* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Acids, Bases and Salts
Chemical equilibrium wikipedia , lookup
Analytical chemistry wikipedia , lookup
Biological aspects of fluorine wikipedia , lookup
Nuclear chemistry wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Peptide synthesis wikipedia , lookup
Electrolysis of water wikipedia , lookup
Liquid–liquid extraction wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Determination of equilibrium constants wikipedia , lookup
Citric acid cycle wikipedia , lookup
Green chemistry wikipedia , lookup
Acid throwing wikipedia , lookup
Thermometric titration wikipedia , lookup
Nitrocellulose wikipedia , lookup
Nitric acid wikipedia , lookup
Genetic code wikipedia , lookup
Sulfuric acid wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Butyric acid wikipedia , lookup
Biosynthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Biochemistry wikipedia , lookup
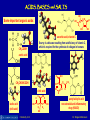
ACIDS, BASES and SALTS
For thousands of years people have known that
vinegar, lemon juice and many other foods taste
sour.
However, it was not until a few hundred years ago that it was discovered why these things taste
sour - because they are all acids.
The term acid, in fact, comes from the Latin term
acere, which means "sour".
Acids taste sour, are corrosive to metals,
change litmus (a dye extracted from lichens) red,
and become less acidic when mixed with bases.
Bases feel slippery, change litmus blue,
and become less basic when mixed with acids.
Acids react with bases to form salts.
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
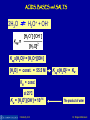
Arrhenius Acids:
Substances that when placed in water,
will dissociate to produce H+ ions:
HCl(aq) → H+(aq) + Cl-(aq)
1884
Arrhenius Bases:
Substances that when placed in water
will dissociate to yield OH- ions:
NaOH(aq) → Na+(aq) + OH-(aq)
Nitric Acid - HNO3
Chloric Acid - HClO3
Perchloric Acid - HClO4
Sulfuric Acid - H2SO4
Phosphoric Acid - H3PO4
Acetic Acid - HC2H3O2
Chemistry 21A
How these acids and
bases dissociate?
Swedish chemist Svante
Arrhenius, received the Nobel
Prize in Chemistry in 1903
One of the founders of the
science of Physical Chemistry
Potassium Hydroxide – KOH
Calcium Hydroxide - Ca(OH)2
Barium Hydroxide - Ba(OH)2
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
the Brønsted-Lowry theory is an acid-base theory, proposed independently by
Danish Johannes Nicolaus Brønsted and English Thomas Martin Lowry in 1923. In this
system, an acid is defined as any chemical species (molecule or ion) that is able
to lose, or "donate" a hydrogen ion (proton), and a base is a species with the
ability to gain or "accept" a hydrogen ion (proton). It follows that if a compound is
to behave as an acid, donating a proton, there must be a base to accept the proton. So
the Brønsted–Lowry concept can be defined by the reaction:
acid
+
base
conjugate base + conjugate acid
CH3CO2H +
H2O
CH3CO2-
+
H3O+
H2O +
NH3
OH-
+
NH4+
There is strong evidence
that the hydrogen ion is
never found free as H+.
The bare proton is so
strongly attracted
by the electrons of
surrounding water
molecules that H30+
forms immediately.
Brønsted Acids:
Any substance that can transfer a proton (H+) to another substance
Brønsted Bases:
Any substance that can accept a proton (H+) from another substance
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Hydrochloric Acid - HCl
Chloric Acid - HClO3
Perchloric Acid - HClO4
Sulfuric Acid - H2SO4
Phosphoric Acid - H3PO4
Acetic Acid - HC2H3O2
Chemistry 21A
Show acid/base
conjugate pairs.
Potassium Hydroxide – KOH
Calcium Hydroxide - Ca(OH)2
Barium Hydroxide - Ba(OH)2
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
AMPHIPROTIC
a compound or ion that can
either donate or accept H+ ions,
i.e. can act both as an acid and as a base
H2O, HSO4- , HPO42-, HSO3- etc.
HSO4- + H3O+
H2SO4 + H2O
HSO4- + OH-
SO42- + H2O
CH3CO2H +
H2 O
H2O + NH3
Chemistry 21A
CH3CO2OH-
+
+
H 3O+
NH4+
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Classical acid (and salt) naming system:
Anion (Salt)
Prefix
Anion (Salt)
Suffix
Acid Prefix Acid Suffix Example
per
ate
per
hypo
ic acid
perchloric acid (HClO4)
ate
ic acid
chloric acid (HClO3)
ite
ous acid
chlorous acid (HClO2)
ite
hypo
ous acid
hypochlorous acid
(HClO)
ide
hydro
ic acid
hydrochloric acid (HCl)
sulf
nitr
phosph
carbon
brom
iod
Chemistry 21A
sulfur
nitr
phosphor
carbon
brom
iod
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
fertilizers, explosives
Nitric Acid - HNO3
Strong Acids
Nitrous Acid - HNO2
Hydrohalic acids: HCl, HBr, HI
Hypochlorous Acid - HClO bleach
Nitric acid: HNO3
Chlorous Acid - HClO2
Sulfuric acid: H2SO4
Chloric Acid
- HClO3
Perchloric acid: HClO4
Perchloric Acid - HClO4
Sulfuric Acid - H2SO4
car batteries
Sulfurous Acid - H2SO3
involved metabolism such as adenosine diphosphate
Phosphoric Acid - H3PO4 (ADP) and triphosphate (ATP); DNA, RNA
Phosphorous Acid - H3PO3
soft drinks, seltzer water
Carbonic Acid - H2CO3
Acetic Acid - HC2H3O2
vinegar, pickles
Oxalic Acid - H2C2O4
kidney and bladder stones
Boric Acid - H3BO3
treatment of skin irritations; insecticide
Silicic Acid - H2SiO3
orthosilicic acid H4SiO4, is the form
predominantly absorbed by humans
and is found in numerous tissues
including bone, tendons, aorta, liver
and kidney.
Chemistry 21A
Hydrofluoric Acid - HF glass etching
Hydrochloric Acid - HCl stomach acid
Hydrobromic Acid - HBr
Hydroiodic Acid - HI
Hydrosulfuric Acid - H2S rotten eggs smell
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
SULFURIC ACID
World production in 2001 was 165 million tonnes,
Hot concentrated sulfuric acid is an oxidizing agent
with an approximate value of US$8 billion.
Fe(s) + 2H2SO4(conc)
FeSO4(aq) + 2H2O(l) + SO2(g)
Concentrated sulfuric acid is very good at removing the water from sugars.
C12H22O11(s) + nH2SO4 (l)
12C(s) + 11H2O (l) + nH2SO4(l)
Making hydrogen peroxide (H2O2) by reacting
barium peroxide with sulfuric acid.
BaO2(s) + H2SO4(aq)
Chemistry 21A
BaSO4(s) + H2O2(aq)
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
HO
Some important organic acids:
HO
O
C H
C
C H
H2
HO
O
OH
ascorbic acid, vitamin C
Scurvy is a disease resulting from a deficiency of vitamin C,
which is required for the synthesis of collagen in humans
CH3COOH
acetic acid
OH
O
CH3CHOHCOOH
HC
citric acid
O
lactic acid
HO
(milk acid)
O
OH
O
C
H2
C
H2
HO
Chemistry 21A
CH3
O
HC
C
H
CH
O
acetylsalicylic acid
OH
non-steroidal anti-inflammatory
drug (NSAID)
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
glycine
an α-amino acid, with
the amino group on the
left and the carboxyl
group on the right
L- and D-alanine
MONOSODIUM GLUTAMATE (MSG)
as a food ingredient has been the subject of health studies. A report from
the Federation of American Societies for Experimental Biology (FASEB)
compiled in 1995 on behalf of the FDA concluded that MSG was safe for
most people when "eaten at customary levels.
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
glycylglycine
glycine
glycine
When two amino acids
react (“head-to-tail”) they
form a peptide bond
(in reaction between the
acid of one molecule and
amine of another
molecule).
Thus, a PEPTIDE
(bond)
is formed,
A polypeptide is a chain of amino acids.
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Bases
lye, oven and drain cleaner Sodium Hydroxide - NaOH
Potassium Hydroxide - KOH
glass cleaner Ammonium Hydroxide - NH4OH
caustic lime, mortar, plaster Calcium Hydroxide - Ca(OH)
2
laxatives, antacids Magnesium Hydroxide - Mg(OH)2
Barium Hydroxide - Ba(OH)2
antacids, deodorants Aluminum Hydroxide - Al(OH)3
Ferrous Hydroxide or Iron (II) Hydroxide - Fe(OH)2
Ferric Hydroxide or Iron (III) Hydroxide - Fe(OH)3
an absorbent in surgical dressings Zinc Hydroxide - Zn(OH)2
Lithium Hydroxide - LiOH
Strong bases
Sodium Hydroxide - NaOH
Potassium Hydroxide – KOH
Calcium Hydroxide - Ca(OH)2
Barium Hydroxide - Ba(OH)2
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
CH3NH2
methylamine
CH3CH2NH2
ethylamine
atropine
Injections of ATROPINE are
used in the treatment of
bradychardia (an extremely
the first effective treatment for malaria
low heart rate)
Atropine occurs in the deadly
All organic bases (like
nightshade plant (Atropa belladonna)
inorganic ones) react
with acids to form salts.
VINCRISTINE, one of the most potent
ANTILEUKEMIC DRUGS in use today,
was isolated in a search for diabetes
treatments from Vinca rosea (now
Catharanthus roseus) in the 1950's
Chemistry 21A
Morphine, C17H19NO3,
is the most abundant of
opium’s 24 alkaloids,
accounting for 9 to 14%
morphine
of opium-extract by mass.
Named after the Roman god of dreams, Morpheus.
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
2H2O
Chemistry 21A
H3O+ + OH-
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
2H2O
H3O+ + OH-
Keqx[H2O]2 = [H3O+][OH-]
[H2O] = const. = 55.5 M
Keqx[H2O]2 = Kw
KW = const.
at 25oC
Kw = [H3O+][OH-] = 10-14
Chemistry 21A
The product of water
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
2H2O
H3O+ + OH-
Keqx[H2O]2 = [H3O+][OH-]
The product of water
if
[H3O+] = [OH-] = 10-7
THE SOLUTION IS
NEUTRAL
Chemistry 21A
T (°C)
0
5
20
70
100
at 25oC
Kw = [H3O+][OH-] = 10-14
Kw
0.114 × 10-14
0.186 × 10-14
0.681 × 10-14
15.85 × 10-14
51.3 × 10-14 12.29
pKw
14.94
14.73
14.17
12.80
6.14
neutral pH
7.47
7.37
7.08
6.40
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
H3O+ + OH-
2H2O
Keqx[H2
O]2
= [H3
O+][OH-]
[H2O+] = const. = 55.5 M
Keqx[H2O]2 = Kw
at 25oC
Kw = [H3
[H3
O+]
=
=
[OH-]
10-14
=
For convenience instead of
exponential numbers, negative
logarithms of these numbers are used.
pH = -log[H+] or -log[H3O+]
IN THE NEUTRAL
SOLUTION
at 25oC
pH = -log [1 x 10-7] = -(-7.00) = 7.00
KW = const.
O+][OH-]
THE pH CONCEPT
10-7
Chemistry 21A
[H3O+] = 10-pH
pOH = -log[OH-]
Dr. Dragan Marinkovic
[H+]
pH Example
1 x 100
0
HCl
1 x 10-1
1
Stomach acid
1 x 10-2
Acids
(acidic
1 x 10-3
Solutions) 1 x 10-4
2
Lemon juice
3
Vinegar
4
Soda (Coca-Cola)
1 x 10-5
5
Rainwater
1 x 10-6
6
Milk
1 x 10-7
7
Pure water
1 x 10-8
8
Egg whites
1 x 10-9
9
Baking soda
1 x 10-10
10 Tums® antacid
Neutral
Bases
(basic
1 x 10-11
Solutions) 1 x 10-12
11 Ammonia
12 Mineral lime - Ca(OH)2
1 x 10-13
13 Drano®
1 x 10-14
14 NaOH
Chemistry 21A
ACIDS,
BASES
and
SALTS
pH = -log [H+]
Note: concentration is commonly
abbreviated by using square brackets, thus
[H+] = hydrogen ion concentration.
When measuring pH, [H+] is in units of
moles of H+ per liter of solution.
The pH of blood is maintained
within the narrow range of
7.35 to 7.45.
Normal urine pH averages about 6.0.
Saliva has a pH between 6.0 and 7.4.
Tear pH was measured in 44 normal subjects.
The normal pH range was
6.5 to 7.6; the mean value was 7.0.
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
pH in living systems
Chemistry 21A
Compartment
pH
Gastric acid
0.7
Lisosomes
4.5
Granules of chromaffin cells
5.5
Urine
6.0
Neutral H2O at 37 °C
6.81
Cytosol
7.2
Cerebrospinal fluid (CSF)
7.3
Blood
7.34 – 7.45
Mitochondrial matrix
7.5
Pancreas secretions
8.1
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Calculate the molar concentration of H3O+ in water
solutions with the following OH- molar concentrations:
a) 6.9x10-5
b) 0.074
Calculate the molar concentration of OH- in water
solutions with the following H3O+ molar concentrations:
a) 0.0087
b) 9.9x10-10
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Calculate the molar concentration of H3O+ in water
solutions with the following OH- molar concentrations:
a) 6.9x10-5
b) 0.074
Kw = [H3O+][OH-] = 10-14
[H3O+] = 10-14/[OH-] = 10-14/[6.9x10-5] = 1.449x10-10 M
[H3O+] = 10-14/[OH-] = 10-14/[7.4x10-2] = 1.35x10-13 M
Calculate the molar concentration of OH- in water
solutions with the following H3O+ molar concentrations:
a) 0.0087
b) 9.9x10-10
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Calculate the molar concentration of H3O+ in water
solutions with the following OH- molar concentrations:
a) 6.9x10-5
b) 0.074
Kw = [H3O+][OH-] = 10-14
[H3O+] = 10-14/[OH-] = 10-14/[6.9x10-5] = 1.449x10-10 M
[H3O+] = 10-14/[OH-] = 10-14/[7.4x10-2] = 1.35x10-13 M
Calculate the molar concentration of OH- in water
solutions with the following OH- H3O+ molar concentrations:
a) 0.0087
b) 9.9x10-10
[OH-] = 10-14 /[H3O+] = 10-14/[8.7x10-3] = 1.15x10-12 M
[OH-] = 10-14 /[H3O+] = 10-14/[9.9x10-10] = 1x10-5 M
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Determine the pH of water solutions
with the following characteristics.
Classify each solution as acidic, basic
or neutral.
Convert the following pH values
in both [H+] and [OH-] values.
Chemistry 21A
a) [H+] = 7.5x10-6
b) [OH-] = 2.5x10-4
c) [OH-] = 8.6x10-10
a) pH = 3.95
b) pH = 4.00
c) pH = 11.86
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Determine the pH of water solutions
with the following characteristics.
Classify each solution as acidic, basic
or neutral.
a) [H+] = 7.5x10-6
b) [OH-] = 2.5x10-4
c) [OH-] = 8.6x10-10
pH = -log [H+]
pOH = -log [OH-]
pH + pOH = 14
a) pH = -log [H+] = -log(7.5x10-6) = 5.12
b) pOH = -log [OH-] = -log(2.5x10-4) = 3.6
Convert the following pH values
in both [H+] and [OH-] values.
Chemistry 21A
pH = 14 - pOH = 14 - 3.6 = 10.4
a) pH = 3.95
b) pH = 4.00
c) pH = 11.86
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Determine the pH of water solutions
with the following characteristics.
Classify each solution as acidic, basic
or neutral.
a) [H+] = 7.5x10-6
b) [OH-] = 2.5x10-4
c) [OH-] = 8.6x10-10
pH = -log [H+]
pOH = -log [OH-]
pH + pOH = 14
pH = -log [H+] = -log(7.5x10-6) = 5.12
pOH = -log [OH-] = -log(2.5x10-4) = 3.6
Convert the following pH values
in both [H+] and [OH-] values.
a) pH = 3.95
b) pH = 4.00
c) pH = 11.86
[H3O+] = 10-pH = 10-3.95 = 1.12x10-4
Chemistry 21A
pH = 14 - pOH = 14 - 3.6 = 10.4
[H3O+] = 10-pH
[OH-] = 10-pOH
[OH-] = 10-pOH = 10-10.05 = 8.91x10-11
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
pH meter
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Properties of Acids
Types of Acids
Monoprotic - a solution that produces one mole of H+ ions per mole of acid HCl , HNO3
Diprotic - a solution that produces two moles of H+ ions per mole of acid H2SO4
Triprotic - a solution that produces three moles of H+ ions per mole of acid H3PO4
Polyprotic - two ore more H+ per mole of acid
STRONG ACIDS
Acids that are essentially 100%
Ionized in aqueous solutions
Hydrohalic acids: HCl, HBr, HI
Nitric acid: HNO3
Sulfuric acid: H2SO4
Perchloric acid: HClO4
Chemistry 21A
WEAK ACIDS
Acids that are partially ionized
(usually less than 5%) in equilibrium.
Nitrous Acid - HNO2
Sulfurous Acid - H2SO3
Phosphorous Acid - H3PO3
Carbonic Acid - H2CO3
Acetic Acid - HC2H3O2
Boric Acid - H3BO3
Silicic Acid - H2SiO3
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Properties of Acids
Acids react with:
Metals
Metal oxides
Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g)
FeO(s) + 2HCl(aq) → FeCl2(aq) + H2O(l)
Hydroxides/bases Al(OH)3(s) + 3CH3COOH(aq) → Al(CH3COO)3(aq) + 3H2O(l)
Carbonates 3CaCO3(s) + 2H3PO4(aq) → Ca3(PO4)2(aq) + 3CO2(g) + 3H2O(l)
Bicarbonates KHCO3(aq) + HNO3(aq) → KNO3(aq) + CO2(g) + H2O(l)
Strong acids react with salts
of weak acids
Na3BO3(aq) + 3HBr(aq) → H3BO3(aq) + 3NaBr(aq)
The major products of al these reactions
(metal compounds with acids) are SALTS.
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Properties of Acids
Making dilutions from stock solutions:
If we have 1 L of 3 M HCl, what is M if we dilute acid to 6 L?
M1 = 3 mol/L, V1 = 1 L, V2 = 6 L
M1V1 = M2V2
M1V1/V2 = M2
M2 = (3 mol/L x 1 L) / (6 L) = 0.5 M
Why does the formula work?
Because we are equating mol to mol: M1V1 = 3 mol M2V2 = 3 mol
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Properties of Acids
What volume of 0.5 M HCl can be prepared from 1 L of 12 M HCl?
M1 = 12 mol/L, V1 = 1 L, M2 = 0.5 L
M1V1 = M2V2
M1V1/M2 = V2
V2 = (12 mol/L x 1 L) / (0.5 L) = 24 L
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Properties of Acids
1. How many mL of a 14 M stock solution must be used to make 250 mL of a 1.75 M
solution?
2. You have 200 mL of 6.0 M HF. What concentration results if this is diluted to a
total volume of 1 L?
3. 100 mL of 6.0 M CuSO4 must be diluted to what final volume so that the resulting
solution is 1.5 M?
4. What concentration results from mixing 400 mL of 2.0 M HCl with 600 mL of 3.0 M
HCl?
5. What is the concentration of NaCl when 3 L of 0.5 M NaCl are mixed with 2 L of
0.2 M NaCl?
6. What is the concentration of NaCl when 3 L of 0.5 M NaCl are mixed with 2 L of
water?
7. Water is added to 4 L of 6 M antifreeze until it is 1.5 M. What is the total volume of
the new solution?
8. There are 3 L of 0.2 M HF. 1.7 L of this is poured out, what is the concentration of
the remaining HF?
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Properties of Bases
Slimy or soapy feel on fingers, due to
saponification of the lipids in human skin.
Concentrated or strong bases are
caustic (corrosive) on organic matter
and react violently with acidic substances
Strong Bases
NaOH - Sodium Hydroxide
KOH - Potassium Hydroxide
Ca(OH)2 - Calcium Hydroxide
Ba(OH)2 - Barium Hydroxide
NaOH(s) + H2O(l) → Na+(aq) + OH-(aq)
Weak Bases
Aluminum Hydroxide - Al(OH)3
Iron (II) Hydroxide - Fe(OH)2
Iron (III) Hydroxide - Fe(OH)3
Zinc Hydroxide - Zn(OH)2
Bases react with:
Acids Al(OH)3(s) + 3CH3COOH(aq) → Al(CH3COO)3(aq) + 3H2O(l)
“Acidic oxides” 2NaOH(aq) + CO2(g) → Na2CO3(aq) + H2O(l)
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Salts
In medicine, saline (also saline solution) is a
general term referring to a sterile solution of
sodium chloride (table salt) in water. It is used for
intravenous infusion, rinsing contact lenses, and
nasal irrigation.
In medicine, normal saline (NS) is the
commonly-used term for a solution of 0.91%
w/v of NaCl, about 300 mOsm/L. Less
commonly, this solution is referred to as
physiological saline or isotonic saline,
Saline solution for
intravenous infusion. The
white port at the base of the
bag is where additives can
be injected with a
hypodermic needle. The
port with the blue cover is
where the bag is spiked
with an infusion set.
Chemistry 21A
NaCl(s) + H2O(l) → Na+(aq) + OH-(aq)
NaOH
HCl
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Salts
According to chemistry, the term "salt" is used for ionic compounds that is composed
of positively charged cations (usually metal or ammonium ions) and the negatively
charged anions, so that the product remains neutral and without a net charge.
The anions may be inorganic (Cl-) as well as organic (CH3COO-)
and monoatomic (F-) as well as polyatomic ions (SO42-).
Salt's solution in water is called electrolytes.
Both, the electrolytes and molten salts conduct electricity.
Salts with OH- are basic salts (CaOHCl, BaOHNO3)
and with H+ are acidic salts (NaHSO4).
Usually salts are solid crystals having high melting point.
Taste - It differes from salt to salt. It can elicit all the five basic tastes, like
salty (sodium chloride),
sweet (lead diacetate very toxic!),
sour (potassium bitartrate),
bitter (magnesium sulfate),
and umami or savory (monosodium glutamate).
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Salts
Silver
electroplating.
The anode is a
silver bar and
the cathode is
an iron spoon.
anode
cathode
+
Anode: The positive terminal of an electrical current flow.
Cathode: The negative terminal of an electric current system.
-
Diagram of a Hoffman voltameter used
for the electrolysis of water to produce
hydrogen and oxygen gases
2H2O → 2H2 + O2
Cathode (reduction): 2H+(aq) + 2e− → H2(g)
Anode (oxidation): 2H2O(l) → O2(g) + 4H+(aq) + 4e
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Salts
Anode: The positive terminal of an electrical current flow.
Anions – negative ions – in aqueous solutions
move towards ANODE, e.g. Cl-, NO3-, SO42CATIONS – positive (usually metal) ions, in aqueous solutions
move towards CATHODE, K+, Al3+, Ba2+
Cathode: The negative terminal of an electric current system.
EQUIVALENT OF SALT is the amount that will produce 1 mol
of positive electrical charge when dissolved and dissociated.
Number of salt equivalents in
1 L of 1 M of MgCl2 is 2(+) x 1 M = 2 eq
Chemistry 21A
Number of salt equivalents in
3.12x10-2 mol of Fe(NO3)3 is
3(+) x 3.12x10-2 = 9.36x10-2 eq = 93.6 meq
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Salts
When crysralluized from aqueous solutions
many salts crystallise as hydrates:
CuSO4•5H2O - copper (II) sulfate pentahydrate
CoCI2•6H2O - cobalt (II) chloride hexahydrate
SnCl2•2H2O - stannous (tin II) chloride dihydrate
When heated, these salts lose their
crystalline water and become
“anhydrous salts”.
HYDRATE
is a salt containing specific numbers of
water molcules as part of solid crystalline structure.
CuSO4•5H2O
CoCI2•6H2O
CuSO4
WATER OF HYDRATION is water retained as
part of the solid crystalline structure of some salts.
CoCI2
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Salts
Metal + nonmetal
Syntheses of salts –
reactions yielding salts
Metals
Metal oxides
Fe(s) + S(s) → FeS(s)
Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g)
FeO(s) + 2HCl(aq) → FeCl2(aq) + H2O(l)
Hydroxides/bases Al(OH)3(s) + 3CH3COOH(aq) → Al(CH3COO)3(aq) + 3H2O(l)
Carbonates 3CaCO3(s) + 2H3PO4(aq) → Ca3(PO4)2(aq) + 3CO2(g) + 3H2O(l)
Bicarbonates KHCO3(aq) + HNO3(aq) → KNO3(aq) + CO2(g) + H2O(l)
Strong acids react with salts
of weak acids
Na3BO3(aq) + 3HBr(aq) → H3BO3(aq) + 3NaBr(aq)
Two soluble salts when mixed forming a new insoluble salt
BaCl2(aq) + K2SO4(aq) → BaSO4(s) + 2KCl(aq)
“Acidic oxides” 2NaOH(aq) + CO2(g) → Na2CO3(aq) + H2O(l)
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
The strength of Acids and Bases
Types of Acids
Monoprotic - a solution that produces one mole of H+ ions per mole of acid HCl , HNO3
Diprotic - a solution that produces two moles of H+ ions per mole of acid H2SO4
Triprotic - a solution that produces three moles of H+ ions per mole of acid H3PO4
Polyprotic - two ore more H+ per mole of acid
WEAK ACIDS
Acids that are partially ionized
(usually less than 5%) in equilibrium.
STRONG ACIDS
Acids that are essentially 100%
Ionized in aqueous solutions
Hydrohalic acids: HCl, HBr, HI
Nitric acid: HNO3
Sulfuric acid: H2SO4
Perchloric acid: HClO4
Chemistry 21A
% dissociation
moderately
weak
6.7 Nitrous Acid - HNO
2
34 Sulfurous Acid - H2SO3
Phosphorous Acid - H3PO3
0.2 Carbonic Acid - H2CO3
1.3 Acetic Acid - HC2H3O2
0.01 Boric Acid - H3BO3
Silicic Acid - H2SiO3
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
The strength of Acids and Bases
ACID DISSOCIATION CONSTANT
The equilibrium constant for
the dissociation of an acid.
HA + H2O
A− + H3O+
CH3COOH + H2O
CH3COO- + H3O+
n all but the most concentrated solutions it can be assumed
that the concentration of water, [H2O], is constant,
approximately 55 mol·dm−3. On dividing K by the constant
terms and writing [H+] for the concentration of the hydronium
ion the expression
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
The strength of Acids and Bases
Polyprotic acids are acids that can lose more than one proton.
The constant for dissociation of the first proton may be
denoted as Ka1 and the constants for dissociation of
successive protons as Ka2, etc.
Phosphoric acid, H3PO4, is an example of a
polyprotic acid as it can lose three protons.
equilibrium
H3PO4
H2PO4− + H+
H2PO4−
HPO42− + H+
HPO42−
Chemistry 21A
PO43− + H+
Ka1
Ka2
Ka3
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
The strength of Acids and Bases
Slimy or soapy feel on fingers, due to
saponification of the lipids in human skin.
Concentrated or strong bases are
caustic (corrosive) on organic matter
and react violently with acidic substances
NaOH(s) + H2O(l) → Na+(aq) + OH-(aq)
Strong Bases
NaOH - Sodium Hydroxide
KOH - Potassium Hydroxide
Ca(OH)2 - Calcium Hydroxide
Ba(OH)2 - Barium Hydroxide
Chemistry 21A
Weak Bases
Aluminum Hydroxide - Al(OH)3
Iron (II) Hydroxide - Fe(OH)2
Iron (III) Hydroxide - Fe(OH)3
Zinc Hydroxide - Zn(OH)2
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
The strength of Acids and Bases
NH3(aq) + H2O
BASE DISSOCIATION CONSTANT
The equilibrium constant for
the dissociation of a base.
NaOH(aq)
NH4+(aq) + OH-(aq)
Na+(aq) + OH-(aq)
B + H 2O
HB+ + OH−
Using similar reasoning to that used
before
In water, the concentration of the
hydroxide ion, [OH−], is related to the
concentration of the hydrogen ion by
Kw = [H+][OH−], therefore
Substitution of the expression for
[OH−] into the expression for Kb give
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Analyzing Acids and Bases
Titration is a common laboratory method
of quantitative chemical analysis
that is used to determine the unknown
concentration of a known reactant.
Because volume measurements play a key
role in titration, it is also known as volumetric analysis
The EQUIVALENCE POINT,
or STOICHIOMETRIC POINT,
of a chemical reaction occurs
during a chemical titration
when the amount of titrant
added is equivalent, or equal,
to the amount of analyte
present in the sample.
known volume of
unknown
concentration (of acid)
Titration curve of a strong base titrating a strong acid
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Analyzing Acids and Bases
The volume of titrant added from the buret is measured. For our example,
lets assume that 18.3 mL of 0.115 M NaOH has been added to 25.00 mL of
nitric acid solution. The following setup shows how the molarity of the
nitric acid solution can be calculated from this data.
= or 0.0842 M HNO3
ENDPOINT - The volume or amount of
acid or base added to a solution to
neutralize the unknown solution during
a titration. When using an indicator, the
endpoint occurs when enough
titrant has been added to change
the color of the indicator.
Titration curve of a strong base titrating a strong acid
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Analyzing Acids and Bases
pH meter
ENDPOINT - The volume or
amount of acid or base added to a To determine endpoint indicators can be used
solution to neutralize the unknown (paper or soluble indicator dyes) or a pH meter
solution during a titration.
Indicator
Low pH color
Transition pH range High pH color
Gentian violet (Methyl violet)
yellow
0.0–2.0
blue-violet
Thymol blue (first transition)
red
1.2–2.8
yellow
Thymol blue (second transition)
yellow
8.0–9.6
blue
Methyl orange
red
3.1–4.4
orange
Bromocresol purple
yellow
5.2–6.8
purple
Bromothymol blue
yellow
6.0–7.6
blue
Phenol red
yellow
6.8–8.4
red
Cresol Red
yellow
7.2–8.8
reddish-purple
Phenolphthalein
colorless
8.3–10.0
fuchsia
Alizarine Yellow R
yellow
10.2–12.0
red
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Titration Calculations
HNO3(aq) + NaOH(aq)
NaNO3(aq) + H2O(l)
acid/base
molar ratio
1 : 1
H2SO4(aq) + 2NaOH(aq)
Na2SO4(aq) + 2H2O(l)
1 : 2
H3PO4(aq) + 3NaOH(aq)
Na3PO4(aq) + 3H2O(l)
1 : 3
M1V1 = M2V2
30 mL of 0.10 M NaOH neutralized 25.0 mL of hydrochloric acid.
Determine the concentration of the acid
NaOH(aq) + HCl(aq) -----> NaCl(aq) + H2O(l)
0.03 L x 0.1 mol/L = 0.025 L x M2
M2 = 0.003 mol/0.035 L = 0.12 mol/L
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Titration Calculations
50 mL of 0.2 mol L-1 NaOH neutralized 20 mL of sulfuric acid.
Determine the concentration of the acid
1.Write the balanced chemical equation for the reaction
NaOH(aq) + H2SO4(aq) -----> Na2SO4(aq) + 2H2O(l)
2.Extract the relevant information from the question:
NaOH
V = 50mL, M = 0.2M
H2SO4
V = 20mL, M = ?
3.Check the data for consistency
NaOH
V = 50 x 10-3L, M = 0.2M
H2SO4
V = 20 x 10-3L, M = ?
4.Calculate moles NaOH
n(NaOH) = M x V = 0.2 x 50 x 10-3 = 0.01 mol
5.From the balanced chemical equation find the mole ratio
NaOH:H2SO4
2:1
6.Find moles H2SO4
NaOH: H2SO4 is 2:1
So n(H2SO4) = ½ x n(NaOH) = ½ x 0.01 = 5 x 10-3 moles H2SO4 at the
equivalence point
7.Calculate concentration of H2SO4: M = n ÷ V
n = 5 x 10-3 mol,
V = 20 x 10-3L
M(H2SO4) = 5 x 10-3 ÷ 20 x 10-3 = 0.25M or 0.25 mol L-1
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Hydrolysis Reactions of Salts
Hydrolysis is a chemical reaction with water.
CH3COONa(aq) + H2O(l)
Salts of weak acids and/or bases
hydrolyze in aqueous solutions.
CH3COOH(aq) + Na+(aq) + OH-(aq)
basic pH
NH4Cl(aq) + H2O(l)
NH4OH(aq) + H3O+(aq) + Cl-(aq)
acidic pH
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
BUFFER
A solution with the ability to resist changing pH
when acids (H+) or bases (OH-) are added.
BUFFERS usually consist of a pair of compounds
one of which has the ability to react with H+
and the other with the ability to react with OH-.
CH3COOH(aq) + OH-(aq)
CH3COO-(aq) + H+(aq)
CH3COO-(aq) + H2O(l)
CH3COOH(aq)
This is how a buffer solution
resists changes ion pH
Chemistry 21A
BUFFER CAPACITY
the amount of acid (H+)
that can be absorbed by
a buffer without causing a
significant change in pH.
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
Antacids create
buffered solutions
In blood plasma, the carbonic acid and
hydrogen carbonate ion equilibrium buffers the pH.
In this buffer, carbonic acid (H2CO3)
is the hydrogen-ion donor (acid) and
hydrogen carbonate ion (HCO3-) is
the hydrogen-ion acceptor (base).
H2CO3(aq)
Al(OH)3
Wyeth amphojel tablets
of aluminum hydroxide
CaCO3
Chemistry 21A
H+(aq) + HCO3-(aq)
Additional H+ is consumed by HCO3and additional OH- is consumed by H2CO3.
The value of Ka for this equilibrium is 7.9 × 10-7.
The pH of arterial blood plasma is 7.40.
If the pH falls below this normal value,
a condition called acidosis is produced.
If the pH rises above the normal value,
he condition is called alkalosis.
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
The equilibrium
constant will be:
In 1 L of solution, there are going to be about 55 moles of water.
Kc (a constant) times the concentration of water
(another constant) on the left-hand side.
The product of those is then given the name Ka.
An introduction to pKa
pKa bears exactly the same relationship to Ka as pH does to the hydrogen ion concentration:
The higher the value for pKa, the weaker the acid.
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
acid
Ka (mol dm-3) pKa
hydrofluoric acid
5.6 x 10-4
3.3
formic acid
1.6 x 10-4
3.8
acetic acid
1.7 x 10-5
4.8
hydrogen sulfide
8.9 x 10-8
7.1
Phosphoric acid, H3PO4, is
an example of a polyprotic acid
as it can lose three protons.
equilibrium
H3PO4
Chemistry 21A
H2PO4− + H+
pKa value
pKa1 = 2.15
H2PO4−
HPO42− + H+
pKa2 = 7.20
HPO42−
PO43− + H+
pKa3 = 12.37
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
[base]
pKa
=
pH
-
log
-------------
[acid]
pH = -log[H+] or -log[H3O+]
same equation
Henderson-Hasselbalch equation
Relationship between the pH, pKa
and the concentrations of acid
and base in the buffer.
Chemistry 21A
[base]
pH
=
pKa
+
log
-------------
[acid]
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
If we need to make
a buffer solution
of a certain pH, we would usually select an acid with the
pKa near the desired pH and then adjust the concentration
of the acid and the conjugate base (the anion of the acid)
to give the desired pH. We can assume that the amount of
acid that dissociates is very small and can be neglected.
This means that the buffer concentration of the acid
and the anion are “equal” to made-up concentrations.
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
Calculate the pH of buffers that contain the acid and conjugate base in following concentrations:
a) [HPO42-] = 0.33 M, [PO43-] = 0.52 M
pH = pKa + log{[PO43-]/[PO42-]} = 12.66 + log(0.52 M/0.33 M) = 12.66 + 0.20 = 12.86
b) [CH3COOH] = 0.40 M, [CH3COO-] = 0.25
pH = pKa + log[CH3COO-]/[CH3COOH] = 4.74 + log(0.25 M/0.40 M) = 4.74 - 0.2 = 4.54
What ratio of concentrations of NaH2PO4 and Na2HPO4 in solution
would give a buffer with pH = 7.65?
pH = pKa + log{[PO42-]/[PO4-]}
7.65 = 7.21 + log{[PO42-]/[PO4-]}
Chemistry 21A
log{[PO42-]/[PO4-]} = 0.44
[PO42-]/[PO4-] = 100.44 = 2.75
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
1.00 mol of ammonium chloride is added to 2.00 L of 1.00 mol.L-1 NH3(aq)
(Kb = 1.8 x 10-5 mol.L-1)
a. Calculate the pH of the solution.
b. 10.00 mL of the solution is placed into a 1000 mL volumetric flask and
made up to the mark with pure water. What is the pH of this solution?
[acid]
pOH
=
pKb
+
log
-------------
[base]
NH4+(aq) + H2O(l)
H3O+(aq) + NH3(aq)
[acid] = [NH4+] = 1 mol/2 L = 0.5 mol/L
[base] = [NH3] = [NH4OH] = 1 mol/L
pOH = -log(1.8x10-5mol/L) + log(0.5/1 molL-1)
pOH = 4.74 - 0.30 = 4.44
pH = 14 - pOH = 9.56
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
1.00 mol of ammonium chloride is added to 2.00 L of 1.00 mol.L-1 NH3(aq)
(Kb = 1.8 x 10-5 mol.L-1)
a. Calculate the pH of the solution.
b. 10.00 mL of the solution is placed into a 1000 mL volumetric flask and
made up to the mark with pure water. What is the pH of this solution?
b. Buffer solution diluted 1:100 with pure water, ratio of conjugate acid
and base remains unchanged. Therefore pH is unchanged. pH = 9.56
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
Chemistry 21A
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
1.00 mol of ammonium chloride is added to 2.00 L of 1.00 mol.L-1 NH3(aq)
(Kb = 1.8 x 10-5 mol.L-1)
a. Calculate the pH of the solution.
b. 10.00 mL of the solution is placed into a 1000 mL volumetric flask and
made up to the mark with pure water. What is the pH of this solution?
c. 10.0 mL of 0.100 mol.L-1 HCl(aq) is added to 100 mL of each of the
above solutions. What is the final pH in each case?
d. 1.00 mL of 0.100 mol.L-1 NaOH(aq) is added to fresh 100 mL portions
of each solution. What is the final pH in each case?
[NH4+] = 1.00 mol/2.00 L = 0.500 mol.L-1,
[NH3] = 1.00 mol.L-1 (given).
neutralization reaction:
HCl(aq) + NH3(aq) ==> NH4+(aq) + Cl-(aq) (complete)
i.e. 0.0010 mol of NH3 will be converted to [NH4ˆ+]
100 mL of solution contain:
amount of NH4+ = 0.500 mol.L-1 x 0.100 L = 0.0500 mol
NH4+(aq) + H2O(l) <===> H3O+(aq) + NH3(aq)
amount of NH3 = 1.00 mol.L-1 x 0.100 L = 0.100 mol
init/mol 0.0500
0.100 ch/mol
amount of HCl = 0.100 mol.L-1 x 0.010 L = 0.0010 mol
+0.0010
-0.0010
0.0510
final/mol
0.099
pH = pKa - log10(a/b) = -log10{Kw/Kb} - log10{[NH4+]/[NH3]}
= 9.26 - log10{0.0051/0.099} = 9.26 + 0.29 => pH = 9.55
Chemistry 21A
14 - 4.74 = 9.26
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
1.00 mol of ammonium chloride is added to 2.00 L of 1.00 mol.L-1 NH3(aq)
(Kb = 1.8 x 10-5 mol.L-1)
a. Calculate the pH of the solution.
b. 10.00 mL of the solution is placed into a 1000 mL volumetric flask and
made up to the mark with pure water. What is the pH of this solution?
c. 10.0 mL of 0.100 mol.L-1 HCl(aq) is added to 100 mL of each of the
above solutions. What is the final pH in each case?
d. 1.00 mL of 0.100 mol.L-1 NaOH(aq) is added to fresh 100 mL portions
of each solution. What is the final pH in each case?
solution b. We are adding the same amount of HCl to a solution which is
100 times more dilute, i.e. contains 0.0100 times the amounts of NH4+ & NH3:
amount of NH4+ = 0.0500 mol x 0.00100 = 0.000500 mol The amounts of HCl and NH3 are equal;
we no longer have a buffer, but exact
amount of NH3 = 0.100 mol x 0.00100 = 0.00100 mol
neutralization, since all the NH3 is converted
amount of HCl = 0.100 mol.L-1 x 0.010 L = 0.0010 mol
to NH4+ we have 0.00150 mol of NH4+ in 110 mL
[NH4+] = 0.00150 mol/0.110 L = 0.0136 mol.L-1
of solution. This is a solution of a weak acid.
Ka = Kw/Kb = 1.00 x 10-14/1.8 x 10-5
= 5.6 x 10-10
Chemistry 21A
Since [NH4+]>>Ka we can use the approximation [NH4+]>>[H3O+]
(sqrt = square root.)
[H3O+] = sqrt(C0.Ka) =sqrt(0.00150 x 5.6 x 10-10) mol.L-1
= 9.17 x 10-7 => pH = 6.04
Dr. Dragan Marinkovic
ACIDS, BASES and SALTS
Buffers
1.00 mol of ammonium chloride is added to 2.00 L of 1.00 mol.L-1 NH3(aq)
(Kb = 1.8 x 10-5 mol.L-1)
a. Calculate the pH of the solution.
b. 10.00 mL of the solution is placed into a 1000 mL volumetric flask and
made up to the mark with pure water. What is the pH of this solution?
c. 10.0 mL of 0.100 mol.L-1 HCl(aq) is added to 100 mL of each of the
above solutions. What is the final pH in each case?
d. 1.00 mL of 0.100 mol.L-1 NaOH(aq) is added to fresh 100 mL portions
of each solution. What is the final pH in each case?
d. (Outline solution only) Similar to c, but a smaller amount of base added.
The neutralisation reaction is now:
NaOH(aq) + NH4+(aq) ---> Na+(aq) + NH3(aq) + H2O(l) (complete)
i. solution a: amount of NH4+ = 0.0500 mol
amount of NH3 = 0.100 mol
amount of NaOH = 0.00010 mol
ii. solution b amount of NH4+ = 0.00050 mol
+
+
NH4 (aq) + H2O(l) <===> H3O (aq) + NH3(aq)
amount of NH3 = 0.00100 mol
init/mol 0.0500 0.100 ch/mol -0.0001 +0.0001
amount of NaOH = 0.00010 mol still a buffer;
final/mol 0.0499 0.100 pH = 9.57
similar to above final [NH +] = 0.00040 mol ; f
4
inal [NH3] =0.00110 mol pH = 9.73
Chemistry 21A
Dr. Dragan Marinkovic