Chapter 2 biochem
... Concentrations of H+ and OH- ions in body fluids can greatly affect chemical reactions ◦ Ex: Blood pressure, breathing rate ...
... Concentrations of H+ and OH- ions in body fluids can greatly affect chemical reactions ◦ Ex: Blood pressure, breathing rate ...
Energy represents the capacity to do work. Cells must
... 2. How did plants acquire photosynthesis in evolution? Name three features of chloroplasts that are indicative of their origin. (It is referred to as endosymbiosis or the endosymbiotic theory) Click 3. Photosynthesis can be divided in two different processes. What are these processes? What are ...
... 2. How did plants acquire photosynthesis in evolution? Name three features of chloroplasts that are indicative of their origin. (It is referred to as endosymbiosis or the endosymbiotic theory) Click 3. Photosynthesis can be divided in two different processes. What are these processes? What are ...
Higher Human Biology HW 3
... 29. Which of the following statements is CORRECT? A. Slow twitch muscle fibres are good for long distance running because they have fewer mitochondria and greater blood supply than fast twitch muscle fibres. B. Slow twitch muscle fibres are good for long distance running because they have more mito ...
... 29. Which of the following statements is CORRECT? A. Slow twitch muscle fibres are good for long distance running because they have fewer mitochondria and greater blood supply than fast twitch muscle fibres. B. Slow twitch muscle fibres are good for long distance running because they have more mito ...
Download PDF
... structure of proteins, and how this translates into differences in the function of these proteins. We will also cover the synthesis of biopolymers – peptide synthesis from protected amino acids and DNA synthesis from nucleoside phosphoramidites. 2. Energy metabolism. Biological systems use sugars an ...
... structure of proteins, and how this translates into differences in the function of these proteins. We will also cover the synthesis of biopolymers – peptide synthesis from protected amino acids and DNA synthesis from nucleoside phosphoramidites. 2. Energy metabolism. Biological systems use sugars an ...
Cell Metabolism - Cathkin High School
... 29. Which of the following statements is CORRECT? A. Slow twitch muscle fibres are good for long distance running because they have fewer mitochondria and greater blood supply than fast twitch muscle fibres. B. Slow twitch muscle fibres are good for long distance running because they have more mito ...
... 29. Which of the following statements is CORRECT? A. Slow twitch muscle fibres are good for long distance running because they have fewer mitochondria and greater blood supply than fast twitch muscle fibres. B. Slow twitch muscle fibres are good for long distance running because they have more mito ...
Chapter 6: Metabolism and Energy
... Terminal high-energy bond is more exergonic than others Activation energy is usually less than 7 kcal/mole Cells contain a pool of ATP, ADP and phosphate Cells do not stockpile ATP but create it as needed ATP constantly recycled BIOCHEMICAL PATHWAYS: THE ORGANIZATIONAL UNITS OF METABOLISM Reactions ...
... Terminal high-energy bond is more exergonic than others Activation energy is usually less than 7 kcal/mole Cells contain a pool of ATP, ADP and phosphate Cells do not stockpile ATP but create it as needed ATP constantly recycled BIOCHEMICAL PATHWAYS: THE ORGANIZATIONAL UNITS OF METABOLISM Reactions ...
Chapter 1
... 14. Distinguish between anaerobic and aerobic respiration. During cellular respiration, the oxidative processes that occur in the absence of oxygen are called anaerobic respiration. The oxidative processes that require the presence of oxygen for their reactions are called aerobic respiration. 15. Ex ...
... 14. Distinguish between anaerobic and aerobic respiration. During cellular respiration, the oxidative processes that occur in the absence of oxygen are called anaerobic respiration. The oxidative processes that require the presence of oxygen for their reactions are called aerobic respiration. 15. Ex ...
Chapter 4
... 14. Distinguish between anaerobic and aerobic respiration. During cellular respiration, the oxidative processes that occur in the absence of oxygen are called anaerobic respiration. The oxidative processes that require the presence of oxygen for their reactions are called aerobic respiration. 15. Ex ...
... 14. Distinguish between anaerobic and aerobic respiration. During cellular respiration, the oxidative processes that occur in the absence of oxygen are called anaerobic respiration. The oxidative processes that require the presence of oxygen for their reactions are called aerobic respiration. 15. Ex ...
lecture notes-metabolism pathways-web
... Metabolic Pathways - Bioenergetics - Reducing power: supply hydrogen atom in biosynthesis. Nicotinamide Adenine Dinucleotide (NADH) Flavin Adenine Dinucleotide (FADH2) NADH and FADH2 are major electron carriers in the oxidation of fuel molecules and for ATP generation. ...
... Metabolic Pathways - Bioenergetics - Reducing power: supply hydrogen atom in biosynthesis. Nicotinamide Adenine Dinucleotide (NADH) Flavin Adenine Dinucleotide (FADH2) NADH and FADH2 are major electron carriers in the oxidation of fuel molecules and for ATP generation. ...
ILS Unit 6 Semester 2 Name Teacher
... These complex cells, called eukaryotic cells, appeared about 2 billion years ago. They are the ancestors of most algae, fungi, plants, and animals. These are complex organisms with energy needs that only aerobic respiration provides. Without the marvelous process of aerobic respiration, early bacter ...
... These complex cells, called eukaryotic cells, appeared about 2 billion years ago. They are the ancestors of most algae, fungi, plants, and animals. These are complex organisms with energy needs that only aerobic respiration provides. Without the marvelous process of aerobic respiration, early bacter ...
2-Phospho
... • Electrons are transferred from NADH or FADH2 to the electron transport chain • Electrons are passed through a number of proteins including cytochromes (each with an iron atom) to O2 • The electron transport chain generates no ATP directly • It breaks the large free-energy drop from food to O2 int ...
... • Electrons are transferred from NADH or FADH2 to the electron transport chain • Electrons are passed through a number of proteins including cytochromes (each with an iron atom) to O2 • The electron transport chain generates no ATP directly • It breaks the large free-energy drop from food to O2 int ...
RESPIRATION PPT...Campbell Powerpoint presentation
... • Electrons are transferred from NADH or FADH2 to the electron transport chain • Electrons are passed through a number of proteins including cytochromes (each with an iron atom) to O2 • The electron transport chain generates no ATP directly • It breaks the large free-energy drop from food to O2 int ...
... • Electrons are transferred from NADH or FADH2 to the electron transport chain • Electrons are passed through a number of proteins including cytochromes (each with an iron atom) to O2 • The electron transport chain generates no ATP directly • It breaks the large free-energy drop from food to O2 int ...
Biochemistry Review
... C. A cell can release only stored energy D. A cell can create energy and does not need to get it from elsewhere in the organism ...
... C. A cell can release only stored energy D. A cell can create energy and does not need to get it from elsewhere in the organism ...
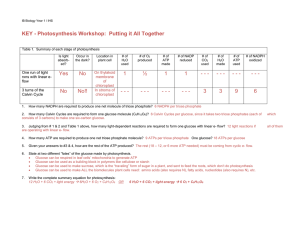
KEY - Photosynthesis Workshop: Putting it All Together
... consists of 3 carbons) to make one six-carbon glucose. ...
... consists of 3 carbons) to make one six-carbon glucose. ...
CHAPTER-IV LIPID METABOLISM BETA
... BETA-OXIDATION Beta-oxidation is the process by which fatty acids, in the form of acyl-CoA molecules, are broken down in mitochondria and/or peroxisomes to generate acetyl-CoA, the entry molecule for the citric acid cycle. The beta oxidation of fatty acids involve three stages: 1. Activation of fatt ...
... BETA-OXIDATION Beta-oxidation is the process by which fatty acids, in the form of acyl-CoA molecules, are broken down in mitochondria and/or peroxisomes to generate acetyl-CoA, the entry molecule for the citric acid cycle. The beta oxidation of fatty acids involve three stages: 1. Activation of fatt ...
normal myocardial metabolism: fueling cardiac contraction
... chain and protons move to the outside of the inner mitochondrial membrane to generate a proton gradient for ATP synthase activation. This is accomplished by an enzyme complex of 5 highly specialized proteins that are typically encoded by proper mitochondrial DNA. Electron flux is regulated to mainta ...
... chain and protons move to the outside of the inner mitochondrial membrane to generate a proton gradient for ATP synthase activation. This is accomplished by an enzyme complex of 5 highly specialized proteins that are typically encoded by proper mitochondrial DNA. Electron flux is regulated to mainta ...
Metabolism of RBC
... which moderates oxygen binding to Hb). The NADH generated from glycolysis can be used to reduce methemoglobin (Fe3+) to normal Hb (Fe2+) or To convert pyruvate to lactate, so that NAD+ can be regenerated and used for glycolysis. ...
... which moderates oxygen binding to Hb). The NADH generated from glycolysis can be used to reduce methemoglobin (Fe3+) to normal Hb (Fe2+) or To convert pyruvate to lactate, so that NAD+ can be regenerated and used for glycolysis. ...
Chem 115 POGIL Worksheet - Week 10 Periodic Trends Why? The
... Trends in sizes of atoms are the most important to understand, because other trends can often be rationalized on that basis. The most commonly used measure of size of an atom is its bonding atomic radius, also called the covalent radius.1 The bonding atomic radius of an element is taken as one half ...
... Trends in sizes of atoms are the most important to understand, because other trends can often be rationalized on that basis. The most commonly used measure of size of an atom is its bonding atomic radius, also called the covalent radius.1 The bonding atomic radius of an element is taken as one half ...
answers_ch04
... histidine and aspartate. Serine would serve as a nucleophile, histidine as an acid/base catalyst and aspartate as an activating and orientating group. The actual mechanism for the hydrolysis of acetylcholine is described in section 19.16.3.2. ...
... histidine and aspartate. Serine would serve as a nucleophile, histidine as an acid/base catalyst and aspartate as an activating and orientating group. The actual mechanism for the hydrolysis of acetylcholine is described in section 19.16.3.2. ...
Cellular Energy and Enzymatic Function
... • Substrates bind to active site on enzyme • Binding induces conformational change in enzyme--better ”fit” for substrate • Active sites are highly specific and ...
... • Substrates bind to active site on enzyme • Binding induces conformational change in enzyme--better ”fit” for substrate • Active sites are highly specific and ...
Test 2a
... (8 points)Tell me about the different types of catalysis you see occurring in this mechanism. In step 1 His 12 is acting as a general base to remove a proton from the 2' OH of the ribose, and His 119 is acitng as a general acid to donate its proton to the phosphate group between the two ribose sugar ...
... (8 points)Tell me about the different types of catalysis you see occurring in this mechanism. In step 1 His 12 is acting as a general base to remove a proton from the 2' OH of the ribose, and His 119 is acitng as a general acid to donate its proton to the phosphate group between the two ribose sugar ...
For lecture notes click here
... Glycolysis. During glycolysis, the cell gains 2 molecules of ATP directly for each glucose molecule broken down anaerobically to pyruvic acid. Two molecules of NADH are also produced. In most cells, electrons are passed from NADH to FAD by means of an intermediate in the intermembrane space and th ...
... Glycolysis. During glycolysis, the cell gains 2 molecules of ATP directly for each glucose molecule broken down anaerobically to pyruvic acid. Two molecules of NADH are also produced. In most cells, electrons are passed from NADH to FAD by means of an intermediate in the intermembrane space and th ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.