* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Lymphopoiesis wikipedia , lookup
Molecular mimicry wikipedia , lookup
Immune system wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Hepatitis B wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Adaptive immune system wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Innate immune system wikipedia , lookup
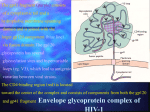
Recent Advances in Antiretroviral Therapy Hail M. Al-Abdely, MD Consultant, Infectious Diseases Rationale for Drug Combinations CD4 + cell NRTIs Work here NNRTIs Work here PIs Work here Development of AIDS is like an impending train wreck Viral Load = Speed of the train CD4 count = Distance from cliff HIV infection J. Coffin, XI International Conf. on AIDS, Vancouver, 1996 Viral Dynamics of HIV-1 Infection Latently infected CD4 lymphocytes Productively infected CD4 lymphocytes <1% T 1/2 ~1.6d 99% Uninfected CD4 lymphocytes 2.6 days per generation T1/2 ~5.7 hrs HIV <1% Uninfected activated CD4 lymphocytes Long-lived cell populations Perelson et.al. Science 271:1582 (1996) CD4 lymphocytes infected with defective virus Viral dynamics it takes 2.6 days to produce a new generation of viral particles estimated total HIV production is 10.3 x 109 virions per day at least 99% of the virus pool is produced by recently infected cells retroviral therapy should be able to reduce viral load within a few days GOALS OF THERAPY • Clinical goals: Prolongation of life and improved quality of life • Virologic goals: Reduction in viral load as much as possible for as long as possible to: 1) halt disease progression, and 2) prevent/reduce resistant variants • Immunologic goals: Achieve immune reconstitution that is quantitative (CD4 to normal range) and qualitative (pathogen-specific immune response) • Therapeutic goals: Rational sequencing of drugs in a fashion that achieves virologic goals, but also: 1) maintains therapeutic options; 2) is relatively free of side effects; and 3) is realistic in terms of probability of adherence • Epidemiologic goals: Reduce HIV transmission Antiretroviral Drugs Approved by FDA for HIV Generic Name Class Firm FDA Approval Date zidovudine, AZT NRTI Glaxo Wellcome March 87 didanosine, ddI NRTI Bristol Myers-Squibb October 91 zalcitabine, ddC NRTI Hoffman-La Roche June 92 stavudine, d4T NRTI Bristol Myers-Squibb June 94 lamivudine, 3TC NRTI Glaxo Wellcome November 95 saquinavir, SQV, hgc PI Hoffman-La Roche December 95 ritonavir, RTV PI Abbott Laboratories March 96 indinavir, IDV PI Merck & Co., Inc. March 96 nevirapine, NVP NNRTI Boehringer Ingelheim June 96 nelfinavir, NFV PI Agouron Pharmaceuticals March 97 delavirdine, DLV NNRTI Pharmacia & Upjohn April 97 zidovudine and lamivudine NRTI Glaxo Wellcome September 97 saquinavir, SQV, sgc PI Hoffman-La Roche November 97 efavirenz, EFV NNRTI DuPont Pharmaceuticals September 98 abacavir, ABC NRTI Glaxo Wellcome February 99 amprenavir PI Glaxo Wellcome April 99 Deaths per 100,000 Population Trends in Age-Adjusted* Rates of Death due to HIV Infection, USA, 1982-1998 18 16 14 12 10 8 6 4 2 0 82 84 86 88 *Using the age distribution of the projected year 2000 US population as the standard. 90 Year 92 94 96 **Preliminary 1998 data 98** Good News Highly active antiretroviral therapy has Changed our view toward HIV from inevitably fatal to a manageable disease over several decades Bad News 1. 2. 3. 4. 5. Incomplete response Complexity of treatment Short and long term side effects Cross resistance Drug-drug interactions Bad News 1. Incomplete response 2. Complexity of treatment 3. Short and long term side effects 4. Cross resistance 5. Drug-drug interactions Bad News 1. Incomplete response 2. Complexity of treatment 3. Short and long term side effects 4. Cross resistance 5. Drug-drug interactions Bad News 1. Incomplete response 2. Complexity of treatment 3. Short and long term side effects 4. Cross resistance 5. Drug-drug interactions Bad News 1. 2. 3. 4. Incomplete response Complexity of treatment Short and long term side effects Cross resistance 5. Drug-drug interactions Bad News 1. Incomplete response • • 2. 3. 4. 5. Complete RNA suppression and sustained CD4 increase happens only in 60-80%. Effectiveness is even lower in patients with high replication rates and extensive antiretroviral experience. Complexity of treatment Short and long term side effects Cross resistance Drug-drug interactions Immunotherapy Immunotherapy • Directions – Augmentation of specific immune response to control viral replication. – Preventive Vaccines. Clues to immune control of HIV? Subject JP: Subject 161J: Sx: Dx: Sx: Dx: F/U: Fever, Rash, Headache HIV ELISA Neg. HIV RNA >700,000 Extensive Rx AIDS at 11 mo. Rapid CD4 cell decline Viral Load >750,000 F/U: Fever, Rash, Headache HIV ELISA Neg. HIV ELISA Pos. No Rx Well at 19 yrs. CD4 1000 Viral Load < 500 Acute HIV-1 infection Stimulation of HIV-1-specific immune CD4 cells (Helper cells) Generation of HIV-1-specific killer cells (CTL) Loss of CTL function due to inadequate HIV-1-specific helper cells Infection of activated helper cells Loss of HIV-1-specific helper cells Progression T helper cells are the central orchestrator of the immune system CTL Function T helper cell B Cell Function Antibody Production NK Cell Function APC Function Cytokine production Acute HIV-1 infection Stimulation of HIV-1-specific immune CD4 cells (Helper cells) Antiviral Rx Generation of HIV-1-specific killer cells (CTL) Maintenance of CTL function due to adequate HIV-1-specific helper cells Nonprogression Protection of activated helper cells Maintenance of HIV-1specific helper cells HIV-1-specific T helper cells in individuals treated during acute infection (n=7) 100 0 Months 2 Months 10 1 MB JC KM ND SJ DK KS Barriers to the Development of an Effective AIDS Vaccine • Sequence variation • Protective immunity in natural infection not clearly established • Desire to achieve sterilizing immunity • Lack of adequate animal model to study vaccine protection with HIV • Latency and integration of HIV into host genome • Transmission by cell-associated virus • Limited knowledge about mucosal transmission and immune responses • Financial disincentives • Ethical issues