Spin Quantum Number - stpats-sch3u-sem1-2013
... Electrons are found in atomic orbitals, or atomic space, surrounding the nucleus of an atom. For a neutral atom, the number of electrons in the element is equal to the atomic number. For example, nitrogen’s atomic number is 7 and so, an atom of nitrogen has 7 electrons. However, these 7 electrons do ...
... Electrons are found in atomic orbitals, or atomic space, surrounding the nucleus of an atom. For a neutral atom, the number of electrons in the element is equal to the atomic number. For example, nitrogen’s atomic number is 7 and so, an atom of nitrogen has 7 electrons. However, these 7 electrons do ...
Electronic structure (download)
... Define atomic orbitals Distinguish between the Bohr orbit and atomic orbital Apply quantum numbers and atomic orbitals to building atoms and the periodic table Describe periodic trends in terms of electronic ...
... Define atomic orbitals Distinguish between the Bohr orbit and atomic orbital Apply quantum numbers and atomic orbitals to building atoms and the periodic table Describe periodic trends in terms of electronic ...
Quantum Mechanical Model of the Atom
... The Compton Effect: When a high energy xray photon collides with a “free electron”, it gives some of its energy to the electron and a lower energy photon scatters off the electron. ...
... The Compton Effect: When a high energy xray photon collides with a “free electron”, it gives some of its energy to the electron and a lower energy photon scatters off the electron. ...
Electrons in the Atom
... Think of orbitals as sort of a "border” for spaces around the nucleus inside which electrons are allowed. No more than 2 electrons can ever be in 1 orbital. The orbital just defines an “area” where you can find an electron. ...
... Think of orbitals as sort of a "border” for spaces around the nucleus inside which electrons are allowed. No more than 2 electrons can ever be in 1 orbital. The orbital just defines an “area” where you can find an electron. ...
Mr. Knittel`s Final Review Sheet I Answers
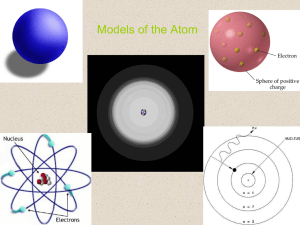
... the original model to our current understanding of the atom. In Dalton’s atomic theory atoms were seen as indivisible pieces of matter, and were likened to billiard balls (hard, compact, spheres). J. J. Thomson’s work with cathode ray tubes led him to the discovery of the electron—a subatomic partic ...
... the original model to our current understanding of the atom. In Dalton’s atomic theory atoms were seen as indivisible pieces of matter, and were likened to billiard balls (hard, compact, spheres). J. J. Thomson’s work with cathode ray tubes led him to the discovery of the electron—a subatomic partic ...
Exam 1
... ms - spin quantum number; relationship to electron spin Determining possible values for n, , m, and ms n = 1, 2, 3, … Examples: If n = 3, = 0, 1, 2 = 0, 1, 2, … (n-1) If = 2, m = -2, -1, 0, +1, +2 m = -, …, + ms = - 1/2, +1/2 Electron configuration for atoms Pauli principle Aufbau princi ...
... ms - spin quantum number; relationship to electron spin Determining possible values for n, , m, and ms n = 1, 2, 3, … Examples: If n = 3, = 0, 1, 2 = 0, 1, 2, … (n-1) If = 2, m = -2, -1, 0, +1, +2 m = -, …, + ms = - 1/2, +1/2 Electron configuration for atoms Pauli principle Aufbau princi ...
Atomic Radius and Ionization Energy
... Trend in Atomic Radius • Measure the molecule that forms when two atoms of the same element combine • Atomic radius = half of the distance between the nuclei of the two atoms • Measured in picometers (1 pm = 10-12 m) ...
... Trend in Atomic Radius • Measure the molecule that forms when two atoms of the same element combine • Atomic radius = half of the distance between the nuclei of the two atoms • Measured in picometers (1 pm = 10-12 m) ...
Chpater 5.3 PPT
... due to increased nuclear charge. Ionization energies decrease down a group due to further distance from nucleus and electron ...
... due to increased nuclear charge. Ionization energies decrease down a group due to further distance from nucleus and electron ...
Quarter Exam (Old Test)
... ____ 36. How many unpaired electrons are in a phosphorus atom (atomic number 15)? Show the work the for the orbital notation (or arrow diagram) for this element. It should help you answer this question. a. 1 c. 3 b. 2 d. 0 ____ 37. Which of the following items is a mixture (NOT a compound)? a. table ...
... ____ 36. How many unpaired electrons are in a phosphorus atom (atomic number 15)? Show the work the for the orbital notation (or arrow diagram) for this element. It should help you answer this question. a. 1 c. 3 b. 2 d. 0 ____ 37. Which of the following items is a mixture (NOT a compound)? a. table ...
The Bohr model for the electrons
... Define atomic orbitals Distinguish between the Bohr orbit and atomic orbital Apply quantum numbers and atomic orbitals to building atoms and the periodic table Describe periodic trends in terms of electronic ...
... Define atomic orbitals Distinguish between the Bohr orbit and atomic orbital Apply quantum numbers and atomic orbitals to building atoms and the periodic table Describe periodic trends in terms of electronic ...
Chapter 3 Study Guide
... a. Thomson- discovered the electron by experimenting with cathode ray tubes. i. Used a magnet to determine the charge on the beam inside the tube. ii. Proposed the ‘plum pudding’ model (see notes for picture and description) of the atom. b. Rutherford- discovered the positively charged proton in the ...
... a. Thomson- discovered the electron by experimenting with cathode ray tubes. i. Used a magnet to determine the charge on the beam inside the tube. ii. Proposed the ‘plum pudding’ model (see notes for picture and description) of the atom. b. Rutherford- discovered the positively charged proton in the ...
Vocabulary Terms Defined
... quantum (93) the minimum quantity of energy that can be lost or gained by an atom (plural: quanta) A quantum is the minimum amount of any physical entity involved in an interaction. continuous spectrum (94) the emission of a continuous range of frequencies of electromagnetic radiation excited state ...
... quantum (93) the minimum quantity of energy that can be lost or gained by an atom (plural: quanta) A quantum is the minimum amount of any physical entity involved in an interaction. continuous spectrum (94) the emission of a continuous range of frequencies of electromagnetic radiation excited state ...
Document
... Rule 1. Electron can exist in stationary states; requires fixed energy. Rule 2. Possible stationary states determined by quantization of angular momentum (mevr) in units of h/2π (mevr = nh/2π) Rule 3. Transitions between stationary states occur with emission or absorption of a quantum of energy, ΔE ...
... Rule 1. Electron can exist in stationary states; requires fixed energy. Rule 2. Possible stationary states determined by quantization of angular momentum (mevr) in units of h/2π (mevr = nh/2π) Rule 3. Transitions between stationary states occur with emission or absorption of a quantum of energy, ΔE ...
08_lecture_ppt
... Electronic Quantum Numbers in Atoms 1. Principle quantum number, n – Energy level – Average distance from nucleus ...
... Electronic Quantum Numbers in Atoms 1. Principle quantum number, n – Energy level – Average distance from nucleus ...
08_lecture_ppt - Chemistry at Winthrop University
... Electronic Quantum Numbers in Atoms 1. Principle quantum number, n – Energy level – Average distance from nucleus ...
... Electronic Quantum Numbers in Atoms 1. Principle quantum number, n – Energy level – Average distance from nucleus ...
Electronic structure_(download)
... Atomic number dictates how many are filled – how many electrons are added Filling orbitals follows a fixed pattern: lowest energy ones first But need to know... how many electrons in an orbital? ...
... Atomic number dictates how many are filled – how many electrons are added Filling orbitals follows a fixed pattern: lowest energy ones first But need to know... how many electrons in an orbital? ...
Chem 150 Problem Set Introductory Quantum Chemistry 1
... hydrogen atom, the most probable distance of the electron from the nucleus is 0.53 x 10 -10 m. Why are these two statements different? b) Why is Schrődinger’s wave equation to describe the location of a particle very different from the description obtained from classical physics? c) In the quantum m ...
... hydrogen atom, the most probable distance of the electron from the nucleus is 0.53 x 10 -10 m. Why are these two statements different? b) Why is Schrődinger’s wave equation to describe the location of a particle very different from the description obtained from classical physics? c) In the quantum m ...
5. Quantum mechanics of chemical binding
... • E(R) potential energy surface is a consequence of the Born-Oppenheimer approximation; ...
... • E(R) potential energy surface is a consequence of the Born-Oppenheimer approximation; ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.