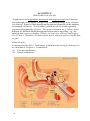
* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download king saud university - KSU Faculty Member websites
Thomas Young (scientist) wikipedia , lookup
Electromagnetic mass wikipedia , lookup
Mass versus weight wikipedia , lookup
Negative mass wikipedia , lookup
Gibbs free energy wikipedia , lookup
Internal energy wikipedia , lookup
Nuclear binding energy wikipedia , lookup
Anti-gravity wikipedia , lookup
Conservation of energy wikipedia , lookup
Chien-Shiung Wu wikipedia , lookup
Nuclear drip line wikipedia , lookup
Bernoulli's principle wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
State of matter wikipedia , lookup
Elementary particle wikipedia , lookup
History of subatomic physics wikipedia , lookup
Work (physics) wikipedia , lookup
Atomic nucleus wikipedia , lookup
KING SAUD UNIVERSITY COLLEGE OF APPLIED MEDICAL SCIENCES SHORT NOTES* (HAND-OUTS) ON THE PHYSICS FOR NURSES (Reference: Physics of The Health Sciences, 3rd Ed., Nave and Nave). The 3 fundamental standards for qualitative physical measurements in the SI system are: (1) Length (symbol l ) measured in meter (2) Mass (symbol m ) measured in kilogram (3) Time (symbol t ) measured in seconds (symbol m ) (symbol kg ) (symbol s ) Other base units: (1) Electric current (symbol I ) measured in ampere (symbol A ) (2) Thermodynamic temperature measured in kelvin (symbol K ) (3) Amount of substance measured in mol (symbol mol) (4) Luminous intensity measured in candela (symbol cd ) Other supplementary units used to express angular momentum: radian (rad) for plane angles, steradian (sr) for solid angles. * Common scientific (multiples) and (submultiples) are: PARAMETER SYMBOL Atomic Mass Unit Avogadro's Number U NA Boltzmann's Constant Electron Mass Elementary Charge (The electron) Electron-Volt Neutron Mass Proton Mass Plank's Constant Speed of light in vacuum K me E eV mn Mp H C Density of Air (at 20 C) Density of Water (at 20 C) Acceleration due to gravity (gravity) Standard Atmospheric Pressure G - VALUE / UNIT 1.6 x 10 -27 kg 6 x 1023 molecules (atoms)/mol 1.38 x 10-23 J/K 1/1840 u 1.6 x 10-19 C 1.6 x 10-19 J 1u 1u 6.6 x 10-34 J.s 3 x 108 m/s (3 x 1010 cm/s) 1.2 kg/m3 103 kg/m3 9.8 m/s2 10 Pa (N/m2) * Matter: Is material of which all things are made. It can exist in three physical states: Solid, liquid, and gas, but may also exist in liquid crystal and plasma states. 1 Law of conservation of matter and energy states that: Matter can neither be created, nor destroyed, but remains constant in a closed system. However, it can be converted from one form to another by physical or chemical means. For example, ice changes from a solid to a liquid (water) form which is a physical change and is reversible. Wood, however, can be burned to form aches. This is a chemical change and the process is irreversible. But some chemical changes are reversible. For example, chemical combination of oxygen with the haemoglobin (Hb) of red blood cells that takes place in the lungs, is reversed when oxygen is released from haemoglobin in the tissues. Matter and Energy Scientist Albert Einstein stated that: Matter and energy are interconvertible, that is, one can be changed into the other, or mathematically expressed: E = mc2 The relationship shows that conversion of only a minute amount of matter Of mass (m) will release very great quantities of energy (E) (as in a nuclear explosion). Conversely, a massive amount of energy must be used or (converted) to produce a small quantity of matter (m). Atom: Smallest building unit of matter, e.g., the hydrogen atom ( H ), the carbon atom ( C ), the zinc atom ( Zn ), etc .. Molecule: A combination of atoms bound together by electrostatic forces. Examples: A molecule of water (H2O), a molecule of carbon dioxide (CO2), a molecule of benzene, etc .. Particle: Is either a charged or neutral body of infinitely small dimensions: Examples are: The beta ( ) particle, the alpha ( ) particle, the proton (p), the neutron(n), the meson (), the keon (K), etc .. Electron (e): Is a negative particle of negligible mass of only 1/1840 u where u = 1.66 x 10-27 kg. A proton (p): Is a positive particle of mass 1.0 u. It exists in nuclei of atoms. Its number inside the nucleus equals the Atomic Number (Z), and it well defines (represents) the element. In an electrically neutral atom (that is, not ionized): Z=p Where Z is the atomic number, p is the number of protons. Total number of electrons equals the total number of protons, that is e=p 2 Neutron (n): Is a neutral particle (no charge), that is (q = 0), of mass = 1 u and exists in nuclei of atoms. (*) The Atomic Mass Unit (A) = p + n . The law of Nuclear Equilibrium assumes that (n = p) for the light elements (such as, hydrogen, helium, lithium, etc..). In the heavy elements (e.g., uranium, radium, lithium, etc..), this law is violated and nuclei of these elements are unstable (in a state of inequilibrium), that is they are (excited) and become what is known as radioactive elements. Scalar quantities (scalars): Quantities having magnitude (size or amount) only. For example, energy, time, mass, speed, etc .. Vector quantities (vectors): Quantities with both magnitude and direction. for example, force, velocity, acceleration, magnetic field strength, etc .. MECHANICS (KINEMATICS) Force is the agent by which a body's state of motion is changed. It explains the reasons why objects speed up, slow down, or change direction of travel. Force (F) is measured in Newton (N), where 1N = 1 kg.m/s2 Also, 1N = (1 kg) x ( g ), where ( g ) is the ( acceleration due to gravity) = 9.81 m/s2 . therefore, 1N = 9.81 kg, (approximately 10 kg weight). Energy (E): Energy is the ability to do work. It can be in a potential form (P.E.), or a kinetic form (K.E.). Potential energy is due to a position or state. Kinetic energy is due to motion. Energy is measured in joule (J), same unit as work, or in Electron-Volt (eV). The human body converts chemical energy (obtained from the food we eat and the oxygen we breath) into energy of movement (kinetic energy). Electrical energy is converted into light energy in lamps (e.g., the operating room lights). Also, light energy is converted to electrical energy in a solarpowered medical device. Power (P): Is the rate of doing work. Its unit is watt (W), or (J/s) which is expressed as: Power (Watts) = [Work done (j)] / time taken (s) 3 or P = W/t (watts) Unit of power is the watt (W), and 1W = 1 J/s (1 horse power (h.p) = 746 J) Displacement (x): Is change in position of a particle (or body). It is a vector quantity (with both magnitude and direction). Speed (s): Is a scalar quantity (has magnitude only), and is the ratio of total distance traveled to the total time taken, so that average speed = total distance/ total time (unit: m/s). Velocity (v): Is the speed in a given direction at a given instant in time. It is a vector quantity. Average velocity ( v ): v = (s) / t Constant velocity: Is the special case when the velocity remains constant (speed not changing and direction remains the same), therefore, v = s/t From average velocity ( v ): v = (s) / t and, in general, s = (sf – si) [final and initial speeds] therefore, t = (tf – ti) [final and initial times] Usually we start with si = 0, ti = 0, therefore v = (s) / t reduces to v = s/t Therfore, v = s/t and s = (v t) In summary: Velocity is the change in displacement / change in time: v = dx/dt (unit: m/s) Acceleration (a): Is the rate of change of velocity. In the case of motion in a straight line, average acceleration is a = (v) / t (unit m/s2). If initial velocity is v0 (for t = 0) and velocity at time t = v, then average acceleration equals or 4 a = (v – v0) / t v = v0 + at Acceleration is a vector quantity and = The change in velocity/change in time: a = dv/dt a = d2x/dt2 (unit: m/s2) Acceleration a is the derivative of velocity with respect to time: a = [dv/dt] It is always caused by the action of one or more forces. Example: A box full of medicines is pushed down a smooth incline with initial velocity of 10 m/s and accelerates at constant rate of 5 m/s2. What will its velocity be after 3 seconds? v = v0 + at = 10 + 5.(3) = 25 m/s Acceleration due to gravity (g): (Also known as the gravitational force): If air resistance = 0 , then, a falling coin and a falling piece of paper will both simultaneously hit the ground (earth) at the same time – because both are acted upon by the same gravitational force (g), which is always directed into a downwardly direction towards the center of the earth (This is a natural phenomenon!). g = – 9.8 m/s2 = (980 cm/ s2 ) Acceleration is always downwarded and equals (– g) . Density ( ): Equals the mass /unit volume, = m/V (unit: kg/m3). Material Air Water Magnesium Iron Lead Gold Mass (m): 5 (kg/m3) 0.001 x 103 1.000 x 103 1.750 x 103 7.860 x 103 11.30 x 103 19.30 x 103 Is a physical quantity and is amount of matter in a material, or the number of elementary particles (atoms or molecules) a material has. The kilogram (kg) is the SI unit of mass. To describe mass in terms of the elementary particles, another unit is used: The mole. 1 mole of matter contains (6 x 1023) elementary particles. This is a fundamental physical constant known as Avogadro's number. Weight (W): Is the total effect of the gravitational force, g , on a mass m: This is the same as W = mg F = mg (unit: N) So that the weight (W) of 1 kg mass exactly = 9.81 N. Work (W): If a patient has collapsed on the floor and succeeded on lifting him up, work have been done, because we have applied a force so as to move him through a distance upwards in the direction of the force. Work done = (force) x (distance) moved in direction of force. W = Fm (Fm) is called Newton meters) And 1 J = 1 Nm WORKED EXAMPLE (in gravitational potential energy): (1) A 50 kg patient seated in a wheelchair has to be lifted onto a doctor's table (couch) which was 25 cm higher than the wheelchair. Calculate work to be done to carry such a job and how much potential energy will the patient possess as a result? W = force x distance Hint: Minimum force (F) required to lift the patient up equals the (weight), W, of the patient. Weight (W) = mg = 50 x 9.81 N Convert the 25 cm into meters, = 0.25 m Calculate work done in lifting the patient: Work done (W) = force x distance = Weight x height = (mg) x h = (50 x 0.81) x 0.25 (Nm) = 120 (Nm), = 120J (Nm = J) Therefore, 120 J of work have to be done in lifting the patient. Therefore, P.E. will be increased by these 120 J. WORKED EXAMPLE (in kinetic energy): 6 Kinetic energy is energy possessed by a moving body, e.g., a patient falling from a trolley or stretcher. K.E. = ½ mv2 Example: A baby of 2 kg mass accidentally fell onto the floor from a place 1.5 m high. Calculate the K.E. the baby possess at the instant he hits the floor and how fast will he then be moving? First, find the P.E. P.E. = (mg) x h (weight x height). = (2 x 9.81) x 1.5 (J) = 29.4 J Thus, when the baby was high over the floor, his P.E. was 29.4 J greater than when he was on the floor. When he fell he had converted all the 29.4 J of P.E. into K.E. The moment he stroked the floor, his K.E. was 29.4 J. How fast he was moving? K.E. = ½ mv2 v2 = [2 x K.E.] / m = [2 x 29.4] / 2 = 29.4 m2/s2 V = 29.4 = 5.4 (m/s) Newton's Laws of Motion: First Law: A particle (or a body) of mass m will remain at rest, and a particle (or a body) of uniform motion (zero acceleration and constant velocity) stays in that motion in a straight line unless acted upon by external resultant forces (F). Types of forces: Mechanical, electric, magnetic, gravitational, electrostatic, nuclear, resistive, etc .. Second Law: Acceleration of a particle (or object) is directly proportional to resultant force F acting on it, and inversely proportional to its mass m , so that: F = ma (unit: N) Definition of a Newton: A force of (1N) will produce an acceleration of 1 m/s2 in a body of mass (1 kg), if it is free to move and not acted upon by any other forces. Circular Motion and the second law: 7 The Centrifuge: A particle of mass m moving in a circular path of radius ( r ) with uniform speed (v) will have a central force (Fs) due to the centripetal acceleration: Fs = ( mv2 ) / r Clinical and research laboratory centrifuges use this central force to push heavier material (like RBCs, WBCs, proteins, etc .. ) towards the center of rotation, leaving behind lighter material (e.g., plasma). m (test tube) Fs 0 Rotating drum r Simple manual centrifuge 8 Advanced digital centrifuge 9 Centrifugation can be made more efficient (settling even smaller elements) by increasing the velocity of rotation, v , (as in the ultracentrifuge, usually with velocities up to 100,000 rpm). Third Law: (Action) and (reaction) are equal and opposite: F12 = F21 Frictional forces: These forces can resist relative motion between two surfaces in contact, whether they are of mechanical nature ( e.g., baby balance bridge, a sliding x-ray table top, a piston, etc), or of a biological nature such as (synovial joint articular surfaces of the knee joint, shoulder joint, etc ..), or the motion through a viscous medium such as air or water. Frictional forces are always opposite in direction to applied forces and can have undesirable effects such as: (1) Reducing the efficiency of machines (output becomes less than The input). (2) Produces heat (dissipated thermal energy). (3) Damaging surfaces in contact: This causes slow movement of the contacting surfaces (and pain in the human joints). Because of this, the modern plastic gastric (or rectal) catheters and tubes are used instead of the old rubber type to reduce the frictional effect. For machines' moving mechanical parts, oil and other lubricants are used to reduce friction. For catheters and tubes, special antiseptic water-soluble oils are used so as to overcome frictional forces. Crater (heavy box) Static frictional force, s N (Normal force) Applied force, F Rough surface N N = W = mg (W = mg) is the weight of the block and represents the gravitational force that acts on the block. N is the normal force (force pressing the surfaces together) In this case it equals the weight of the box. When the block is 10 static, the static friction force (force opposing the motion) is s and it = F. When F > s , the block will move and start to accelerate to the right. Then, s = µs N Where, µs is a constant known as the coefficient of static friction. Its value will depend on the nature of the two surfaces in contact. Typically, µs will range between ( 0.05) and ( 1.5 ) Material µs Human synovial joints Ice on ice Lubricated metal on metal Glass on glass Rubber on concrete Steel on steel 0.01 0.10 0.15 0.94 1.00 0.74 NB/ The only external forces acting on a box are: The normal external force (F), the force of gravity (mg), and the force of static friction, s . Static friction force: the relationship s = µs N Is used to calculate the maximum resistance force an object can exert before it starts to move. Example: An 80 kg box of medicines has coefficient of static friction (µs ) of (0.4) . What force is needed to start moving the box? s = µs N = (0.4) . (80) = 32 kg force. NB/ The normal force (N) in this case is the weight of the box. Kinetic friction force: the relationship k = µk N (µk) is the coefficient of kinetic (motion) friction, is used to calculate frictional resistance force required to (keep) a body (moving). Example: If coefficient of kinetic friction (µs ) of the above box is (0.3). What force is needed to keep it moving with constant velocity once started? k = µk N = (0.3) . (80) = 24 kg force. 11 N.B./ 1. Whenever there is relative motion between two surfaces which are in in contact, frictional force ( ) will resist this motion. 2. Magnitude (size) of this force depends on the nature (texture) or the roughness of the two surfaces in contact (are they very smooth, or smooth, or rough, or very rough). 3. ( ) is present even with (perfectly) polished surfaces because of: a. Microscopic irregularities. b. Surface adhesion. c. Electrical properties of the surfaces. Disadvantages of frictional forces: a. Increased effort needed to operate any mechanical device. b. Produces heat (by exhausting – using up- mechanical energy). c. Wears or damages surfaces rubbing against each other. Advantages of frictional force: * In the absence of friction ( = 0) humans will not walk because of absence of (traction). * Friction must be maximized when useful, and reduced when harmful. For example, in clinical applications such as (insertion of gastric tubes, rectal catheters, angio-catheters, measures must be taken to reduce the burning and irritation caused by (friction) from these tubes and catheters. Modern plastic tubing and catheters are found to be superior to rubber. * Fluid also has frictional forces that are much smaller than forces between solid-solid. For this reason, lubricants are used such as (oil) to separate the two surfaces, thus limit friction to the much smaller fluid friction. In the absence of lubricants, water can be used in some cases. This is the why water is taken with capsules or tablets Work (W): If (F) is a constant (pushing force) acting on a mass m, x is the displacement or motion of of the mass m in the direction of the force, then total work (W) done by the pushing the mass m is defined as: W = Fx cos No work will be done (that is W = 0) if the force F is applied in a direction perpendicular (that is 90°) to the direction of the force. Why? This is because W = Fx cos , and (cos ) in this case = cos 90 = 0 Unit of force is the joule (j) and equals (N-m) because W = Fx . 12 Kinetic Energy (K.E.): Is energy due to motion of a particle or body of mass (m). It is one form of mechanical energy: K.E. = ½ mv2 (unit: J ) Average Power ( P ): Power is the rate of doing work, where P = W /dt (unit: watt (W), or horse-power [h.p] ) and 1W = 1 J/s 1 horse-power (h.p.) = 746 W. Potential Energy (P.E.): Is energy due to position or state of a mass (m) or an object. It is a (stored) energy that can be trans-formed to K.E. or other forms of energy. Total energy of a system is always conserved (constant), that is, energy cannot be created, nor it can be destroyed. It can only be (transformed/ converted) into other types of energy, e.g, heat. Linear Momentum ( P ): Momentum is a vector, which is also conserved (total momentum of a system is always constant), and P = mv (unit: kg.m/s ) Conservation of Energy: Total mechanical energy of a closed system is constant, that is: Initial energy = Final energy And the total mechanical energy for a system is Etotal = (K.E.) + (P.E.) PROBLEMS: 1. A 1.5 years old girl has a mass of 10 kg. What is its weight in newtons? (assume (g) = 9.81 m/s2) (Answer: 98.1 N). 2. How fast will a patient trolley be travelling after it has fallen from a high Ward under the effect of gravity for 10 s? (neglect air resistance) (Answer: 98.1 m/s). 3. How much work is done in lifting a 100 kg patient from the floor onto A stretcher 1 m high above the floor? (Answer: 981 J). 4. How much power is required to do this lift in 1s? 13 (Answer: 981 W). 5. What force is required to accelerate a stretcher trolley at 1 m/s, if its mass was 20 kg and it carries a patient of 80 kg? (Answer: 100 N). 6. How fast will it be travelling if this force is maintained for 5 s? (Answer: 5 m/s). 7. How much energy would be needed to bring the trolley to a halt (to rest)? (Answer: 1250 J). SIMPLE MACHINES Include levers (e.g., the pair of scissors, the children swing, etc .. ), pulleys, and carts (used for carrying oxygen and other medical gas cylinders around the hosp- ital). Most machines have a < 100% (less than unity) efficiency, that is their output is always less than their input and the difference is (expended) as waste energy (thermal energy – or heat) because of these effects of friction. This is why machines have to be lubricated (oiled) to reduce frictional forces. For purpose of improving machines' efficiency (so as to overcome friction), machines (e.g., the gas cylinder's carts) are made with longer effort (applied) arms, or shorter resistive (load) arms. Class 1, Class 2, and Class 3 Levers class-1 lever load lever arm fulcrum class--2 lever bellows A lever in which the fulcrum is between the load and the effort. The weight or resistance that is moved using a simple machine. A beam, free to pivot around a point. The point at which a lever arm pivots. A lever in which the load is between the fulcrum and the effort. A device that takes in air when the sides are spread apart and expels it through a tube when the sides are brought together. A ratio of the load or resistance to the effort or force. mechanical advantage equal-arm balance A scale used to compare masses placed at equal distances from the fulcrum. class-3 lever A lever in which the effort is between the fulcrum and the load. The Wheel and Axle wheel and axle windlass 14 Two wheels of different diameters attached and rotating on the same axis. A wheel and axle system used to apply force to a rope while winding it around the axle (also called a winch). The Inclined Plane inclined plane slope A flat surface set at an angle, used to change the direction of a force. The slant of an inclined plane. The Wedge wedge A double inclined plane that tapers to a point or sharp edge, used to change the direction of force. Pulleys pulley fixed pulley movable pulley compound pulley sheave block and tackle chain hoist hydraulic A wheel with a grooved rim in which a rope can run to change the direction of the pull and so lift a load. A pulley attached in position above a load to be lifted. A pulley attached to a load that is being lifted. Two or more pulleys working together. A grooved wheel used in a pulley. Another name for a pulley system. A compound pulley system using chain instead of rope. Operated by means of fluid pressure. A class-2 lever has its fulcrum at one end of a lever arm. The load is between the fulcrum and the effort. With this kind of lever, the direction of effort is not changed. Pushing up on the class-2 lever arm pushes up on the load. Pushing down on the lever arm pushes down on the load. To gain a mechanical advantage, the load is places closer to the fulcrum than to the effort. The class-2 lever always reduces effort. 15 In a class-3 lever, the fulcrum is at one end, and the effort is applied between the fulcrum and the load. With this kind of lever, the direction of effort is not changed. The load moves in the same direction as the effort. The gain offered by a class-3 lever is one of distance. 16 FLUID MECHANICS It is the study of fluids, It is subdivided into fluid dynamics and fluid statics depending on whether the fluid is in motion or not. Fluids at rest: Fluids ( )الموائعinclude both the liquids and the gases. They are a collection of randomly moving atoms or molecules held together by weak cohesive forces, and by forces exerted by the walls of the container (the vessel). Fluids in motion (Fluid dynamics): Fluids have a flow which can be: (1) Steady (laminar) flow: In this flow, each particle follows a smooth noncrossing path. The velocity of the fluid at any point remains constant. (2) Non-steady (turbulent) flow: In this flow, and above a certain critical speed, flow becomes turbulent (irregular, and characterized by small whirl-pool like regions). Pressure (P) in a liquid: Pressure is defined as force per unit area. And force is proportional to the depth (height h) of the liquid and to its density (d). For a vessel (container) of water in a shape of 1 m3 : Weight = mg = (dA)g = dghA Liquid pressure at the bottom of the container = Weight/area, and PL = (dghA)/A = dgh This is not the (absolute) pressure on the bottom, because it did not take into Account the atmospheric pressure (atm) on top of the liquid. Therefore, absolute pressure = liquid pressure + atm pressure PL is the liquid pressure (commonly known as gauge pressure). It usually measures pressure (excess) over atm pressure. For our example of 1 m3 of water (density of water is 1000 kg/ m3) , PL = (1000).(9.8).(1) = 9800 (N/ m2) = 9800 pascals (Pa) = 9.8 kPa 17 Therefore, 1 m of water exterts (has weight pressure) of 9.8 kPa) Pascal (Pa) is the SI unit of pressure. In the British system of units: Water pressure of 1m depth (9.8 kPa) = PL = 62.4h pound/ft2 = 0.43h pound/in2 Where h is the depth of water in feet. The above relationship gives only the pressure caused by the weight of the liquid – not considering the atmospheric pressure (atm). Atm = 14.7 pound/ in2 (760mm Hg). Example: At what depth in water will the liquid pressure (PL) and atm pressure be equal to each other? For equalness, (0.43h) = 14.7 h = (14.7)/0.43 = 34 Therefore, at depth 34 feet, total pressure (absolute) is twice atm pressure. That means: Standard atm pressure = 14.7 pound/ in2 Liquid pressure ( PL ) at 34 feet = 29.4 pound/ in2 Example: Pressure required for intravenous fluid (IV) infusion into a vein can be done by lifting the fluid bottle (container) above the point of infusion. If liquid level in a bottle is 18 inches (= 1.5 ft) above the point where the needle enters the vein, and the liquid is assumed to have the same density as water (1 gm/ cm3 , or 62.4 pound/ ft2). What is the infusion liquid pressure at the needle tip? PL = hd = (1.5).(62.4) = 93.6 pound/ ft2 = 0.65 pound/ in2 NB/ (1) No details about the bottle or container shape, material, or type are necessary for calculating the pressure, that is pressure is determined only by: a. The density (d) of the liquid. b. The depth (h) of the liquid. (2) For irregular shaped containers, liquid pressure (PL) is the same and depends only upon the height (h) not on the shape or volume. 18 h NB/ Height h in all commonly connected containers will be the same, unless the vessels are so small that capillary action is significant. General relationship for empirical pressure is PL = dgh Where, (d) is the density in kg/m3 , (g) = 9.8 m/s2 , and (h) depth in meters. For a depth of water of 1 m heigh will exert a pressure at the bottom of the container of PL = (1000).(9.8).(1) = 9.8 kPa However, in the British system (weight) measures are commonly used rather than (mass) measures. Then the term 'weight density', dw is used, and PL = (dg)h = (dw)h (Empirical Unit) (British Unit) Where dw = dg . Weight density (dw) of water = 62.4 pounds/ft3. Therfore, pressure at a depth of 1 foot = PL = (dw)h = (62.4) . (1) = 62.4 pounds/ ft2 = 0.43 pounds/ in2 With standard atmospheric (atm) pressure of 14.7 pounds/ in2, the absolute (total) pressure at a depth (h) of 1 foot in water is PA = 14.7 + 0.43 * Pressure at the bottom of a column of liquid is directly proportional to the height (h) of the column. Therefore, (h) is generally used as a pressure measuring (or indicating) instrument. The simplest of pressure measurement devices is the (manometer) that consists of a simple vertical tube of liquid (usually mercury of water). Blood pressure instrument (sphygmomanometer), 19 atm pressure instrument, pressure measuring instrument of suction units, etc.. all use (cm Hg), or (cm H2O), or (mm Hg) measures. Pressure is said to be 20 cm Hg (or 200 mmHg) if pressure at the bottom of the mercury tube is enough to push mercury into the tube to a height (h) of 20cm. Note that: 20 cm Hg = 272 cm H2O 1 cm Hg = 13.6 cm H2O TRANSMISSION OF PRESSURE Hydrostatic (no-flow) conditions: Distribution of pressure in a static liquid makes possible the use of fluids to transmit pressure in the Intravenous (IV) apparatus generally used in hospitals for fluid infusion. Note that: 1. Pressure at depth (h) below the surface of a liquid is proportional to the depth (h) and the density (d), but has nothing to do with the shape, type, or configuration of the vessel or (tubing). 2. Pressure is exerted equally in all directions in a static liquid. The weight density (dw) for H2O = 62.4 pounds/ft2 . Therfore, at a depth of 1 foot = PL = (dw)h = (62.4) . (1) = 62.4 pounds/ft2 = 0.43 pounds/in2 Because water pressure is often used, it is useful to use the empirical relatIonship: PL = (62.4h) pounds/ft2 = 0.43h) pounds/in2 where (h) is the depth (height) of water (in feet) in both cases. Pressure distribution: Since pressure at a depth of 1 foot is 62.4 pounds/ft2, therefore at 10 feet the pressure will be 624 pounds/ft2. This pressure acts in all directions at this depth. Therefore, the sideways pressure against the walls of the liquid container will be 624 pounds/ft2 at 10 feet, and 62.4 pounds/ft2 at 1 foot. We have to note that: 1. Pressure increases with depth (h) (compare the results above). 2. Horizontal pressure also increases with depth (h). If holes are punched on the sides, increase in horizontal pressure with increased depth can be noticed. 20 Pascal Principle: Principle states: (Any change of pressure in an enclosed fluid is transmitted not diminished to all parts of the liquid). In an open liquid, static pressure is caused by its weight and equals: Pressure = (Weight density) x (depth at particular point) = (dw) . h In an enclosed liquid, any external pressure to this open liquid pressure must be added to this pressure to get the absolute (total) pressure, e.g. Example: If atm pressure was 15 pounds/in2 on every exposed object at the see level. What is the pressure at a depth (h) below the surface of a liquid? Pressure = (Weight density) x (depth at particular point) = [(dw) . h] + 15 pounds/in2 NB/ This transmission of the (15 pounds/in2) to all parts of the liquid is an example of Pascal's principle. The hydraulic press is a good apllication of Pascal's principle. It is used to overcome a large resistance (weight) by the exertion of a small effort force (weight). In fact, this is one of the basic functions of a machine. 1000 pounds A2 F1 A1 F2 F2 A1 = 1 in2, A2 = 100 in2. A small effort force of 10 pounds (F1) is exerted on A piston of cross-sectional area A1 of 1 in2. This pressure id transmitted to all parts of the liquid and acts on the bottom of a large piston of an area of 100 in2. With a pressure of 10 pounds/in2 on this piston, the upward force F2 can be calculated as follows: 21 P1 = P2 (F1)/ A1 = (F2)/A2 (10)/ 1 = (F2)/100 and F2 = (10 pounds/in2 ) . (100 in2) = 1000 pounds Mechanical advantage (%) = Fr /Fe = (1000/10) x 100 = 100% This mechanical advantage is also equal to the ratio of the areas of the two pistons. Medical applications of the hydraulic press: 1. Dentist's chairs. 2. Some x-ray and radiotherapy tables. 3. Car lifts (in garages), and car hydraulic brakes. Pressure transmission through Intravenous IV fluids tubing: Because of the fact that pressure (acts) in all directions, it can therefore, be transmitted through any tubing for administration of IV fluids. S h Pressure = dw under hydrostatic (no flow) conditions. For the apparatus above, Pressure is determined by the height (h) of the liquid surface (S) above the Point of infusion input (I) to the patient. This pressure will be transmitted through the tubing even if it has curls, turns, or even coils. But the pressure can be obstructed by (kinks) and (constrictions)! Since liquid pressure is controlled or determined by the height (h), pressure at the patient (under static no flow 22 conditions) will be the same in A, B, or C. When the fluid is flowing, however, then there will be some (drop) in pressure compared to the static pressure. This is due to fluid friction. If the length of A, B, or C tubing is the same, then frictional effects will be about the same and pressure in them will be the same under (slow) flow conditions. Under (slow) flow conditions, pressure at the patient's arm (site of fluid insertion) depends only on the height (h) and not on the path (A, B, C…) taken by tubing to reach the point of insertion. SUMMARY: * Static (no flow) pressure in an open liquid is caused by it's weight and equals to the weight density times the depth at the point considered: PL = (dw).h * Pressure acts equally in all directions. 23 THERMAL ENERGY (HEAT) Thermal energy (the heat phenomenon) is due to kinetic energy (K.E.) of the random motion of particles (atoms and molecules) in solids, liquids, and in gases. These particles are always in motion. Type of motion will depend on whether the material is a solid, liquid, or gas: a. In solids: Molecules vibrate (oscillate) about fixed positions. This is why solids do not (change) their shape easily! b. In liquids and gases: Molecules move randomly. For this reason they both do not have fixed shapes. c. Because these particles are in constant motion, they possess K.E. In the solids and liquids the molecules will also posses P.E. because of the intermolecular forces of attraction between them (strongest in solids, but weak or even absent in gases). Total K.E. and P.E. of the molecules in a system is called internal energy of the system. Temperature: Is the measurement of the rate of this movement (that is, the degree of hotness). At higher temperatures, solids melt. With more temperature, they boil. At still higher temperature, they vaporize (change into gas). This is known as the change of state. Temperature of a body depends on the average K.E. of its particles (which also depends on their speed of motion). At higher temperatures, particles are in great motion. At lower temperatures, they move slowly. Temperatures scales: These include: The Celsius (C), the Fahrenheit (F), and the Kelvin (K). The Kelvin is widely used in scientific research and in hospitals (clinical use). Normal body temperature measures: 98.4 F, 37 C, or 310 K . Units of heat are: The joule (J) and the calorie (cal). Heat equivalence states that: 1 cal = 4.2 J . Heat flow: Heat flows (is transferred) in solids by conduction, in gases by convection, and in vacuum by radiation. Heat flow in solids: Particles (vibrate) or (oscillate) about approximately fixed positions. This is why solids do not change their shapes easily. Heat flow in liquids and gases: Particles move randomly. They, therefore, have no fixed shapes. They take the shape of the containers or vessels. 24 Heat as a form of kinetic energy: Because all these particles are in constant motion, they posses K.E. of the moving atoms and molecules which is called (heat) or (thermal energy). Effects of temperature change: 1. At higher temperatures a substance will expand. If temperature is reduced it will contract. Clinical mercury thermometers work on this principle by expanding and contracting with temperature change. If this expansion and contraction is prevented, then a change in pressure (P) will occur. 2. At very high temperature, solids melt (change from solid state to liquid). 3. At extremely higher temperatures, liquids boil (change to gas state). Water (ice) melts at 0°C. It boils at 100°C. Absolute Zero (0K): This is the stage at which atomic particles (which are usually moving), are at rest. At 0K, a body will have no heat energy. 0°C = 273K = 32° F 100°C = 373K = 212° F Clinically, normal body temperature = 98.4°F = 37°C = 310K. SI unit of heat is the joule ( j ). The calorie (cal) may be used also. 1 cal = 4.2 j (called heat equivalence). NB/ 1 Calorie is the amount of heat required to cause 1°C rise in temperature in 1 gm of (water). 1 gm of most other substances requires (less) heat than (water) to raise their temperature by 1°C. This is called the specific heat s). It is the heat energy in joules required to raise the temperature of 1 kg of a substance by 1°C. Heat energy required = smt Specific heat (s) of water = 4200 J/kg/°C Specific heat of copper = 420 J/kg/°C Thermal capacity (C ): Is the heat required to cause 1°C temperature rise in A body component (no mass defined). Heat Transfer: (1) Heat conduction: Occurs in solids. Heat is transferred through the material Through atomic vibrations (oscillations) produced by collisions between the 25 Neighbouring atoms rate of this transfer depends on: a. Cross sectional area. b. Temperature difference between the ends of the material. c. Length (distance) heat has to travel. Rate of heat flow = [ kA (T2 – T1)] / L (2) Heat convection: Occurs in liquids and gases (heat moves through by circulation of heated liquid or gas forming conventional currents). When liquid or gas is heated, it becomes less dense (lighter) and rises up to be replaced by cooler liquid or gas. (3) Radiation: In vacuum (or space). It is how heat reaches the earth from the sun. Vibrating atoms and molecules emit electromagnetic rays or radiations (waves). Amount of heat energy radiated depends on the temperature of the body: E α T4 (T = temperature in kelvin). Thus, if teperature of a body is doubled, the Radiated heat energy will be (16) times as much. A body may absorb heat energy from external source and at the same time Be radiating heat energy: a. If a body absorbs more than it radiates, its temperature will rise. b. If it radiates more than it absorbs, its temperature will drop (fall). c. If the heat absorbed balances the heat radiated, temperature will remain (constant). Surface colour and texture: Matt and black surfaces are efficient absorbers and radiators of heat. White And glossy surfaces will not. Digital thermometers 26 ELECTRICITY Law of electrostatic charges states: (Like charges repel each other, and unlike charges attract each other). Attraction or repulsion forces are (inversely) proportional to the distance between the charges and (directly) proportional to the sizes of the charges involved: F = k (Q1 x Q2) / d2 (Q is a –ve or +ve charge). Electric potential and potential difference (p.d.): Have unit of Volt (V) and exist between two charges. Electric potential of the earth (ground potential) = 0V , that is neutral, because the earth is too large. All electrical equipment have to be connected to the Earth (or ground) so that charges (the current) can pass ' safely ' to the ground and prevent operators from being electrified (effects of an electric shock). The electric current (I): Is the rate of flow of electric charges. Electron flow is either from the –ve to the +ve or from the +ve to the –ve pole of a battery in a direct current (DC) circuit. In an alternating current (AC) circuit, the current flow is in both directions. Unit of current is the ampere (A). The electron Volt (eV): Is another unit of energy and equals the work done in moving an electron through a potential difference of 1 V . 1 keV = 1000 eV, 1 MeV = 106 eV. Electric conductor: Is the material with many 'free' or unbound electrons and can easily conduct electricity. Examples are metals, e.g., copper, aluminium, zinc, iron, ferrous, silver, gold, etc… Also some of the liquids like (salty water), and some fluids and gases like (wet air), are good conductors. Silver and gold are the best metallic conductors – but they are expensive! Electric insulator: Is the material with very few free electrons and will not, therefore, conduct electricity. Good insulators include porcelain, glass, wood, plastics, dryair, waxed paper, and some oils (e.g., transformer oil). Mica is the best known conductor. Electric resistance ( resistor), (R): It is any electric device (made of carbon or coiled wire) used to reduce electric currents. Its unit is the ohm (). When connected 'in series', the 27 resistors are additive (that is, they add together): R1 R2 Rtotal = R1 + R2 When connected 'in parallel', R1 R2 Rtotal = (R1 x R2) / R1 + R2). Ohm's Law: States that: Potential difference (V) across a conductor is directly proportional to the current (I) passing through the conductor if the resistance (R) remains constant: Voltage (V) 0 Current ( I ) Capacitance (Capacitor) (C): Electric device that store electrical charge. Its unit is the farad (F). NB/ Capacitors in series are just like resistors in parallel. Capacitors in parallel are just like resistors in series. Symbol of a capacitor is C Electrical power (P): Electrical energy (E): 28 P = VI (watts) = I2R (watts) E = Pt (joules) Alternating EMF and current (A.C.): Alternating EMF (voltage) and alternating current (AC) are generated from generator (dynamos) as a single-phase (1 ) or three phase (3 ) electrical supplies. Nominal (mains) single- phase electrical supply = 240V, and is supplied by two cables or wires (L and N). Voltage on (L) = 240V, and voltage on (N) = 0V. Also an earth lead or cable (E) has 0V in it and is used as a safety wire, usually connected to all electrical equipment. Single-phase supply is used for simple lighting (AC lamps), low-power x-ray units, low-power operating-theatre equipment, dental apparatus, ENT scopes, fibre optics, etc.. 3-phase supply usually give 415V ( = √3 x 240) and is supplied by three lines (L1, L2, and L3). Voltage between any two of these lines = 415V. It is used for lifts and for the large x-ray units(general, C.T., MRI), theatre units, radiotherapy, etc.. Transformers: Is a device to transform electrical energy from low to high (then called a high-tension transformer), or from high to low (then called a stepdown transformer). Heat produced by transformers is due to (power losses). Step-down transformers are used to provide low-power voltage (6V – 30V) to small lamps in endoscopes, laryngoscopes, ENT lamps, small dental motors, pocket calculators, telephones, mobiles, etc.. Hightension transfomers are used for all high-tension units - like x-ray tubes, radiotherapy equipment, operating-rooms lamps, physiotherapy units, electro-surgery equipment, etc.. 29 ELECTROMAGNETIC RADIATIONS Are transverse waves (rays) with no mass or charge and travel as magnetic and electric fields in vacuum. They includes all radio waves, the TV waves, the radar, microwaves, infrared rays IR (heat), white light rays, ultraviolet rays UV, X-rays, and gamma rays . They all have the same velocity (c) in vacuum = 186,000 mi/sec. c = 3 x 108 m/sec (known as the speed of light in vacuum) = 3 x 1010 cm/sec They have properties of reflection, refraction, and diffraction and can also penetrate matter to different extent (x-rays and are more penetrating than others). White paper can penetrate glass, some paper, and transparent plastics. All electro-magnetic radiations (rays) can cause varying degree of ionization in matter. This is why they form a biological risk ! X-rays (X-radiation): Because x-rays (like other radiations) can be absorbed through many processes by the human body, they can produce scatter (secondary) radiation which may be harmful to the nursing staff, patient's attendants, and visitors. Late effects could include erythema, burns, ulceration, and cancer. Best way to protect from x-rays is by using distance (staying away from the x-ray source, or by lead-rubber shielding and radiation barriers. Radiofrequency waves: Both radiofrequency waves and microwaves have low photon energy because of their low frequency range (500 kHz – 2,500 MHz). When they interact with matter or the human tissues, they only cause some molecular agitation and chemical effects in the form of heat. Over-exposure to radiofrequency or to microwaves can cause internal or external burns of the human body's skin (skin burns). For this reason, the high energy radiofrequency and microwaves are usually used in diathermy (deep heat therapy) to relief muscular pain and inflammation of skeletal bones. These waves are limited in their frequency range (and in power) because of their interference with communication networks (Radio, TV, telephones, and mobiles). They are now limited to a frequency range of 900 MHz – 2,500 MHz. Infra-red (IR) radiation: Generally known as (heat or thermal waves) and have a wavelength range of (7,500 – 30,000) Angstroms . It is widely used in the form of (IR) lamps for superficial heat therapy (less penetrating than radiofrequency and microwaves). Also it is used in medical thermography to photograph surface veins, skin, the eye, for detecting burns, and detecting breast cancer. 30 Ultra-violet (UV) radiation: These have wavelengths in the range (2,500 – 4,000 Angstroms), therefore, they are strongly absorbed by any matter and can cause ionization in the human skin, leading to (sun-burn) and (skin cancer) at the higher frequency range. They always penetrate up to 1 mm of tissue, thus, it is used in skin therapy to treat acnes and warts. Because it kills bacteria, it is generally used in air and instruments sterilization in operating and surgery rooms. Because it is harmful to the eyes (destroys the retina), UV operators must wear special eye goggles. 31 X-RAYS (ATOMIC RADIATION) X-rays are transverse electromagnetic rays of high energy and very short wavelengths (0.02 – 10) Angstroms produced inside an x-ray tube by the interaction of electrons with a target made of the tungsten metal. They can penetrate most materials and human tissue. Very little energy is absorbed by these structures leading to ionization and therefore to scatter rays. The scatter rays can cause biologic damage if they were excessive. Low atomic number (Z) materials like human tissue, plastics, wood, glass, paper, etc.., are easily penetrated by x-rays. High atomic number materials like all metals, some non-metals, and human bones greatly absorb x-radiation. (Barium sulphate) powder is a high (Z) material which is commonly used as contrast material for diagnosing the stomach and intestines. Lead metal is also used as a protective material in some of the x-ray clothing (aprons) and for covering doors, windows, and protective barriers. X-rays have some effects: 1. They cause ionization in matter. 2. Affect photographic films. 3. Cause fluorescence in some phosphors. Radiation quality: Is its penetration power. Radiation intensity: Is the amount of radiation energy per unit area. An x-ray film (a radiograph) for paranasal sinuses 32 RADIOACTIVITY (NUCLEAR RADIATION) Is emission of gamma ( ), beta ( ), or alpha ( ) radiation from unstable nuclei (radioactive nuclei) of atoms. Nuclei are unstable if they were in the nuclear equilibrium state, that is they have more protons or neutrons inside them. Higher atomic number (Z) elements, like uranium and radium, are naturally unstable, and therefore radioactive – they emit alpha, beta, and gamma rays. They give-off excess neutrons or protons as excess particles. Beta is an electron (negative particle) emitted from excess neutrons, with a mass of ( 1/1840u ). Alpha particle is made from 2 protons and two neutrons and therefore it has a positive charge and a mass of ( 4u ). Both beta and alpha are particles and, therefore, have masses. Gamma rays ( ) are a type of 'electromagnetic' radiation and is emitted as an energy difference (excess). Most elements have isotopes. These are materials with same Z, but with different atomic masses (A). As example, hydrogen has only three isotopes: Hydrogen, deuterium, and tritium: ( 1H , 2H , 3H ) All the three forms of hydrogen are chemically the same but they differ in their physical properties. H-2 combines with oxygen to form heavy-water. H-3 is a radioactive gas. Some other element's isotopes are radioactive, e.g., cobalt-60 (Co-60) is commonly used in radiotherapy (treating cancer with radiation). Iodine-131 (I-131) and caesium are also used for diagnoses in nuclear medicine. Radioactive gold is also used in the treatment of cancer of the cervix. Half-life Time (HLT): Is the time in which activity (power of radioactivity) is reduced to half (50%). Some elements (like uranium) have long HLT (millions of years), some, like Co-60, have an average HLT of 5 years only, some, like I-131, has short HLT of 8 days. 33 Properties of alpha particles: An alpha particle is identical to a helium nucleus having two protons and two neutrons. It is a relatively heavy, high-energy particle, with a positive charge of +2 from its two protons. Alpha particles have a velocity in air of approximately one-twentieth the speed of light, depending upon the individual particle's energy. Conditions that lead to alpha particle emission: When the ratio of neutrons to protons in the nucleus is too low, certain atoms restore the balance by emitting alpha particles. For example: Polonium-210 has 126 neutrons and 84 protons, a ratio of 1.50 to 1. Following radioactive decay by the emission of an alpha particle, the ratio becomes 124 neutrons to 82 protons, or 1.51 to 1. Alpha emitting atoms tend to be very large atoms (that is, they have high atomic numbers). With some exceptions, naturally occurring alpha emitters have atomic numbers of at least 82 (the element lead). Radionuclide alpha emitters: There are many alpha emitting radioactive elements, both natural and manmade.: Alpha Emitters Atomic Number (Z) Americum-241 Plutonium-236 Uranium-238 Thorium-232 Radium-226 Radon-222 Polonium-210 95 94 92 90 88 86 84 How do you know you are near alpha emitters and alpha particles? You must have specialized equipment to detect alpha radiation. Generally, this equipment is expensive and requires an expert to operate it. The one alpha-emitting radionuclide that you can easily measure yourself is radon. Inexpensive home test kits are available to test your home for radon from hardware and often grocery stores. 34 How do you protect yourself and your family from alpha particles? Protecting yourself from external exposure to alpha radiation is easy, since alpha particles can not penetrate the outer dead layers of skin or clothing. However, tissue that is not protected by the outer layer of dead cells, such as eyes or open wounds, must be carefully protected. Exposure pathways of concern are inhalation or ingestion of alpha emitters, which continue to emit alpha particles. Alpha emitting radionuclides taken into the body release alpha particles directly to sensitive living tissues. As their high energy transfers directly to tissue, it causes damage that may lead to cancer. The most significant way people come in contact with alpha emitters is in their home, school, or place of business. Radon, is a heavy gas and tends to collect in low-lying areas such as basements. Testing for radon in your home and taking any corrective action necessary is the most effective way to protect you and your family from alpha emitters. Properties of beta particles: Beta particles are high-energy, high-speed electrons or positrons emitted by certain types of radioactive nuclei such as potassium-40. The beta particles emitted are a form of ionizing radiation also known as beta rays. The production of beta particles is termed beta decay. They are designated by the Greek letter beta (β). There are two forms of beta decay, β− and β+, which respectively give rise to the electron and the positron. Unstable atomic nuclei with an excess of neutrons may undergo β− decay, where a neutron is converted into a proton, an electron and an electron-type antineutrino (the antiparticle of the neutrino): This process is mediated by the weak interaction. The neutron turns into a proton through the emission of a virtual W− boson. At the quark level, W− emission turns a down-type quark into an up-type quark, turning a neutron (one up quark and two down quarks) into a proton (two up quarks and one down quark). The virtual W− boson then decays into an electron and an antineutrino. Beta decay commonly occurs among the neutron-rich fission byproducts produced in nuclear reactors. Free neutrons also decay via this process. This is 35 the source of the copious amount of electron antineutrinos produced by fission reactors. β+ decay (positron emission) Unstable atomic nuclei with an excess of inverse beta decay is a key step in the fusion processes that produce energy inside the sun. Properties of beta particles: Beta particles have an electrical charge of -1. Beta particles have a mass of 549 millionths of one atomic mass unit, or AMU, which is about 1/2000 of the mass of a proton or neutron. The speed of individual beta particles depends on how much energy they have, and varies over a wide range. It is their excess energy, in the form of speed, that causes harm to living cells. When transferred, this energy can break chemical bonds and form ions. Conditions leading to beta particle emission: Beta particle emission occurs when the ratio of neutrons to protons in the nucleus is too high. In this case, an excess neutron transforms into a proton and an electron. The proton stays in the nucleus and the electron is ejected energetically. 36 This process decreases the number of neutrons by one and increases the number of protons by one. Since the number of protons in the nucleus of an atom determines the element, the conversion of a neutron to a proton actually changes the radionuclide to a different element. Often, gamma ray emission accompanies the emission of a beta particle. When the beta particle ejection doesn't rid the nucleus of the extra energy, the nucleus releases the remaining excess energy in the form of a gamma photon. The decay of technetium-99, which has too many neutrons to be stable, is an example of beta decay. A neutron in the nucleus converts to a proton and a beta particle. The nucleus ejects the beta particle and some gamma radiation. The new atom retains the same mass number, but the number of protons increases to 44. The atom is now a ruthenium atom. Examples of beta emitters are phosphorous-32, tritium (H-3), carbon-14, strontium-90, and lead-210. Which radionuclides are beta emitters? There are many beta emitters: tritium cobalt-60 strontium-90 technetium-99 iodine-129 and -131 cesium-137 Beta Particles in the Environment: Applications of beta emitters: Beta emitters have many uses, especially in medical diagnosis, imaging, and treatment: 37 Iodine-131 is used to treat thyroid disorders, such as cancer and graves disease (a type of hyperthyroidism). Phosphorus-32 is used in molecular biology and genetics research. Strontium-90 is used as a radioactive tracer in medical/agricultural studies. Tritium is used for life science and drug metabolism studies to ensure the safety of potential new drugs. It is also used for luminous aircraft and commercial (Exit) signs, for luminous dials, gauges and wrist watches. Carbon-14 is a very reliable tool in dating of organic matter up to 30,000 years old. Beta emitters are also used in a variety of industrial instruments, such as industrial thickness gauges, using their weak penetrating power to measure very thin materials. Behavior of beta particles in the environment: Beta particles travel several feet in open air and are easily stopped by solid materials. When a beta particle has lost its energy, it is like any other loose electron. Whether in the outdoor environment or in the body, these electrons are then picked up by a positive ion. Man's exposure to beta particles: There are many both natural and man-made beta emitting radionuclides. Potassium-40 and carbon-14 are weak beta emitters that are found naturally in our bodies. Some decay products of radon emit beta particles, but its alphaemitting decay products pose a much greater health risk. Beta emitters that eject energetic particles can pose a significant problem to health. Their use requires special consideration of both benefits and potential, harmful effects. Key beta emitters used in medical imaging, diagnostic and treatment procedures are phosphorus-32, and iodine-131. For example, people who have taken radioactive iodine will emit beta particles. They must follow strict procedures to protect family members from exposure. Radioactive iodine may enter the environment during a nuclear reactor accident and find its way into the food chain. Industrial gauges and instruments containing concentrated beta-emitting radiation sources can be lost, stolen, or abandoned. If these instruments then enter the scrap metal market, or someone finds one, the sources they contain can expose people to beta emitters. Gamma Rays: 38 Gamma-rays have the smallest wavelengths and the most energy of any other wave in the electromagnetic spectrum. These waves are generated by radioactive atoms and in nuclear explosions. Gamma-rays can kill living cells, a fact which medicine uses to its advantage, using gamma-rays to kill cancerous cells. Gamma-rays travel to us across vast distances of the universe, and absorbed by the Earth's atmosphere. Different wavelengths of light penetrate the Earth's atmosphere to different depths. Instruments aboard high-altitude balloons and satellites like the Compton Observatory provide our only view of the gamma-ray sky. Gamma rays production: Gamma rays are often produced alongside other forms of radiation such as alpha or beta. When a nucleus emits an α or β particle, the daughter nucleus is sometimes left in an excited state. It can then jump down to a lower level by emitting a gamma ray in much the same way that an atomic electron can jump to a lower level by emitting visible light or ultraviolet radiation. Gamma rays, x-rays, visible light, and UV rays are forms of electromagnetic radiation. The only difference is the frequency and hence the energy of the photons. Gamma rays are the most energetic. An example of gamma ray production follows. First 60Co decays to excited 60Ni by beta decay: Then the 60Ni drops down to the ground state (see nuclear shell model) by emitting two gamma rays in succession: Gamma rays of 1.17 MeV and 1.33 MeV are produced. 39 A (Cobalt-60) radiotherapy unit. 40 LIGHT ENERGY AND OPTICS Light is a form of electromagnetic energy. It has a frequency, wavelength, and amplitude and can undergo reflection, refraction, interference, and polarization. It usually travels in vacuum (free-space) at the constant speed of ( c ) of 186,000 mi/s (3 x 108 m/s). Refraction: When light travels through a material (medium), its speed (v) will be less than in vacuum. Also, when it travels from a material with a certain refractive index (m1) to another with a refractive index (m2), it will be refracted (bent) toward the normal. Lenses refract the light to form images of different objects. A convex lens focuses (gathers) light at the focal length ( ), unit meter, on the centre line of the lens. A concave lens diverges light away from centreline. The lens strength = ( 1/ ). It has a unit of (dioptres). Convex lenses are +ve lenses, and concave lenses are –ve lenses. The cornea and the crystalline lens are the two lenses of the human eye. Normal focal length ( ) of the human eye when viewing distant objects is 2.2 cm. This, however, changes with age. The lens strength of the human eye is 45 dioptres. Total internal reflection of light occurs when sin > 1.0 Fibre optics principle depends on this law. Light is usually transmitted from one end of the fibre to the other end by total internal reflection. Fibre Optics are usually used in endoscopy, bronchoscopy, laryngoscopy gastroscopy, etc .. Common Vision Defects: Myopia (short-sight): Eye's focal length ( ) too short and falls (before) the retina. This is corrected by using a -ve lens (concave). Hyperopia (long-sight): ( ) too long and falls (in front of) the retina. Corrected by a +ve lens (convex). 41 Astigmatism: Occurs when the human eye lens is asymmetric (not symmetric) and is of non-ideal optical properties (e.g., its curvature). 42 ACOUSTICS (THE SCIENCE OF SOUND) Sound waves are longitudinal mechanical and compressional waves that can travel through solids, liquids, and gases by transmitting energy as by vibration (oscillation). Speed of light depends on the physical properties of the medium, for example, its density. At a boundary (called interface), sound is partially transmitted and partially reflected. The speed of sound in air is 344 m/s, but it different for different media through which the sound is travelling, e.g., the speed in body tissues is roughly 1540 m/s, in water it is 1498 m/s, and in glass it is 5170 m/s. Therefore, it is faster in the denser material (more in liquids than in gas). Sound intensity: Is measured in (decibels). Audiometry is defined as the testing for hearing loss as a function of frequency. It consists of: (1) (2) 43 Pure tone audiometry. Speech audiometry. ULTRASOUND Refers to sound waves of frequencies above 20 kHz (20,000 hertz). Pulsed ultrasound is clinically used for both diagnosis and therapy. An ultrasound probe (transducer) will transmit and receive ultrasound waves. It is made of a piezoelectric material. A-Mode Ultrasound: Is used to measure organ's thickness or depth, represented by vertical lines on the VDU (monitor) screen. . B-Moda Ultrasound: Uses a movable probe to produce bright spots (image) on the VDU screen. The Doppler-Ultrasound: Is used to scan organ's motion (like fetal heart beats and blood flow). 44