Quantum Mechanical Model
... Integral values from l to -l (including 0) Related to the orientation of the orbital in space relative to the other orbitals ...
... Integral values from l to -l (including 0) Related to the orientation of the orbital in space relative to the other orbitals ...
Name: ______ Date: Period: ______ Review of Bohr`s Atomic Model
... To review the placement of electrons within an atom using Bohr’s model. To become familiar with the basic terminology and concepts concerning the current quantum mechanical model of the atom. Bohr Model of the Atom ...
... To review the placement of electrons within an atom using Bohr’s model. To become familiar with the basic terminology and concepts concerning the current quantum mechanical model of the atom. Bohr Model of the Atom ...
Bohr Model of the Hydrogen Atom
... (c) What is the energy of the ground state, measured in eV (electron volts)? As you likely know from high school physics and/or chemistry courses, the Bohr model predicts the energies of the hydrogen atom incredibly accurately: in fact, not until the introduction of relativistic quantum mechanics wa ...
... (c) What is the energy of the ground state, measured in eV (electron volts)? As you likely know from high school physics and/or chemistry courses, the Bohr model predicts the energies of the hydrogen atom incredibly accurately: in fact, not until the introduction of relativistic quantum mechanics wa ...
Chem 2 AP Ch 7 MC Review Key
... B) No, fluorescent materials only emit purple and green visible light. C) Yes, fluorescent materials emit a broad spectrum of light. D) Yes, after storing enough visible light energy, the fluorescent material can emit ultraviolet light. ...
... B) No, fluorescent materials only emit purple and green visible light. C) Yes, fluorescent materials emit a broad spectrum of light. D) Yes, after storing enough visible light energy, the fluorescent material can emit ultraviolet light. ...
Chapter 4
... Orientation of orbital around nucleus Symbol – m s–1 p–3 d–5 f–7 Every orientation can hold 2 e-!! Figures 4-13, 4-14, 4-15 on page 102-103 ...
... Orientation of orbital around nucleus Symbol – m s–1 p–3 d–5 f–7 Every orientation can hold 2 e-!! Figures 4-13, 4-14, 4-15 on page 102-103 ...
Physical Science
... Naming Ionic Compounds – roman numeral for multiple oxidation number cations, name to formula and formula to name Metallic Bonds and Properties of Metals – electron sea model, delocalized electrons, metal properties, alloys (substitutional/interstitial) What is a covalent bond – shared electro ...
... Naming Ionic Compounds – roman numeral for multiple oxidation number cations, name to formula and formula to name Metallic Bonds and Properties of Metals – electron sea model, delocalized electrons, metal properties, alloys (substitutional/interstitial) What is a covalent bond – shared electro ...
Quantum Numbe
... 14. The sublevels are given letters “s,p,d,f” 15. Sublevels of one energy level can overlap those of another 16. Each sublevel holds a maximum number of pairs of electrons: s-1; p-3; d-5; f-7 17. Also describes the probability shape of an orbital ...
... 14. The sublevels are given letters “s,p,d,f” 15. Sublevels of one energy level can overlap those of another 16. Each sublevel holds a maximum number of pairs of electrons: s-1; p-3; d-5; f-7 17. Also describes the probability shape of an orbital ...
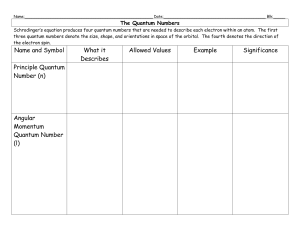
e-the-quantum-numberssv-2
... The Quantum Numbers Schrodinger’s equation produces four quantum numbers that are needed to describe each electron within an atom. The first three quantum numbers denote the size, shape, and orientations in space of the orbital. The fourth denotes the direction of the electron spin. ...
... The Quantum Numbers Schrodinger’s equation produces four quantum numbers that are needed to describe each electron within an atom. The first three quantum numbers denote the size, shape, and orientations in space of the orbital. The fourth denotes the direction of the electron spin. ...
topic-2.doc
... Electrons are in orbit around the nucleus, are involved in chemical reactions. o Orbital: three-dimensional space where an electron will most likely be found 90% of the time o First energy level: one s orbital, holds 2 electrons o Second energy level: one s and three p orbitals, holds 8 electrons Ch ...
... Electrons are in orbit around the nucleus, are involved in chemical reactions. o Orbital: three-dimensional space where an electron will most likely be found 90% of the time o First energy level: one s orbital, holds 2 electrons o Second energy level: one s and three p orbitals, holds 8 electrons Ch ...
Bohr`s equation for the hydrogen atom Bohr derived an equation to
... it is normal to write the orbital quantum number as l but we are using L to make it clearly different from1.) (d) the spin quantum number (s), which may have values of + ½ or – ½ . Related to these quantum numbers is the Pauli exclusion principle which states that no two electrons in an atom may exi ...
... it is normal to write the orbital quantum number as l but we are using L to make it clearly different from1.) (d) the spin quantum number (s), which may have values of + ½ or – ½ . Related to these quantum numbers is the Pauli exclusion principle which states that no two electrons in an atom may exi ...
Lesson 9 Core notation File
... values of the spin quantum number that describe the electron. Quantum theory was able to explain the experimental results if the spin quantum number was taken to be either +1/2 or -1/2. Question: Why should you care which way electrons are spinning? Answer: Spin determines how many electrons can occ ...
... values of the spin quantum number that describe the electron. Quantum theory was able to explain the experimental results if the spin quantum number was taken to be either +1/2 or -1/2. Question: Why should you care which way electrons are spinning? Answer: Spin determines how many electrons can occ ...
Chemistry - chem.uwec.edu
... Write a set of quantum numbers for the third electron and a set for the eighth electron of the F atom. ...
... Write a set of quantum numbers for the third electron and a set for the eighth electron of the F atom. ...
... I 10. (1 0) The decay between two excited states of the nucle~isof 4 ' ~ iemits gamma ray of 1.3117 MeV. Tht luppe, state has a lifetime of 1.4ps, the lower state 3.0 ps. A) What is the fractional uncertainty AEIE in tht energy of the gainma ray? B) What is the percentage spread in wavelength of the ...
Chapter 4 Section 2
... particular path have a fixed energy, they do NOT lose energy and fall into the nucleus Energy level—region around nucleus where it is likely to be moving, similar to rungs on a ladder but not equally spaced Quantum—amount of energy needed to move an electron from its current energy level to the next ...
... particular path have a fixed energy, they do NOT lose energy and fall into the nucleus Energy level—region around nucleus where it is likely to be moving, similar to rungs on a ladder but not equally spaced Quantum—amount of energy needed to move an electron from its current energy level to the next ...
Physics 2 Homework 23_2013 We started discussing
... conditions can be of the four shapes which are denoted by the characters: s, p, d, f. Historically, these notations came from the words: sharp, principal, diffusive, fundamental. These shapes are shown below: ...
... conditions can be of the four shapes which are denoted by the characters: s, p, d, f. Historically, these notations came from the words: sharp, principal, diffusive, fundamental. These shapes are shown below: ...
study note 1 06
... The part that oscillates between crest and trough and nodes (fixed). What is the restriction placed on the wavelength of standing The only waves that can occur are those for which a half wavelength waves? can be repeated exactly a whole number of times. > Express this restriction as an equation. L = ...
... The part that oscillates between crest and trough and nodes (fixed). What is the restriction placed on the wavelength of standing The only waves that can occur are those for which a half wavelength waves? can be repeated exactly a whole number of times. > Express this restriction as an equation. L = ...
Test 4
... Chapter 8 1) Define, identify and/or give examples of: electron configuration, Aufbau Principle, Hund’s Rule, Pauli Exclusion Principle, ground state, excited state, degenerate orbital, shielding, effective nuclear charge, valence electrons, valence shell, s, p, d, & f block, atomic radius, periodic ...
... Chapter 8 1) Define, identify and/or give examples of: electron configuration, Aufbau Principle, Hund’s Rule, Pauli Exclusion Principle, ground state, excited state, degenerate orbital, shielding, effective nuclear charge, valence electrons, valence shell, s, p, d, & f block, atomic radius, periodic ...
Review Sheet
... Endothermic vs. exothermic Stoichiometry using energy (using enthalpy of reaction and balanced chemical reactions) Calculating H using Hess’s Law, Enthalpy Diagrams, and/or H° of formations Calorimetry calculations to determine H and q Standard States and enthalpy of formation Using Hess’s Law eq ...
... Endothermic vs. exothermic Stoichiometry using energy (using enthalpy of reaction and balanced chemical reactions) Calculating H using Hess’s Law, Enthalpy Diagrams, and/or H° of formations Calorimetry calculations to determine H and q Standard States and enthalpy of formation Using Hess’s Law eq ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.