The Periodic table
... A region of space within an electron subshell where an electron with a specific energy is most likely to be found. S subshell=1 orbital, p subshell=3 orbitals, d subshell=5 orbitals, f subshell=7 orbitals. Maximum number of electrons in a subshell is always 2. S orbital=spherical, p orbital ...
... A region of space within an electron subshell where an electron with a specific energy is most likely to be found. S subshell=1 orbital, p subshell=3 orbitals, d subshell=5 orbitals, f subshell=7 orbitals. Maximum number of electrons in a subshell is always 2. S orbital=spherical, p orbital ...
Lecture 1.6 PowerPoint
... what each of these numbers indicate and discuss the importance of these numbers. • 1.7 – I can describe the shape, number, and energy level of the s, p, d, and f orbitals. Furthermore, I can draw the s and p orbitals. • 1.8 – I can write the electron configuration and orbital diagram for any element ...
... what each of these numbers indicate and discuss the importance of these numbers. • 1.7 – I can describe the shape, number, and energy level of the s, p, d, and f orbitals. Furthermore, I can draw the s and p orbitals. • 1.8 – I can write the electron configuration and orbital diagram for any element ...
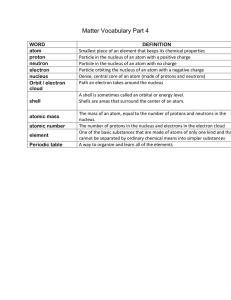
Matter Vocab Part 4
... Particle in the nucleus of an atom with a positive charge Particle in the nucleus of an atom with no charge Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes cal ...
... Particle in the nucleus of an atom with a positive charge Particle in the nucleus of an atom with no charge Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes cal ...
Modern physics 2330
... 6- ( ) Millikan’s oil drop experiment is inaccurate because it is difficult to measure the speed of drop. 7- (..) Thompson cloud chamber experiment is inaccurate because the theory applies only to a single particle, and the particles are all assumed to be identical. 8- ( ) The number, strength, and ...
... 6- ( ) Millikan’s oil drop experiment is inaccurate because it is difficult to measure the speed of drop. 7- (..) Thompson cloud chamber experiment is inaccurate because the theory applies only to a single particle, and the particles are all assumed to be identical. 8- ( ) The number, strength, and ...
Atoms and the Periodic Table
... The smallest unit of an element Atoms are made of even smaller subatomic particles: protons, neutrons, and electrons. Protons carry a positive charge, electrons carry a negative charge and neutrons are ...
... The smallest unit of an element Atoms are made of even smaller subatomic particles: protons, neutrons, and electrons. Protons carry a positive charge, electrons carry a negative charge and neutrons are ...
Energy and Matter - Hicksville Public Schools
... atoms by treating the electron as a wave and by including the idea of quantized energies. Electrons occupy atomic orbitals. An orbital is a region in the space around the atom’s nucleus where an electron with a particular amount of energy can be found 90% of the time. An orbital may occupy up to two ...
... atoms by treating the electron as a wave and by including the idea of quantized energies. Electrons occupy atomic orbitals. An orbital is a region in the space around the atom’s nucleus where an electron with a particular amount of energy can be found 90% of the time. An orbital may occupy up to two ...
Chp7,Quantum_Num
... Orbital Energies and Electron Configurations of Multi-Electron Atoms For the H atom the orbital energy depends only on n, so all orbitals with the same value of n have the same energy. This is not true, however, for any other atom! The H atom orbitals may be used to approximate the orbitals for mult ...
... Orbital Energies and Electron Configurations of Multi-Electron Atoms For the H atom the orbital energy depends only on n, so all orbitals with the same value of n have the same energy. This is not true, however, for any other atom! The H atom orbitals may be used to approximate the orbitals for mult ...
Fall Exam 3
... Orbital energies increase in the order 3d < 3p < 3s because the effective nuclear charge experienced by an electron increases in the order 3s < 3p < 3d. 3s, 3p and 3d orbitals are energetically degenerate because orbital energy depends only on the principal quantum number, n. ...
... Orbital energies increase in the order 3d < 3p < 3s because the effective nuclear charge experienced by an electron increases in the order 3s < 3p < 3d. 3s, 3p and 3d orbitals are energetically degenerate because orbital energy depends only on the principal quantum number, n. ...
Chem20u2(5.2) - Mr. Searcy Chemistry 20
... Heisenberg uncertainty principle on the modern view of electrons in atoms. 5. Identify the relationships among a hydrogen atom’s energy levels, sublevels, and atomic orbitals. II. The following questions will help to cover these objectives as you read through the section. Briefly answer each questio ...
... Heisenberg uncertainty principle on the modern view of electrons in atoms. 5. Identify the relationships among a hydrogen atom’s energy levels, sublevels, and atomic orbitals. II. The following questions will help to cover these objectives as you read through the section. Briefly answer each questio ...
Periodic Trends
... remove an electron from a gaseous atom. The energy required to remove the first electron is called the first ionization energy. ...
... remove an electron from a gaseous atom. The energy required to remove the first electron is called the first ionization energy. ...
Constructive Interference
... exist in stable configurations around nuclei Wavefunctions and energies for these configurations determine most properties of matter ...
... exist in stable configurations around nuclei Wavefunctions and energies for these configurations determine most properties of matter ...
CHEM1611 Worksheet 2: Atomic Accountancy Model 1: Atomic
... ml: the magnetic orbital quantum number identifies the subshell and the orientation of the orbital For each value of l, it has values from l…0…-l ms: the spin quantum number which describes the spin of the electron. It has values of +½ or -½ which are sometimes called ‘spin up’ and ‘spin down’ respe ...
... ml: the magnetic orbital quantum number identifies the subshell and the orientation of the orbital For each value of l, it has values from l…0…-l ms: the spin quantum number which describes the spin of the electron. It has values of +½ or -½ which are sometimes called ‘spin up’ and ‘spin down’ respe ...
Section 13.2 - CPO Science
... around you come from this property of elements to emit or absorb light of only certain colors. ...
... around you come from this property of elements to emit or absorb light of only certain colors. ...
key - gcisd
... 11. Bohr - His atomic model (planetary model) had atoms built up of successive orbital shells of electrons. Electrons move between shells based on specific amounts of energy being lost or gained. 12. Electron Cloud Model - This model is based on quantum mechanics which states that you cannot know th ...
... 11. Bohr - His atomic model (planetary model) had atoms built up of successive orbital shells of electrons. Electrons move between shells based on specific amounts of energy being lost or gained. 12. Electron Cloud Model - This model is based on quantum mechanics which states that you cannot know th ...
Review Sheet Filled Out
... List the number of facts you know about electrons. Electrons closest to the nucleus have the least amount of energy Electrons farthest away from the nucleus have the most energy – valence e Have a negative charge Have insignificant mass and volume Reside in the 99.996% of the atom outside t ...
... List the number of facts you know about electrons. Electrons closest to the nucleus have the least amount of energy Electrons farthest away from the nucleus have the most energy – valence e Have a negative charge Have insignificant mass and volume Reside in the 99.996% of the atom outside t ...
Chapter 4: Electrons in Atoms I. Properties of Light A
... confined to the space around an atomic nucleus. 2. According to the relationship E = hν, these frequencies corresponded to specific ________________—the quantized energies of Bohr’s orbits. B. Heisenberg Uncertainty Principle 1. German physicist Werner Heisenberg proposed that any attempt to locate ...
... confined to the space around an atomic nucleus. 2. According to the relationship E = hν, these frequencies corresponded to specific ________________—the quantized energies of Bohr’s orbits. B. Heisenberg Uncertainty Principle 1. German physicist Werner Heisenberg proposed that any attempt to locate ...
Smallest sliver of time yet measured sees electrons
... The ejections took between 7 and 20 attoseconds, according to Schultze, depending on how the electron interacted with the nucleus and the other electron. “We only need to know the time centre of the pulses very precisely, and our technique allows us to measure this to very high precision,” Ossiander ...
... The ejections took between 7 and 20 attoseconds, according to Schultze, depending on how the electron interacted with the nucleus and the other electron. “We only need to know the time centre of the pulses very precisely, and our technique allows us to measure this to very high precision,” Ossiander ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.