 3. Citrate undergoes a dehydrogenization reaction where two electrons and two hydrogen protons are e released to form NADH and H+. Also a CO2 molecule is released, creating a molecule of α-ketoglutarate (5 carbons). This reaction is facilitated by the enzyme isocitrate dehydrogenase. 4. The α-ketogl ...
Questions and answers from course Environmental microbiology on
... 3. Give an example of ATP-conservation in a fermentation and respiration process. Fermentation: Glucose ...
... 3. Give an example of ATP-conservation in a fermentation and respiration process. Fermentation: Glucose ...
Oxidative phosphorylation.
... molecules built in to the mitochondial inner membrane . These molecules accept energy-rich electrons from reduced NADH (and also FADH2) and pass them along the chain to the final electron acceptor (oxygen). The electronegative oxygen accepts these electrons, along with hydrogen nuclei (recall dehydr ...
... molecules built in to the mitochondial inner membrane . These molecules accept energy-rich electrons from reduced NADH (and also FADH2) and pass them along the chain to the final electron acceptor (oxygen). The electronegative oxygen accepts these electrons, along with hydrogen nuclei (recall dehydr ...
Fundamentals of Biochemistry 2/e
... Glycolysis converts to two C3 units. The free energy released in this process is harvested to synthesize ATP from ADP and Pi ...
... Glycolysis converts to two C3 units. The free energy released in this process is harvested to synthesize ATP from ADP and Pi ...
Solutions to 7.014 Quiz I
... Yes. Because culture A derives all of its energy from glycolysis, it only produces 2 molecules of ATP/molecule of glucose. Thus, it will have to utilize more glucose to perform same amount of cellular functions as culture B that derives 36 molecules of ATP/molecule of glucose through respiration. ii ...
... Yes. Because culture A derives all of its energy from glycolysis, it only produces 2 molecules of ATP/molecule of glucose. Thus, it will have to utilize more glucose to perform same amount of cellular functions as culture B that derives 36 molecules of ATP/molecule of glucose through respiration. ii ...
Cellular Respiration
... In the above equation oxygen is the electron acceptor in the oxidation of glucose Most organisms are obligate aerobes – they require oxygen and cannot survive without it Obligate anaerobes (some species of bacteria) use other molecules as the final electron acceptor and must live in environments tha ...
... In the above equation oxygen is the electron acceptor in the oxidation of glucose Most organisms are obligate aerobes – they require oxygen and cannot survive without it Obligate anaerobes (some species of bacteria) use other molecules as the final electron acceptor and must live in environments tha ...
Energy
... Pine trees can lose up to 26 gallons of water on a sunny, hot day. Under dry conditions, they lose less water than other types of trees ...
... Pine trees can lose up to 26 gallons of water on a sunny, hot day. Under dry conditions, they lose less water than other types of trees ...
Microbial Metabolism
... “Electrical” because of the difference in charge “pH” because of the difference in [H+] E/C gradient has potential energy = PMF Proton Motive Force (PMF) generates energy sufficient to drive the ATP synthetase ...
... “Electrical” because of the difference in charge “pH” because of the difference in [H+] E/C gradient has potential energy = PMF Proton Motive Force (PMF) generates energy sufficient to drive the ATP synthetase ...
Anaerobic Respiration
... Some organisms, such as yeast and some bacteria, do not require oxygen and can survive on a less efficient way of getting energy Other organisms that generally require oxygen sometimes don’t have enough for all their cells to do aerobic respiration so they can use a less effiecent way of breaking do ...
... Some organisms, such as yeast and some bacteria, do not require oxygen and can survive on a less efficient way of getting energy Other organisms that generally require oxygen sometimes don’t have enough for all their cells to do aerobic respiration so they can use a less effiecent way of breaking do ...
1. glucose is broken down to pyruvate in the cytoplasm;
... glucose converted to pyruvate (two molecules); by glycolysis; pyruvate enters the mitochondria; pyruvate converted to acetyl CoA / ethyl CoA; by oxidative decarboxylation / NADH and CO2 formed; fatty acids / lipids converted to acetyl CoA; acetyl groups enter the Krebs cycle (accept acetyl CoA); FAD ...
... glucose converted to pyruvate (two molecules); by glycolysis; pyruvate enters the mitochondria; pyruvate converted to acetyl CoA / ethyl CoA; by oxidative decarboxylation / NADH and CO2 formed; fatty acids / lipids converted to acetyl CoA; acetyl groups enter the Krebs cycle (accept acetyl CoA); FAD ...
Lecture 5
... You will notice that some ATP is made and some Carbon Dioxide comes out of the process. But again, the main goal here is to make NADH. You will notice that FADH2 is also made. You don’t need to worry about knowing the difference between NADH and FADH2. For our purposes, you can assume they work the ...
... You will notice that some ATP is made and some Carbon Dioxide comes out of the process. But again, the main goal here is to make NADH. You will notice that FADH2 is also made. You don’t need to worry about knowing the difference between NADH and FADH2. For our purposes, you can assume they work the ...
Ions - RCSD
... F a. _______ One enzyme can be used for many different types of chemical reactions. F b. _______ Enzymes are used only once because they change shape after a reaction occurs. c. _______ Enzymes speed up reactions. T F d. _______ Raising the temperature will not change the rate of a reaction that use ...
... F a. _______ One enzyme can be used for many different types of chemical reactions. F b. _______ Enzymes are used only once because they change shape after a reaction occurs. c. _______ Enzymes speed up reactions. T F d. _______ Raising the temperature will not change the rate of a reaction that use ...
Cellular Respiration:
... Anaerobic Respiration: Glycolysis is the anaerobic (without oxygen) process that generates 2 ATP molecules per glucose molecule. It takes place in the cytoplasm (outside mitochondria). Glycolysis in your cells and glycolysis in bacterial cells is almost identical, strongly inferring that glycolysis ...
... Anaerobic Respiration: Glycolysis is the anaerobic (without oxygen) process that generates 2 ATP molecules per glucose molecule. It takes place in the cytoplasm (outside mitochondria). Glycolysis in your cells and glycolysis in bacterial cells is almost identical, strongly inferring that glycolysis ...
Muscle Metabolism - Liberty Union High School District
... tissues in the body that are depleted first and need to be replaced • Phosphagen system: ATP must be made, then broken to give Pi back to creatine • Oxidizing lactic acid: most of lactic acid will be converted into glucose in the presence of oxygen • Metabolic rate: if body temp is high the metaboli ...
... tissues in the body that are depleted first and need to be replaced • Phosphagen system: ATP must be made, then broken to give Pi back to creatine • Oxidizing lactic acid: most of lactic acid will be converted into glucose in the presence of oxygen • Metabolic rate: if body temp is high the metaboli ...
(Test Your Knowledge)
... 9. The phenomenon of viscosity is due to the transport of: a) Work b) Energy c) Force d) Momentum ...
... 9. The phenomenon of viscosity is due to the transport of: a) Work b) Energy c) Force d) Momentum ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.