Chapter 8 Study Guide
... c. Atoms of different elements differ in their physical and chemical properties. d. Atoms of different elements combine in simple, whole number ratios to form compounds e. In chemical reactions, atoms are combined, separated, or rearranged but never created, destroyed, or changed. 3. Describe how Th ...
... c. Atoms of different elements differ in their physical and chemical properties. d. Atoms of different elements combine in simple, whole number ratios to form compounds e. In chemical reactions, atoms are combined, separated, or rearranged but never created, destroyed, or changed. 3. Describe how Th ...
Simple Harmonic Oscillator
... Never express yourself more clearly than you are able to think. Prediction is very difficult, especially about the future. - Niels Bohr ...
... Never express yourself more clearly than you are able to think. Prediction is very difficult, especially about the future. - Niels Bohr ...
probability = ψ 2
... The nucleus is the very dense region consisting of nucleons (protons and neutrons) at the center of an atom. Almost all of the mass in an atom is made up from the protons and neutrons in the nucleus, with a very small contribution from the orbiting electrons. The diameter of the nucleus is in the ...
... The nucleus is the very dense region consisting of nucleons (protons and neutrons) at the center of an atom. Almost all of the mass in an atom is made up from the protons and neutrons in the nucleus, with a very small contribution from the orbiting electrons. The diameter of the nucleus is in the ...
7.4 The Quantum-Mechanical Model of the Atom
... – The orbital size increases with increasing the energy of the orbital (1s < 2s < 3s …) – Higher energy orbitals have several maxima in the radial distribution and one or more spherical nodes (regions with zero probability to find the ...
... – The orbital size increases with increasing the energy of the orbital (1s < 2s < 3s …) – Higher energy orbitals have several maxima in the radial distribution and one or more spherical nodes (regions with zero probability to find the ...
electron cloud - Wickliffe City School
... The trend across a horizontal period is less obvious. Each step from left to right adds a proton and an electron (and 1 or 2 neutrons) and electrons are added to existing energy levels. The effect is that the more positive nucleus has a greater pull on the electron cloud. The nucleus is more positiv ...
... The trend across a horizontal period is less obvious. Each step from left to right adds a proton and an electron (and 1 or 2 neutrons) and electrons are added to existing energy levels. The effect is that the more positive nucleus has a greater pull on the electron cloud. The nucleus is more positiv ...
Particle-like Properties of Electromagnetic Radiation
... Bohr- described the structure of the hydrogen atoms as containing an electron circling the nucleus where specific orbitals of the electron correspond to specific energy levels. B. Schrodinger- developed the quantum mechanical model of the atom - abandoned the idea of an electron as a small particle ...
... Bohr- described the structure of the hydrogen atoms as containing an electron circling the nucleus where specific orbitals of the electron correspond to specific energy levels. B. Schrodinger- developed the quantum mechanical model of the atom - abandoned the idea of an electron as a small particle ...
atomsagain
... •Normally, they will go into their lowest energy states •Each electron goes into the lowest possible energy ...
... •Normally, they will go into their lowest energy states •Each electron goes into the lowest possible energy ...
Electrons as waves
... Spin The last number is a spin indicator • -1/2 or + ½ • These are the two spin states, ( spin to the right or spin to the left) • No two electrons can be identical., or , no two electrons can have the same set of quantum numbers – Pauli’s exclusion principle • A maximum of two electrons can occupy ...
... Spin The last number is a spin indicator • -1/2 or + ½ • These are the two spin states, ( spin to the right or spin to the left) • No two electrons can be identical., or , no two electrons can have the same set of quantum numbers – Pauli’s exclusion principle • A maximum of two electrons can occupy ...
Quantum Notes (Chapter 16)(Powerpoint document)
... For n = 1, there is only 1 (n2) orbital. Higher values of n have several types of orbitals. These are distinguished by a second quantum number: ...
... For n = 1, there is only 1 (n2) orbital. Higher values of n have several types of orbitals. These are distinguished by a second quantum number: ...
Shapes of the Charge Clouds
... therefore specific amounts of energy are absorbed or emitted •Because the energy of an electron is quantized (discreet), there are only certain energy levels (orbits) for electrons •Therefore, this e-m radiation can behave as waves or particles = Wave-Particle Duality Theory •When an electron gains ...
... therefore specific amounts of energy are absorbed or emitted •Because the energy of an electron is quantized (discreet), there are only certain energy levels (orbits) for electrons •Therefore, this e-m radiation can behave as waves or particles = Wave-Particle Duality Theory •When an electron gains ...
The Quantum Model of the Atom
... • Principal quantum number (n): indicates the main energy level occupied by an electron • Only positive integers n= 1, 2, 3, 4… • As n increases, the electron’s energy and average distance from the nucleus increases • Each main energy level has sub-levels • Maximum number of electrons in a princi ...
... • Principal quantum number (n): indicates the main energy level occupied by an electron • Only positive integers n= 1, 2, 3, 4… • As n increases, the electron’s energy and average distance from the nucleus increases • Each main energy level has sub-levels • Maximum number of electrons in a princi ...
November 18
... Given that red light has a wavelength of 700 x 10-9 meters, what is the frequency and how much energy does it have? To find frequency, use first formula, c=lambda x frequency Frequency = 3x 108 m/s / 700 x 10-9 meters = 4.29 x 1014 Hz To find Energy, plug it into the second formula: E = hf = (6.6 x ...
... Given that red light has a wavelength of 700 x 10-9 meters, what is the frequency and how much energy does it have? To find frequency, use first formula, c=lambda x frequency Frequency = 3x 108 m/s / 700 x 10-9 meters = 4.29 x 1014 Hz To find Energy, plug it into the second formula: E = hf = (6.6 x ...
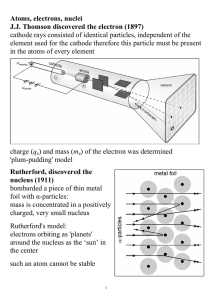
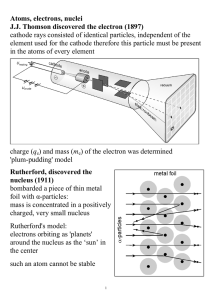
Atoms, electrons, nuclei J.J. Thomson discovered the electron (1897
... Davisson and Germer (1927) used electron beams to induce diffraction through a thin metal foil: interference interference phenomena have been shown with various other particles: duality is a general characteristic of matter Bohr's model (incorrect, but useful) electrons in an atom can only occupy ce ...
... Davisson and Germer (1927) used electron beams to induce diffraction through a thin metal foil: interference interference phenomena have been shown with various other particles: duality is a general characteristic of matter Bohr's model (incorrect, but useful) electrons in an atom can only occupy ce ...
Atomic_Orbitals
... energy of light and understand the origin of the atomic emission spectrum of an element. Objective 4 – Describe the different groups on the periodic table and relate them to their electron configurations. Objective 5 – Interpret group and periodic trends in atomic radii, ionic radii, ionization ...
... energy of light and understand the origin of the atomic emission spectrum of an element. Objective 4 – Describe the different groups on the periodic table and relate them to their electron configurations. Objective 5 – Interpret group and periodic trends in atomic radii, ionic radii, ionization ...
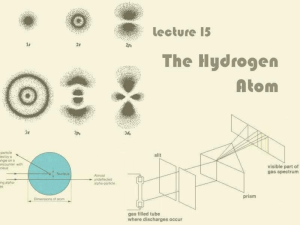
Lecture 15: The Hydrogen Atom
... Electron does not fly round the nucleus like the Earth around the Sun (Rutherford, Bohr) Depending on which energy level it is in, the electron can take one of a number of stationary probability ...
... Electron does not fly round the nucleus like the Earth around the Sun (Rutherford, Bohr) Depending on which energy level it is in, the electron can take one of a number of stationary probability ...
Chapter 2 - Speedway High School
... • Atoms of an element have the same number of protons but may differ in number of neutrons • some are radioactive ...
... • Atoms of an element have the same number of protons but may differ in number of neutrons • some are radioactive ...
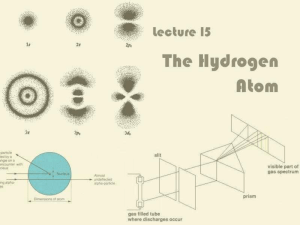
Lecture 15: The Hydrogen Atom
... Depending on which energy level it is in, the electron can take one of a number of stationary probability ...
... Depending on which energy level it is in, the electron can take one of a number of stationary probability ...
CH 6 electrons in atoms
... thinking about the boundaries of a city. If we want to find a citizen of the city, we have a 90% probability of finding that citizen if we look in the boundaries of the city. The same is true for an electron. We believe there is a good chance or a high probability of finding the electron within the ...
... thinking about the boundaries of a city. If we want to find a citizen of the city, we have a 90% probability of finding that citizen if we look in the boundaries of the city. The same is true for an electron. We believe there is a good chance or a high probability of finding the electron within the ...
Biol 1441
... Nonpolar covalent bond: the electrons of the bond are shared equally. Ex: N2 Polar covalent bond: the electrons of the bond are not shared equally. Ex: HCl Ionic Bonds: Two atoms are so unequal in their attraction for valence electrons that the more electronegative atom strips an electron completely ...
... Nonpolar covalent bond: the electrons of the bond are shared equally. Ex: N2 Polar covalent bond: the electrons of the bond are not shared equally. Ex: HCl Ionic Bonds: Two atoms are so unequal in their attraction for valence electrons that the more electronegative atom strips an electron completely ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.