Gas Exchange
... Phosphogens – not a pathway but a class of phosphoamino acids that can store energy from ATP during periods of relaxation and deliver it under conditions of anoxia or exhaustion (phosphoarginine in invertes. phosphocreatine in verts. , echinoderms have both, annelids have 4 others) ...
... Phosphogens – not a pathway but a class of phosphoamino acids that can store energy from ATP during periods of relaxation and deliver it under conditions of anoxia or exhaustion (phosphoarginine in invertes. phosphocreatine in verts. , echinoderms have both, annelids have 4 others) ...
BCH 201 – GENERAL BIOCHEMISTRY 1 – (3 UNITS) DR
... The existence of enzymes has been known for well over a century. Some of the earliest studies were performed in 1835 by the Swedish chemist Jon Jakob Berzelius who termed their chemical action catalytic. It was not until 1926, however, that the first enzyme was obtained in pure form, a feat accompli ...
... The existence of enzymes has been known for well over a century. Some of the earliest studies were performed in 1835 by the Swedish chemist Jon Jakob Berzelius who termed their chemical action catalytic. It was not until 1926, however, that the first enzyme was obtained in pure form, a feat accompli ...
Atom
... • What is the difference between a compound and a molecule? • A molecule is formed when two or more atoms join together chemically. A compound is a molecule that contains at least two different elements. All compounds are molecules but not all molecules are compounds. • Molecular hydrogen (H2), mol ...
... • What is the difference between a compound and a molecule? • A molecule is formed when two or more atoms join together chemically. A compound is a molecule that contains at least two different elements. All compounds are molecules but not all molecules are compounds. • Molecular hydrogen (H2), mol ...
Document
... How are cellular reactions defined? Catabolism: breaking down of complex molecules Anabolism: the building up of complex molecules Metabolism: the sum of all these reactions ...
... How are cellular reactions defined? Catabolism: breaking down of complex molecules Anabolism: the building up of complex molecules Metabolism: the sum of all these reactions ...
Macromolecule Notes
... Enzyme = protein that facilitates the reaction (suffix ”ASE” Substrate = the item that the enzyme breaks down Product = the result of the enzyme breaking down the item Denature = causing an enzyme to not work as well or at all. Can be permanent or temporary. ...
... Enzyme = protein that facilitates the reaction (suffix ”ASE” Substrate = the item that the enzyme breaks down Product = the result of the enzyme breaking down the item Denature = causing an enzyme to not work as well or at all. Can be permanent or temporary. ...
3. What are macromolecules?
... compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form single bonds with another atom and also bond to other carbon molecules forming double, triple, or quadruple bonds. Organic compounds also contain hydrogen. Since hydrogen has only one ele ...
... compounds are made primarily of carbon. Carbon has four outer electrons and can form four bonds. Carbon can form single bonds with another atom and also bond to other carbon molecules forming double, triple, or quadruple bonds. Organic compounds also contain hydrogen. Since hydrogen has only one ele ...
biology 2402
... (Often, catabolic reactions occur in one compartment of a cell, for example, the mitochondria). (Synthetic reaction takes place in another location such as the cytosol or endoplasmic reticulum). Which reaction occur depend on which enzymes are active in a particular cell at a particular time. ATP (a ...
... (Often, catabolic reactions occur in one compartment of a cell, for example, the mitochondria). (Synthetic reaction takes place in another location such as the cytosol or endoplasmic reticulum). Which reaction occur depend on which enzymes are active in a particular cell at a particular time. ATP (a ...
1. dia
... 1.) Reaction / incubation time Initial velocity should be: P(t) graph must be linear, time has to be short enough. If incubation time ↑, time ↑ for denaturation because of high temperature or pH 2.) pH: optimum depends on the amino acid composition of the enzyme proper ionization is necessary for S ...
... 1.) Reaction / incubation time Initial velocity should be: P(t) graph must be linear, time has to be short enough. If incubation time ↑, time ↑ for denaturation because of high temperature or pH 2.) pH: optimum depends on the amino acid composition of the enzyme proper ionization is necessary for S ...
Chapter 3 Chemistry of Life Modern Biology Textbook Holt
... Proteins are affected by environmental conditions such as heat and pH. Explain why the process of cooking an egg cannot be reversed. Answer: The heat that is added to the egg changes the bonds in the proteins and other molecules that make up the egg to such a large extent that the original protein s ...
... Proteins are affected by environmental conditions such as heat and pH. Explain why the process of cooking an egg cannot be reversed. Answer: The heat that is added to the egg changes the bonds in the proteins and other molecules that make up the egg to such a large extent that the original protein s ...
2 Chem Packet
... Types of Molecules Living things need organic compounds called carbohydrates, lipids, nucleic acids, and proteins. Fill in the missing cells in the table. Identify the function of the molecule or the main components (types of atoms) that make up the molecule. ...
... Types of Molecules Living things need organic compounds called carbohydrates, lipids, nucleic acids, and proteins. Fill in the missing cells in the table. Identify the function of the molecule or the main components (types of atoms) that make up the molecule. ...
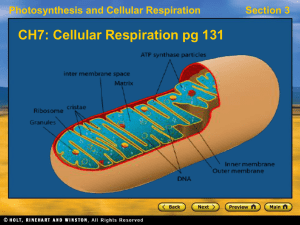
Chapter 18 Metabolic Pathways and Energy Production
... Enzyme Complex IV At enzyme complex IV electrons from cytochrome c are passed to other electron carriers until the electrons combine with hydrogen ions and oxygen (O2) to form water. ...
... Enzyme Complex IV At enzyme complex IV electrons from cytochrome c are passed to other electron carriers until the electrons combine with hydrogen ions and oxygen (O2) to form water. ...
Practice problems for chapter 1, 3 and 5 1) A small amount of salt
... 28) The SI unit for mass is __________. 29) __________ is the abbreviation for the prefix mega-. 30) Milli is the abbreviation for the prefix ________. 31) A common English set of units for expressing velocity is miles/hour. The SI unit for velocity is __________. 32) The density of a gold nugget i ...
... 28) The SI unit for mass is __________. 29) __________ is the abbreviation for the prefix mega-. 30) Milli is the abbreviation for the prefix ________. 31) A common English set of units for expressing velocity is miles/hour. The SI unit for velocity is __________. 32) The density of a gold nugget i ...
enzymes - MBBS Students Club
... acids or bases. In “specific acid or base catalysis” rate of reaction is sensitive to changes in protons , but is independent of conc of other acids or bases present in the solution or at active site. In “general acid or base catalysis” reaction rates are sensitive to all acids & bases present . ...
... acids or bases. In “specific acid or base catalysis” rate of reaction is sensitive to changes in protons , but is independent of conc of other acids or bases present in the solution or at active site. In “general acid or base catalysis” reaction rates are sensitive to all acids & bases present . ...
ENZYMES - Rihs.com.pk
... acids or bases. In “specific acid or base catalysis” rate of reaction is sensitive to changes in protons , but is independent of conc of other acids or bases present in the solution or at active site. In “general acid or base catalysis” reaction rates are sensitive to all acids & bases present . ...
... acids or bases. In “specific acid or base catalysis” rate of reaction is sensitive to changes in protons , but is independent of conc of other acids or bases present in the solution or at active site. In “general acid or base catalysis” reaction rates are sensitive to all acids & bases present . ...
Smooth endoplasmic reticulum
... 3) Chorophyll is a heme group, using a Mg ion instead of Fe or Cu, with a long fatty acid tail attached to make it soluble in the thylakoid membrane. The cytochrome P450 complexes in the membrane of the SER consist of two enzymes: (1) NADPH-cytochrome P450 reductase (2) Cytochrome P450 The reductase ...
... 3) Chorophyll is a heme group, using a Mg ion instead of Fe or Cu, with a long fatty acid tail attached to make it soluble in the thylakoid membrane. The cytochrome P450 complexes in the membrane of the SER consist of two enzymes: (1) NADPH-cytochrome P450 reductase (2) Cytochrome P450 The reductase ...
AP Chemistry Summer Study Guide
... Galvanic / Voltaic Cell: Redox reaction that is spontaneous Gamma Ray: 0 protons, mass number = 0, Low ionizing ability, high energy Halogen: Elements in group 17. Form halides as ions Hydrogen Bonding: Strong dipole that results when H is bonded to F, O, or N Indirect relationship: Relationship bet ...
... Galvanic / Voltaic Cell: Redox reaction that is spontaneous Gamma Ray: 0 protons, mass number = 0, Low ionizing ability, high energy Halogen: Elements in group 17. Form halides as ions Hydrogen Bonding: Strong dipole that results when H is bonded to F, O, or N Indirect relationship: Relationship bet ...
The Photosynthetic Dark Reactions Do Not Operate
... for the carbon reduction enzymes for several minutes in the dark, provided that the concentrations of ATP and NADPH were maintained high. When these photosynthetic substrates were depleted, the enzyme reactions stopped. Because there was some residual enzyme activity in the dark, the pathway was com ...
... for the carbon reduction enzymes for several minutes in the dark, provided that the concentrations of ATP and NADPH were maintained high. When these photosynthetic substrates were depleted, the enzyme reactions stopped. Because there was some residual enzyme activity in the dark, the pathway was com ...
Cellular Respiration
... – 2 molecules NADH are created • Important because NADH are Hydrogen ion/proton and e- carriers ...
... – 2 molecules NADH are created • Important because NADH are Hydrogen ion/proton and e- carriers ...
Macromolecules biologyjunction
... outer electrons and can form four bonds. Carbon can form single bonds with another atom and also bond to other carbon molecules forming double, triple, or quadruple bonds. Organic compounds also contain hydrogen. Since hydrogen has only one electron, it can form only single bonds. Each small organic ...
... outer electrons and can form four bonds. Carbon can form single bonds with another atom and also bond to other carbon molecules forming double, triple, or quadruple bonds. Organic compounds also contain hydrogen. Since hydrogen has only one electron, it can form only single bonds. Each small organic ...
Cellular Respiration
... – Cellular respiration yields CO2, H2O, and a large amount of ATP Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings ...
... – Cellular respiration yields CO2, H2O, and a large amount of ATP Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings ...
Learning Objectives
... Know how pyruvate dehydrogenase complex works (cofactors involved, mechanism, regulation) Know net reaction Know where ATP, CO2, FADH2 and NADH are made Know ultimate # ATP formed Stereospecificity of aconitase, fumarase ...
... Know how pyruvate dehydrogenase complex works (cofactors involved, mechanism, regulation) Know net reaction Know where ATP, CO2, FADH2 and NADH are made Know ultimate # ATP formed Stereospecificity of aconitase, fumarase ...
Medical Biochemistry
... Animal cells contain alcohol dehydrogenase (ADH) which oxidizes ethanol to acetaldehyde. Acetaldehyde is oxidized to acetate by acetaldehyde dehydrogenase (AcDH). Acetaldehyde and acetate are toxic leading to the many side effects (the hangover) that are associated with alcohol consumption. The ADH ...
... Animal cells contain alcohol dehydrogenase (ADH) which oxidizes ethanol to acetaldehyde. Acetaldehyde is oxidized to acetate by acetaldehyde dehydrogenase (AcDH). Acetaldehyde and acetate are toxic leading to the many side effects (the hangover) that are associated with alcohol consumption. The ADH ...
Review on Biochemistry: Protein Chemistry
... Desmosine (a derivative of four Lys residues): found in the fibrous protein elastin. Selenocysteine: Selenium replaces sulfur in cysteine during amino acid synthesis (derived from serine). Amino acids not as constituents of proteins, but play other cellular functions: Ornithine, citrulline: ...
... Desmosine (a derivative of four Lys residues): found in the fibrous protein elastin. Selenocysteine: Selenium replaces sulfur in cysteine during amino acid synthesis (derived from serine). Amino acids not as constituents of proteins, but play other cellular functions: Ornithine, citrulline: ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.