Flexbook - Ions and Ion Formation
... of family 3A. The large jump occurs between the 3rd and 4th ionization energies, so we know that only the first three electrons can be easily removed from this atom. The logic for the formation of anions is very similar to that for cations. A fluorine atom, for example, has a high electron affinity ...
... of family 3A. The large jump occurs between the 3rd and 4th ionization energies, so we know that only the first three electrons can be easily removed from this atom. The logic for the formation of anions is very similar to that for cations. A fluorine atom, for example, has a high electron affinity ...
BIO 16l EXAM2 SUMMER6WKKey
... 16. If a blood research laboratory is attempting to collect the content of human red blood cells, the researchers should use which of the following types of solutions to cause blood cell lysis (burstingf a. hyperosmotic b. isosmotic ...
... 16. If a blood research laboratory is attempting to collect the content of human red blood cells, the researchers should use which of the following types of solutions to cause blood cell lysis (burstingf a. hyperosmotic b. isosmotic ...
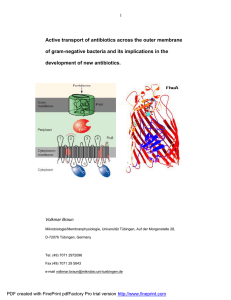
Active transport of antibiotics across the outer membrane of gram
... siderophores, which are taken up across the outer membrane by transport systems of Fe3+-siderophores. A well-studied example is albomycin, which has structural similarities to the natural substrate ferrichrome; albomycin and ferrichrome are both transported by the FhuA protein. A semisynthetic rifam ...
... siderophores, which are taken up across the outer membrane by transport systems of Fe3+-siderophores. A well-studied example is albomycin, which has structural similarities to the natural substrate ferrichrome; albomycin and ferrichrome are both transported by the FhuA protein. A semisynthetic rifam ...
Integrative Assignment - California State University
... They are possibly the most thoroughly studied organism although mice, E. coli, and Drosophila are also in the running. – Henrik Kibak, Professor of Biology at CSU Monterey Bay. Figure below is from the National Geographic Genographic Project. ...
... They are possibly the most thoroughly studied organism although mice, E. coli, and Drosophila are also in the running. – Henrik Kibak, Professor of Biology at CSU Monterey Bay. Figure below is from the National Geographic Genographic Project. ...
Haemoglobin (Roll no. 22
... • Decrease of hemoglobin, with or without an absolute decrease of red blood cells, leads to symptoms of anemia. As absence of iron decreases heme synthesis, red blood cells in iron deficiency anemia are hypochromic (lacking the red hemoglobin pigment) and microcytic (smaller than normal). • Sickle ...
... • Decrease of hemoglobin, with or without an absolute decrease of red blood cells, leads to symptoms of anemia. As absence of iron decreases heme synthesis, red blood cells in iron deficiency anemia are hypochromic (lacking the red hemoglobin pigment) and microcytic (smaller than normal). • Sickle ...
Structural Investigation of the Antibiotic and ATP
... of the polypeptide chain, their limited resolution of approximately 3 A hindered further structural investigations. The model of KNTase presented here is based on X-ray diffraction data from crystals of the site-directed mutant D80Y. These crystals diffract beyond 2.5 A. An a-carbon trace of the new ...
... of the polypeptide chain, their limited resolution of approximately 3 A hindered further structural investigations. The model of KNTase presented here is based on X-ray diffraction data from crystals of the site-directed mutant D80Y. These crystals diffract beyond 2.5 A. An a-carbon trace of the new ...
Metabolism & Enzymes
... 4 polypeptide chains can bind 4 O2; 1st O2 binds now easier for other O2 to bind AP3Biology ...
... 4 polypeptide chains can bind 4 O2; 1st O2 binds now easier for other O2 to bind AP3Biology ...
Micronutrients - Functions - University of Alaska Fairbanks
... • Carnitine is an amino acid required for the transportaion of fatty acids across mitochondrial memebranes for beta-oxidation (energy production). • Vitamin C, along with iron, are cofactors for an enzyme required for the synthesis of carnitine. Neurotransmitter Synthesis • Vitamin C is a co-factor ...
... • Carnitine is an amino acid required for the transportaion of fatty acids across mitochondrial memebranes for beta-oxidation (energy production). • Vitamin C, along with iron, are cofactors for an enzyme required for the synthesis of carnitine. Neurotransmitter Synthesis • Vitamin C is a co-factor ...
Pyruvate dehydrogenase
... pathway occurring in plants and several bacteria, but not animals. . The glyoxylate cycle allows these organisms to use fats for the synthesis of carbohydrates, a task which vertebrates, including humans, cannot perform. Isocitrate --> succinate + glyoxylate (O=CH-COO-)+acetyl-CoA--> malate-->> gluc ...
... pathway occurring in plants and several bacteria, but not animals. . The glyoxylate cycle allows these organisms to use fats for the synthesis of carbohydrates, a task which vertebrates, including humans, cannot perform. Isocitrate --> succinate + glyoxylate (O=CH-COO-)+acetyl-CoA--> malate-->> gluc ...
One of the most famous examples
... the smaller proteins. So larger molecules come out (elute) first, and the smallest come out last. Here again, you would collect the eluted proteins in a series of tubes, and then assay each tube for the presence of the protein being purified. If you calibrate the column by noting the behavior of sph ...
... the smaller proteins. So larger molecules come out (elute) first, and the smallest come out last. Here again, you would collect the eluted proteins in a series of tubes, and then assay each tube for the presence of the protein being purified. If you calibrate the column by noting the behavior of sph ...
ClickThisLinkForEntries
... Cofactors are nonproteins assisting the process of catalytic activity. They can be inorganic or organic; examples are vitamins. They can either bind tightly and permanently (covalently) to enzymes or loosely and reversibly to substrates with weaker bonds; either way of interacting with these two wil ...
... Cofactors are nonproteins assisting the process of catalytic activity. They can be inorganic or organic; examples are vitamins. They can either bind tightly and permanently (covalently) to enzymes or loosely and reversibly to substrates with weaker bonds; either way of interacting with these two wil ...
What is Cellular Respiration?
... The carbon atoms of the sugar molecule are released as carbon dioxide (CO2). The complete breakdown of glucose to carbon dioxide and water requires two major steps: 1) glycolysis and 2) aerobic respiration. Glycolysis produces two ATP. Thirty-four more ATP are produced by aerobic pathways if oxygen ...
... The carbon atoms of the sugar molecule are released as carbon dioxide (CO2). The complete breakdown of glucose to carbon dioxide and water requires two major steps: 1) glycolysis and 2) aerobic respiration. Glycolysis produces two ATP. Thirty-four more ATP are produced by aerobic pathways if oxygen ...
Regeneration of NAD+ Lactic Acid Fermentation
... • Acetaldehyde is formed from pyruvate by decarboxylation. • Pyruvate decarboxylase has thiamine pyrophosphate as a prosthetic group. • Overall reaction becomes: Glucose + 2 ADP + 2 Pi → 2 ethanol + 2 CO 2 + 2 ATP ...
... • Acetaldehyde is formed from pyruvate by decarboxylation. • Pyruvate decarboxylase has thiamine pyrophosphate as a prosthetic group. • Overall reaction becomes: Glucose + 2 ADP + 2 Pi → 2 ethanol + 2 CO 2 + 2 ATP ...
Test 1 Study Guide Chapter 1 – Introduction
... 3. Electrons accepted by various molecules h. Balance sheet: 38 ATP (34 from 10 NAD and 2 FAD) (Tab 5.3) D. Fermentation – a “shortcut” respiration process. It just regenerates NAD+ to run glycolysis. This produces ATP by substrate level phosphorylation only. Inefficient but very fast and no oxygen ...
... 3. Electrons accepted by various molecules h. Balance sheet: 38 ATP (34 from 10 NAD and 2 FAD) (Tab 5.3) D. Fermentation – a “shortcut” respiration process. It just regenerates NAD+ to run glycolysis. This produces ATP by substrate level phosphorylation only. Inefficient but very fast and no oxygen ...
Test 1 Study Guide
... 3. Electrons accepted by various molecules h. Balance sheet: 38 ATP (34 from 10 NAD and 2 FAD) (Tab 5.3) D. Fermentation – a “shortcut” respiration process. It just regenerates NAD+ to run glycolysis. This produces ATP by substrate level phosphorylation only. Inefficient but very fast and no oxygen ...
... 3. Electrons accepted by various molecules h. Balance sheet: 38 ATP (34 from 10 NAD and 2 FAD) (Tab 5.3) D. Fermentation – a “shortcut” respiration process. It just regenerates NAD+ to run glycolysis. This produces ATP by substrate level phosphorylation only. Inefficient but very fast and no oxygen ...
File - Groby Bio Page
... hydrogen carrier molecules Outline the Krebs cycle, in the Krebs cycle including the roles of NAD and FAD, and substrate-level phosphorylation (Grade A-B) ...
... hydrogen carrier molecules Outline the Krebs cycle, in the Krebs cycle including the roles of NAD and FAD, and substrate-level phosphorylation (Grade A-B) ...
Fatty Acid Synthesis Chapter 28, Stryer Short Course
... receptors in non-liver cells • HDLs are “good cholesterol” ...
... receptors in non-liver cells • HDLs are “good cholesterol” ...
Fermentation PowerPoint File
... Fermentation Under anaerobic conditions, fermentation follows glycolysis. During fermentation, cells convert NADH produced by glycolysis back into the electron carrier NAD+, which allows glycolysis to continue producing ATP. ...
... Fermentation Under anaerobic conditions, fermentation follows glycolysis. During fermentation, cells convert NADH produced by glycolysis back into the electron carrier NAD+, which allows glycolysis to continue producing ATP. ...
basic components of living things
... i. Contain esther bonds between glycerol and fatty acid ii.They give higher energy than carbohydrates and proteins iii. They have double bonds in fatty acids Which of the statements above is not a property of solid lipids? ...
... i. Contain esther bonds between glycerol and fatty acid ii.They give higher energy than carbohydrates and proteins iii. They have double bonds in fatty acids Which of the statements above is not a property of solid lipids? ...
STRUCTURE AND FUNCTION
... sufficient amount of carbon compounds and energy to initiate biosynthesis, so it allows the necessary ammonia to enter the cell to be incorporated into proteins and nucleic acids. The model presented and supported by the experimental data suggests that GlnK1 is an important molecule in the regulatio ...
... sufficient amount of carbon compounds and energy to initiate biosynthesis, so it allows the necessary ammonia to enter the cell to be incorporated into proteins and nucleic acids. The model presented and supported by the experimental data suggests that GlnK1 is an important molecule in the regulatio ...
Model Description Sheet
... Antibiotic-resistant bacteria are common and hard to treat. There is potential to create synthetic antibiotics based on natural products like enduracidin and mannopeptimycin to fight drug resistant bacteria like MRSA. MppP, an enzyme from Streptomyces wadayamensis, is required for the biosynthesis o ...
... Antibiotic-resistant bacteria are common and hard to treat. There is potential to create synthetic antibiotics based on natural products like enduracidin and mannopeptimycin to fight drug resistant bacteria like MRSA. MppP, an enzyme from Streptomyces wadayamensis, is required for the biosynthesis o ...
Polyphosphate-ATP Amplification Technology: Principle and its
... Using computer simulation, Chittock et al. also previously proposed an ATP amplification to enhance the sensitivity of ATP detection2), but it has been very difficult to apply this idea because very small amounts of ATP contamination must be eliminated from the reaction mixture before examination. If ...
... Using computer simulation, Chittock et al. also previously proposed an ATP amplification to enhance the sensitivity of ATP detection2), but it has been very difficult to apply this idea because very small amounts of ATP contamination must be eliminated from the reaction mixture before examination. If ...
Nadine Noelting
... PLLEKYCGFHEDNIPQLEDVSQFLQTCTGFRLRPVAGLLSSRDFLGGLAFRVFHCTQYIRHGSKPMYTPE PDICHELLGHVPLFSDRSFAQFSQEIGLASLGAPDEYIEKLATIYWFTVEFGLCKQGDSIKAYGAGLLSS FGELQYCLSEKPKLLPLELEKTAIQNYTVTEFQPLYYVAESFNDAKEKVRNFAATIPRPFSVRYDPYTQR IEVLDNTQQLKILADSINSEIGILCSALQKIK ...
... PLLEKYCGFHEDNIPQLEDVSQFLQTCTGFRLRPVAGLLSSRDFLGGLAFRVFHCTQYIRHGSKPMYTPE PDICHELLGHVPLFSDRSFAQFSQEIGLASLGAPDEYIEKLATIYWFTVEFGLCKQGDSIKAYGAGLLSS FGELQYCLSEKPKLLPLELEKTAIQNYTVTEFQPLYYVAESFNDAKEKVRNFAATIPRPFSVRYDPYTQR IEVLDNTQQLKILADSINSEIGILCSALQKIK ...
Roberts, LM Dept. of Chemistry California State
... Meaning and use of reduction potentials PDH and dehydrogenase complex reaction in general Know the difference between alpha and beta-keto acid decarboxylations Be able to predict regulation of the TCA cycle Understand the basic concepts of the glyoxylate cycle (two unique reactions, 4 carbons feed i ...
... Meaning and use of reduction potentials PDH and dehydrogenase complex reaction in general Know the difference between alpha and beta-keto acid decarboxylations Be able to predict regulation of the TCA cycle Understand the basic concepts of the glyoxylate cycle (two unique reactions, 4 carbons feed i ...
Kinetic mechanism of the dimeric ATP sulfurylase from plants
... (potato) and Glycine max (soybean) and share >60 % amino acid sequence identity [4,17,18,20,22]. For example, the soybean genome contains four ATP sulfurylase genes (Glyma10g38760, Glyma20g28980, Glyma13g06940 and Glyma19g05020) encoding proteins with predicted plastidial and/or mitochondrial locali ...
... (potato) and Glycine max (soybean) and share >60 % amino acid sequence identity [4,17,18,20,22]. For example, the soybean genome contains four ATP sulfurylase genes (Glyma10g38760, Glyma20g28980, Glyma13g06940 and Glyma19g05020) encoding proteins with predicted plastidial and/or mitochondrial locali ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.