The Modern Atomic Model
... •Different orbitals varied by different quantum (energy). •Gaps between energy levels were not equal. ...
... •Different orbitals varied by different quantum (energy). •Gaps between energy levels were not equal. ...
Physics IV - Exam - Winter 2007/08 Please note:
... χ∗m φn dx. With the particle initially in the n an ground state in the potential from part (a) (Ψ = φ1 ), the 1D potential well instantaneously expands to twice it’s original size, with V (x) now given by: V (x) = 0 0 < x < 2L V (x) = ∞ elsewhere Work out the probability, immediately after this chan ...
... χ∗m φn dx. With the particle initially in the n an ground state in the potential from part (a) (Ψ = φ1 ), the 1D potential well instantaneously expands to twice it’s original size, with V (x) now given by: V (x) = 0 0 < x < 2L V (x) = ∞ elsewhere Work out the probability, immediately after this chan ...
Regents Review Packet B2 Answer Key
... 4. Identify the physical property in the table that could be used to differentiate the samples of the three elements from each other. ...
... 4. Identify the physical property in the table that could be used to differentiate the samples of the three elements from each other. ...
Quantum numbers
... When solving Schrödinger's equation for the hydrogen model we find wave functions / orbitals. Each orbital is characterized by a series of numbers called Quantum ...
... When solving Schrödinger's equation for the hydrogen model we find wave functions / orbitals. Each orbital is characterized by a series of numbers called Quantum ...
1 - shawnschmitt
... d. Dependent variable- the variable that changes as a result of the changes to the independent variable e. Control- a group in which the independent variable is not manipulated, used for comparison f. Hypothesis- a possible explaination for observations, a testable idea g. Mole- the amount of partic ...
... d. Dependent variable- the variable that changes as a result of the changes to the independent variable e. Control- a group in which the independent variable is not manipulated, used for comparison f. Hypothesis- a possible explaination for observations, a testable idea g. Mole- the amount of partic ...
Chem 2 AP Ch 7 MC Review
... 2. Is it possible for a fluorescent material to emit radiation in the ultraviolet region after absorbing visible light? Explain your answer. A) No, ultraviolet light has higher energy than visible light. B) No, fluorescent materials only emit purple and green visible light. C) Yes, fluorescent mater ...
... 2. Is it possible for a fluorescent material to emit radiation in the ultraviolet region after absorbing visible light? Explain your answer. A) No, ultraviolet light has higher energy than visible light. B) No, fluorescent materials only emit purple and green visible light. C) Yes, fluorescent mater ...
The Wave Nature of Matter - Waterford Public Schools
... • Instead, mathematical solutions to the wave functions give 3dimensional shapes (orbitals) within which electrons can usually be found ...
... • Instead, mathematical solutions to the wave functions give 3dimensional shapes (orbitals) within which electrons can usually be found ...
Chapter 4: Arrangement of Electrons in Atoms
... 3. The Q.T. describes the wave properties of electrons. 4. By solving the Schrodinger Equation, the probability of finding an electron at a given place around the nucleus is found. These probabilities are used to form atomic orbitals. 5. An Atomic Orbital is a three dimensional region around the nuc ...
... 3. The Q.T. describes the wave properties of electrons. 4. By solving the Schrodinger Equation, the probability of finding an electron at a given place around the nucleus is found. These probabilities are used to form atomic orbitals. 5. An Atomic Orbital is a three dimensional region around the nuc ...
Electron spin and the periodic table
... Elements with roughly half-filled shells such as the transition metals will form compounds in numerous ways. The most versatile element of all is carbon with its 2 2p electrons and four slots for adding more electrons. Ionization energies Many other properties beside chemical behavior show periodici ...
... Elements with roughly half-filled shells such as the transition metals will form compounds in numerous ways. The most versatile element of all is carbon with its 2 2p electrons and four slots for adding more electrons. Ionization energies Many other properties beside chemical behavior show periodici ...
Quantum Numbers
... 2s orbital is not degenerate (e.g., the same energy) with a 2p or a 1s orbital. The ml values are entirely dependent on the l values; each type of orbital has a set degeneracy. For an s-orbital, ml = 0, and degeneracy = 1. For a p-orbital, ml = -1, 0, +1, and degeneracy = 3. For a d-orbital, ml = -2 ...
... 2s orbital is not degenerate (e.g., the same energy) with a 2p or a 1s orbital. The ml values are entirely dependent on the l values; each type of orbital has a set degeneracy. For an s-orbital, ml = 0, and degeneracy = 1. For a p-orbital, ml = -1, 0, +1, and degeneracy = 3. For a d-orbital, ml = -2 ...
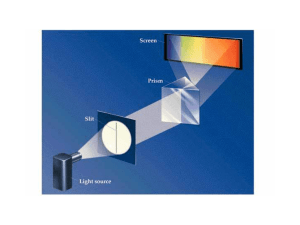
Atomic Spectra
... nm, 434.17 nm, and 410.29 nm. What is the wavelength of the next line of the series? What is the shortest wavelength line in this series? ...
... nm, 434.17 nm, and 410.29 nm. What is the wavelength of the next line of the series? What is the shortest wavelength line in this series? ...
Chapter 3 Make up Test 2004
... ______26. Which of the following statements explains why chemists do not count atoms and molecules directly? A. Atoms and molecules are extremely small B. All of the relationships in a chemical reaction can be expressed as mass ratios C. Matter is neither created nor destroyed in a chemical reaction ...
... ______26. Which of the following statements explains why chemists do not count atoms and molecules directly? A. Atoms and molecules are extremely small B. All of the relationships in a chemical reaction can be expressed as mass ratios C. Matter is neither created nor destroyed in a chemical reaction ...
Some Quantum Considerations II
... n = 1, l = 0, ml = 0, ms = +½. n = 3, l = 1, ml = 0, ms = +½. n = 3, l = 2, ml = -2, ms = -½. n = 2, l = 1, ml = 0, ms = +½. n = 1, l = 1, ml = 1, ms = +½. ...
... n = 1, l = 0, ml = 0, ms = +½. n = 3, l = 1, ml = 0, ms = +½. n = 3, l = 2, ml = -2, ms = -½. n = 2, l = 1, ml = 0, ms = +½. n = 1, l = 1, ml = 1, ms = +½. ...
Q: In which model of the atom do electrons orbit the nucleus? A
... are dumbbell shaped and have three axes along which they are situated? A: p orbitals ...
... are dumbbell shaped and have three axes along which they are situated? A: p orbitals ...
No Slide Title
... know the position (radius) and momentum (energy) of an electron, as defined in the Bohr model of the atom. ...
... know the position (radius) and momentum (energy) of an electron, as defined in the Bohr model of the atom. ...
CHM1045 - Michael Blaber
... 5. An electron transitions from the n = 2 to the n = 4 quantum state in Bohr's model of the hydrogen atom. Is a photon absorbed or emitted for this electron transition? (3 points). What is the energy, frequency and wavelength of the associated photon? (9 points). Is this wavelength visible? (3 poin ...
... 5. An electron transitions from the n = 2 to the n = 4 quantum state in Bohr's model of the hydrogen atom. Is a photon absorbed or emitted for this electron transition? (3 points). What is the energy, frequency and wavelength of the associated photon? (9 points). Is this wavelength visible? (3 poin ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.