* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 2 - Yale University
Survey
Document related concepts
Molecular orbital diagram wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Marcus theory wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Franck–Condon principle wikipedia , lookup
Thermodynamics wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Atomic theory wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Hypervalent molecule wikipedia , lookup
George S. Hammond wikipedia , lookup
Self-assembly of nanoparticles wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Computational chemistry wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Transcript
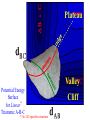
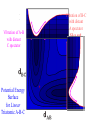
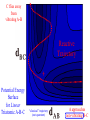
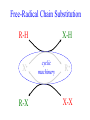
Chem 125 Lecture 37 12/10/08 This material is for the exclusive use of Chem 125 students at Yale and may not be copied or distributed further. It is not readily understood without reference to notes or the wiki from the lecture. Exponents & Three Flavors of Statistics “there’s a divinity that shapes our ends” 1)Same The Boltzmann Factor e thing: Hamlet V:2 k is per individual molecule R is per mole (= k NA) R -H/RT from counting random arrangements of a fixed number of energy bits k =W 2) The Entropy Factor eTS/kT from counting W, the number of molecular structures being grouped 3) The Law of Mass Action Cyclohexane Conformers 10.8 7.0 kcal/mole 5.5 few quantum states 0 few "structures" Chair (stiff) many "structures" many quantum states Twist-Boat (flexible) few quantum states Both classical and quantum views suggest a statistical "entropy" factor (of ~7) favoring twist-boat. This reduces the room-temperature Boltzmann "enthalpy" bias of 10-(3/4) 5.5 = 14,000 in favor of chair to about 2,000. few "structures" Chair (stiff) Experimental Entropy Although we discuss entropy theoretically (in statistical terms), physical chemists can measure it experimentally. The entropy of a perfectly ordered crystalline material at zero Kelvin is zero ( ln 1 ). As the material is warmed it gains entropy in increments of (Heat Absorbed)/Temperature. S = H/T “Floppy” molecules with closely spaced energy levels absorb more energy, and at lower temperatures, and thus gain more S on warming. Cf. Ethane rotation - Lecture 31 K = e-G/RT Exponents & Three Flavors of Statistics 1) The Boltzmann Factor e -H/RT from counting random arrangements of a fixed number of energy bits 2) The Entropy Factor eTS/kT = W weighted from counting W, the number of quantum states being grouped 3) The Law of Mass Action from counting molecules per volume Law of Mass Action Late 1700s : Attempts to assemble. hierarchy of “Affinities” Early 1800s : Amounts [concentration] can shift reaction direction away. from “affinity” prediction. … Mid 1800s : Equilibrium “K” as balance of forward and reverse rates... Law of Mass Action [concentration] 2A [A2] 2 [A] A2 = K [A2] = K [A] 2 Where does the exponent come from? Randomly Distributed “Particles” # Particles # Dimers 50 1 100 9 150 19 200 35 250 59 # of Dimers Randomly Distributed “Particles” Increasing concentration increases both the number number of units and the fraction fraction of units that have near neighbors. [D] = K # of Particles # Particles # Dimers [P] 2 50 1 100 9 150 19 200 35 250 59 Equilibrium, Statistics & Exponents Particle Distribution : Law of Mass Action [A2] = K 2 [A] Energy Distribution : H , Boltzmann Factor -H/RT Ke Counting Quantum States : S S/R Ke Free energy determines what can happen (equilibrium) -G/RT e K= -(3/4)G = 10 Energy & Entropy kcal/mole @ room Temp But how quickly will it happen? (kinetics) Visualizing Reaction Classical Trajectories & The Potential Energy Surface Potential Energy Rolling Ball Maps A-B Vibration A-B Distance Potential Energy “Surface” for Stretching Diatomic Molecule A-B Plateau Pass + (Transition State) Potential Energy Surface for Linear * Triatomic A-B-C * So 2-D specifies structure Valley Cliff Vibration of A-B with distant C spectator Potential Energy Surface for Linear Triatomic A-B-C Vibration of B-C with distant A spectator Slice and fold back Unreactive Trajectory: (A bounces off vibrating B-C) Potential Energy Surface for Linear Triatomic A-B-C C flies away from vibrating A-B Reactive Trajectory Potential Energy Surface for Linear Triatomic A-B-C “classical” trajectory (not quantum) A approaches non-vibrating B-C H3 Surface Henry Eyring (1935) Transition State (“Lake Eyring”) Crazy angle of axes means that classical trajectories can be modeled by rolling marble. H + H-Br Studying Lots of Random Trajectories Provides Too Much Detail Summarize Statistically with Collective Enthalpy (H) & Entropy (S) “steepest descent” path (not a trajectory) Slice along path, then flatten and tip up to create… Transition “State” G Starting Materials Products “Reaction Coordinate” Diagram (for a one-step atom transfer) Not a trajectory, but a sequence of three species each with H and S, i.e. Free Energy (G) Free Energy determines what can happen (equilibrium) -G/RT Since the transition state e (universal) K= -(3/4)G = 10 the velocity is not universal, Velocity and of ts theory is not truly in equilibrium kcal/mole with starting materials, and @ room Temp the theory is approximate. how rapidly (kinetics) ‡ 13 -G /RT 10 e k (/sec) = 13-(3/4)G = 10 ‡ Amount of ts kcal/mole @ room Temp Using Energies to Predict Equilibria and Rates for One-Step Reactions: Free-Radical Halogenation "free-radical chain" • Cl • Cl H CH3 • CH3 Cl Cl H Cl • Cl CH3Cl Are Average Bond Energies “Real” or just a trick for reckoning molecular enthalpy ? Bond Dissociation Energies are real. BondDissn Energies 115 84 85 72 72 58 57 99 111 113 90 89 89 105 111 127 85 85 97 74 74 73 84 63 59 72 57 56 67 51 46 54 122 85 123 136.2 91 92 94 best values as of 2003 Ellison I Larger halogen Poorer overlap with H (at normal bond distance) & less e-transfer to halogen H • H • • • •• F I •• less e-stabilization weaker bond Diagram qualitative; not to scale. All H-Alkyl 100 ± 5 Same trend as H-Halogen Special Cases Ellison II C-H bond unusually strong (good overlap from sp2C) Ditto hard Vinyl 111 No special stabilization SOMO orthogonal to *) hard Phenyl Ditto 113 Are unusual BDE values due to unusual bonds or unusual radicals? C-H bond normal (sp3C , as in alkane) Ditto easy Allyl 89 easy Benzyl 90 Special stabilization SOMO overlaps *) SOMOC • Ditto •• • •• or actually •• Possibility of Halogenation (Equilibrium) H3C H + X X H3C X + H X Cost F 105 37 142 115 Cl ” 58 163 84 Br ” 46 151 72 I ” 36 141 58 136 103 88 71 Return Profit 251 187 160 129 109 19 9 12 Possibility of Halogenation How about rate(Equilibrium) (which depends on Mechanism)? H3C• •H + X• •X H3C X + H X Cost F 105 37 142 115 Cl ” 58 163 84 Br ” 46 151 72 I ” 36 141 58 136 103 88 71 Return Profit 251 187 160 129 109 19 9 12 Is break-two-bonds-then-make-two a plausible Mechanism? at RT (~300K)? 1013 10-106 = 10-93/sec No Way! at ~3000K? 1013 10-10.6 = 250/sec Yes (unless there is a faster one) Henry Eyring H2 H Dissociation followed H by association requires high activation energy. SLOW (1935) Make-as-you-break “displacement” is much easier. FAST HHH H H2 H H Free-Radical Chain Substitution R-H X• R-X X-H cyclic machinery R• X-X Possibility of Halogenation (Mechanism for Reasonable Rate) (Equilibrium) H3C• X• H3C-H + XHX X2 + H33CX 2 HX Cost Step 1 31 136 F 105 136 37 142 37 2 103 Cl ” 103 58 163 58 88 17 46 88 Br ” 46 151 71 34 36 71 I ” 36 141 115 84 72 58 Return Step 2 Profit 78 251 26 187 26 160 22 129 109 24 9 12 How can we predict activation energy? Physical Organic Chemistry Paul D. Bartlett 1907-1997 http://osulibrary.oregonstate.edu/specialcollections/coll/pauling/bond/audio/1997v.1-bookdunitz.html 1939 Jack Dunitz: At the time when I was reading that book I was wondering whether chemistry was really as interesting as I had hoped it was going to be. And I think I was almost ready to give it up and do something else. I didn't care very much for this chemistry which was full of facts and recipes and very little thought in it, very little intellectual structure. And Pauling's book gave me a glimpse of what the future of chemistry was going to be and particularly, perhaps, my future. The Chemical Bond Is there an Atomic Force Law? Feeling & Seeing Molecules and Bonds Understanding Bonding & Reactivity through H = E How chemists learned to treasure Composition, Constitution, Configuration, Conformation and Energy Some Big Questions: The Chemical Bond Is there an Atomic Force Law? How does science know? Feeling & Seeing Molecules and Bonds Compared to what? Understanding Bonding & Reactivity Were chemical through H = bonds E discovered or invented? How chemists learned to treasure Composition, Constitution, Would Configuration, we even have chemical bonds Conformation without our own forbearers? andchemical Energy End of Lecture 37 Dec. 10, 2008 Good Luck on the Final!