Essential Question: What is the current model of the atom? How
... Hund’s rule Heisenberg Uncertainty Prin Orbital notation d orbitals Atomic number Protons ...
... Hund’s rule Heisenberg Uncertainty Prin Orbital notation d orbitals Atomic number Protons ...
Unit 1, Lecture 1
... In order to arrive at the ground state, the lowest energy state, it is necessary to follow some simple rules: Electrons occupy the lowest energy level first (also called the Aufbau Principle) No more than one alpha (or spin up) and one beta (or spin down) electron can occupy a particular orbital at ...
... In order to arrive at the ground state, the lowest energy state, it is necessary to follow some simple rules: Electrons occupy the lowest energy level first (also called the Aufbau Principle) No more than one alpha (or spin up) and one beta (or spin down) electron can occupy a particular orbital at ...
Trends in the periodic table - Brigham Young University
... along the table? • How does this affect the radius? ...
... along the table? • How does this affect the radius? ...
PHYSICS 215 - Thermodynamics and Modern Physics Name:
... Anomalous Zeeman Effect: VB = µBBgmj where g = Landé g-factor = 1 + J(J+1)+S(S+1)-L(L+1) ...
... Anomalous Zeeman Effect: VB = µBBgmj where g = Landé g-factor = 1 + J(J+1)+S(S+1)-L(L+1) ...
Quantum Numbers and Electron Configurations Worksheet
... ml = magnetic quantum number = specifies the orientation of the orbital ml = 0 and +/- l…..so if l = 3, ml = -3, -2, -1, 0, 1, 2, and 3 (this really means is that there are seven possible orientations of the f orbital…so don’t get too hung up on -3, -2, etc…they are abstract…the total # of ml values ...
... ml = magnetic quantum number = specifies the orientation of the orbital ml = 0 and +/- l…..so if l = 3, ml = -3, -2, -1, 0, 1, 2, and 3 (this really means is that there are seven possible orientations of the f orbital…so don’t get too hung up on -3, -2, etc…they are abstract…the total # of ml values ...
Chapter 7_01042016
... • The atomic radii decrease in going from left to right across a period because of the increasing effective nuclear charge. • The valence electrons are drawn closer to the nucleus, decreasing the size of the atom. • Atomic radius increases down a group, because of the increases in the orbital size ...
... • The atomic radii decrease in going from left to right across a period because of the increasing effective nuclear charge. • The valence electrons are drawn closer to the nucleus, decreasing the size of the atom. • Atomic radius increases down a group, because of the increases in the orbital size ...
Atomic Structure and Quantum Theory
... Planck and Blackbody Radiation Einstein and the Photoelectric Effect Spectra Quantization ...
... Planck and Blackbody Radiation Einstein and the Photoelectric Effect Spectra Quantization ...
File
... Electronegativity – ability of an atom to attract an electron TIME AMOUNT Nuclear Chemistry ...
... Electronegativity – ability of an atom to attract an electron TIME AMOUNT Nuclear Chemistry ...
الرقم الجامعي
... an atom. Use the examples of Li versus F atoms to show the relative differences in the 2s and 2p levels in these atoms. ------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------ ...
... an atom. Use the examples of Li versus F atoms to show the relative differences in the 2s and 2p levels in these atoms. ------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------ ...
Notes
... before going onto a different type of room. 4. When filling rooms on a floor, you must place one student in each type of room before pairing them. ...
... before going onto a different type of room. 4. When filling rooms on a floor, you must place one student in each type of room before pairing them. ...
topic 1 sol review homework
... all are diatomics, all have 7 valence electrons, all are halogens 9. The increase in atomic radius of each successive element within a group is due to an increase in the number of a) neutrons b) valence electrons c) unpaired electrons d) principle energy levels 10. According to the modern periodic t ...
... all are diatomics, all have 7 valence electrons, all are halogens 9. The increase in atomic radius of each successive element within a group is due to an increase in the number of a) neutrons b) valence electrons c) unpaired electrons d) principle energy levels 10. According to the modern periodic t ...
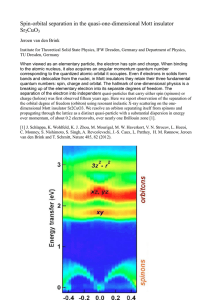
Spin-orbital separation in the quasi-one
... When viewed as an elementary particle, the electron has spin and charge. When binding to the atomic nucleus, it also acquires an angular momentum quantum number corresponding to the quantized atomic orbital it occupies. Even if electrons in solids form bands and delocalize from the nuclei, in Mott i ...
... When viewed as an elementary particle, the electron has spin and charge. When binding to the atomic nucleus, it also acquires an angular momentum quantum number corresponding to the quantized atomic orbital it occupies. Even if electrons in solids form bands and delocalize from the nuclei, in Mott i ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.